1,2,3,4-四氢咔唑 | 942-01-8
中文名称
1,2,3,4-四氢咔唑
中文别名
2,3,4,9-四氢-1H-咔唑;四氢咔唑
英文名称
1,2,3,4-tetrahydrocarbazole
英文别名
2,3,4,9-tetrahydro-1H-carbazole;tetrahydrocarbazole;1,2,3,4-tetrahydro-9H-carbazole;6,7,8,9-tetrahydrocarbazole;1,2,3,4,10,11-hexahydrocarbazole
CAS
942-01-8
化学式
C12H13N
mdl
MFCD00004959
分子量
171.242
InChiKey
XKLNOVWDVMWTOB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:118-120 °C (lit.)
-
沸点:325-330 °C (lit.)
-
密度:1.1459 (rough estimate)
-
闪点:325-330°C
-
保留指数:1809;1775.8
-
稳定性/保质期:
在常温常压下保持稳定,应避免与氧化物直接接触。
计算性质
-
辛醇/水分配系数(LogP):3.3
-
重原子数:13
-
可旋转键数:0
-
环数:3.0
-
sp3杂化的碳原子比例:0.333
-
拓扑面积:15.8
-
氢给体数:1
-
氢受体数:0
安全信息
-
危险品标志:Xi
-
安全说明:S26,S37/39
-
危险类别码:R36/37/38
-
WGK Germany:2
-
海关编码:2933990090
-
RTECS号:FE6300000
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:请将容器密封保存,并存放在阴凉、干燥的地方。
SDS
| Name: | 1 2 3 4-Tetrahydrocarbazole 99% Material Safety Data Sheet |
| Synonym: | 2,3-Tetramethyleneindole; 1H-Carbazole, 2,3,4,9-Tetrahydro- (9CI); 5,6,7,8-Tetrahydrocarbazole |
| CAS: | 942-01-8 |
Synonym:2,3-Tetramethyleneindole; 1H-Carbazole, 2,3,4,9-Tetrahydro- (9CI); 5,6,7,8-Tetrahydrocarbazole
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 942-01-8 | 1,2,3,4-Tetrahydrocarbazole | 99% | 213-385-7 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Runoff from fire control or dilution water may cause pollution.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 942-01-8: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: beige
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 325 - 330 deg C @ 760.00mmHg
Freezing/Melting Point: 118.00 - 120.00 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C12H13N
Molecular Weight: 171.24
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 942-01-8: FE6300000 LD50/LC50:
CAS# 942-01-8: Oral, rat: LD50 = 2650 mg/kg.
Carcinogenicity:
1,2,3,4-Tetrahydrocarbazole - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 942-01-8: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 942-01-8 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 942-01-8 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,3,4,9-四氢-1H-咔唑-1-酮 1-oxo-2,3,4,9-tetrahydrocarbazole 3456-99-3 C12H11NO 185.225 —— 8-bromo-1,2,3,4-tetrahydro-1H-carbazole 27372-41-4 C12H12BrN 250.138 四氢咔唑酮 1,2,3,9-tetrahydro-4H-carbazol-4-one 15128-52-6 C12H11NO 185.225 —— 3-phenylseleno-2,3,4,9-tetrahydro-1H-carbazole 1000683-01-1 C18H17NSe 326.3 —— 4,9-dihydro-1H-carbazole 21865-49-6 C12H11N 169.226 —— 1,2,3,4-Tetrahydro-6-chlor-carbazol-2-carbonsaeure 36077-02-8 C13H12ClNO2 249.697 9-乙烯基-2,3,4,9-四氢-1H-咔唑 9-vinyl-2,3,4,9-tetrahydro-1H-carbazole 60507-53-1 C14H15N 197.28 —— N-allyl-1,2,3,4-tetrahydrocarbazole 51281-98-2 C15H17N 211.307 —— 9-tert-butyl-2,3,4,9-tetrahydro-1H-carbazole 1131890-57-7 C16H21N 227.349 9-苄基-2,3,4,9-四氢-1H-咔唑 9-benzyl-2,3,4,9-tetrahydro-1H-carbazole 17017-63-9 C19H19N 261.367 9-乙酰基-2,3,4,9-四氢-1H-咔唑 N-acetyl-1,2,3,4-tetrahydrocarbazole 13815-69-5 C14H15NO 213.279 —— 1-(1,2,3,4-tetrahydrocarbazol-9-yl)propan-1-one 847355-63-9 C15H17NO 227.306 —— 1,2,3,4-tetrahydrocarbazol-9-carboxylic acid ethyl ester 33568-45-5 C15H17NO2 243.305 —— 1,2,3,4-tetrahydrocarbazol-9-carboxylic acid tert-butyl ester 98598-88-0 C17H21NO2 271.359 —— N-trifluoroacetyl-1,2,3,4-tetrahydrocarbazole 79096-94-9 C14H12F3NO 267.251 —— benzyl 3,4-dihydro-1H-carbazole-9(2H)-carboxylate 1197055-30-3 C20H19NO2 305.376 —— 9-tosyl-2,3,4,9-tetrahydro-1H-carbazole 205642-01-9 C19H19NO2S 325.431 - 1
- 2
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-羟基-5,6,7,8-四氢咔唑 5,6,7,8-tetrahydro-carbazol-2-ol 13314-79-9 C12H13NO 187.241 (6,7,8,9-四氢-5H-咔唑-3-基)甲胺 6-Aminomethyl-1,2,3,4-tetrahydrocarbazole 76061-94-4 C13H16N2 200.283 6,7,8,9-四氢-5H-咔唑-3-胺 2,3,4,9-tetrahydro-1H-carbazol-6-amine 65796-52-3 C12H14N2 186.257 —— 1,2,3,4-tetrahydro-6-hydroxycarbazole 13314-76-6 C12H13NO 187.241 —— 1-methyl-2,3,4,9-tetrahydro-1H-carbazole 17058-12-7 C13H15N 185.269 —— 7-acetyl-1,2,3,4-tetrahydrocarbazole 80053-43-6 C14H15NO 213.279 3-(三氟甲基)-6,7,8,9-四氢-5H-咔唑 6-(trifluoromethyl)-2,3,4,9-tetrahydro-1H-carbazole 2805-84-7 C13H12F3N 239.24 1-乙基-1,2,3,4-四氢咔唑 1-ethyl-1,2,3,4-tetrahydrocarbazole 10257-86-0 C14H17N 199.296 —— 1-propyl-1,2,3,4-tetrahydrocarbazole 1140-49-4 C15H19N 213.323 —— 1-butyl-1,2,3,4-tetrahydrocarbazole —— C16H21N 227.349 —— 1-pentyl-1,2,3,4-tetrahydrocarbazole —— C17H23N 241.376 —— 2-chloro-N-(5,6,7,8-tetrahydro-carbazol-3-ylmethyl)-acetamide 70780-94-8 C15H17ClN2O 276.766 2,3,4,9-四氢-1H-咔唑-1-酮 1-oxo-2,3,4,9-tetrahydrocarbazole 3456-99-3 C12H11NO 185.225 2,3,4,9-四氢-1H-咔唑-1-醇 1-hydroxy-1,2,3,4-tetrahydrocarbazole 1592-62-7 C12H13NO 187.241 —— 9-amino-(1,2,3,4-tetrahydrocarbazole) 67638-19-1 C12H14N2 186.257 —— N-(5,6,7,8-tetrahydro-carbazol-3-yl)-acetamide 109690-31-5 C14H16N2O 228.294 4-羟基-1,2,3,4-四氢咔唑 4-Hydroxy-1,2,3,4-tetrahydrocarbazol 82260-33-1 C12H13NO 187.241 —— 7-Nitro-1,2,3,4-tetrahydro-carbazol 3920-69-2 C12H12N2O2 216.239 3-硝基-5,6,7,8-四氢-9h-咔唑 6-nitro-2,3,4,9-tetrahydro-1H-carbazole 50823-86-4 C12H12N2O2 216.239 四氢咔唑酮 1,2,3,9-tetrahydro-4H-carbazol-4-one 15128-52-6 C12H11NO 185.225 5-羟基-2,3,4,9-四氢咔唑 5-Hydroxy-1,2,3,4-tetrahydro-carbazol 35618-96-3 C12H13NO 187.241 2,3,4,9-四氢-9-甲基-1H-咔唑 9-methyl-2,3,4,9-tetrahydro-1H-carbazole 6303-88-4 C13H15N 185.269 —— 1-acetyl-1,2,3,4-tetrahydro-9H-carbazole 335330-07-9 C14H15NO 213.279 —— 1-methoxy-2,3,4,9-tetrahydro-1H-carbazole 42540-57-8 C13H15NO 201.268 2,3,4,9-四氢-1H-咔唑-8-羧酸 1,2,3,4-tetrahydrocarbazole-8-carboxylic acid 65764-56-9 C13H13NO2 215.252 —— 1-azido-2,3,4,9-tetrahydro-1H-carbazole 61186-49-0 C12H12N4 212.254 —— 1,2,3,4-tetrahydrocarbazole-9-carboxaldehyde 78153-11-4 C13H13NO 199.252 9-乙烯基-2,3,4,9-四氢-1H-咔唑 9-vinyl-2,3,4,9-tetrahydro-1H-carbazole 60507-53-1 C14H15N 197.28 9-乙基-1,2,3,4-四氢咔唑 9-ethyl-1,2,3,4-tetrahydrocarbazole 37391-52-9 C14H17N 199.296 —— N-(5,6,7,8-tetrahydro-carbazol-3-ylmethyl)-phthalimide 68375-17-7 C21H18N2O2 330.386 —— 1,2,3,4-tetrahydro-1,9-dimethylcarbazole 50527-95-2 C14H17N 199.296 —— 1-Benzylamino-1,2,3,4-tetrahydrocarbazol 167476-72-4 C19H20N2 276.381 —— tetrahydro-N-propylcarbazole 37842-83-4 C15H19N 213.323 —— N-dichloromethyl-1,2,3,4-tetrahydrocarbazole 1392276-07-1 C13H13Cl2N 254.159 —— N-allyl-1,2,3,4-tetrahydrocarbazole 51281-98-2 C15H17N 211.307 1,2,3,4-四氢-9H-咔唑-9-乙醇 9-(β-Hydroxyethyl)-1,2,3,4-tetrahydrocarbazole 178174-35-1 C14H17NO 215.295 3-(1,2,3,4-四氢咔唑-9-基)丙腈 3-<1,2,3,4-Tetrahydro-carbazol-9-yl>-propionsaeure-nitril 24115-51-3 C15H16N2 224.305 9-(1,2,3,4-四氢咔唑-9-基甲基)-1,2,3,4-四氢咔唑 9,9'-methylenebis(2,3,4,9-tetrahydro-1H-carbazole) 72755-27-2 C25H26N2 354.495 —— 1,2,3,4-tetrahydro-9-phenyl-9H-carbazole 86844-02-2 C18H17N 247.34 —— 9-phenylethyl-1,2,3,4-tetrahydrocarbazole 38679-87-7 C20H21N 275.393 —— N-(4-fluorophenyl)-2,3,4,9-tetrahydro-1H-carbazol-1-amine 1446264-50-1 C18H17FN2 280.345 —— 9-methyl-6-nitro-2,3,4,9-tetrahydro-1H-carbazole 39581-69-6 C13H14N2O2 230.266 —— 4-(2,3,4,9-tetrahydro-1H-carbazol-1-ylamino)benzonitrile 1446264-38-5 C19H17N3 287.364 —— 1-(3,5-dihydroxy-2,4-dimethyl-6-((2,3,4,9-tetrahydro-1H-carbazol-7-yl)methyl)phenyl)propan-2-one —— C24H27NO3 377.483 —— 9-(2-diethylamino-ethyl)-1,2,3,4-tetrahydro-carbazole 25150-80-5 C18H26N2 270.418 9-苄基-2,3,4,9-四氢-1H-咔唑 9-benzyl-2,3,4,9-tetrahydro-1H-carbazole 17017-63-9 C19H19N 261.367 3-(1,2,3,4-四氢-9H-咔唑-9-基)丙酸 3-<1,2,3,4-Tetrahydro-carbazol-9-yl>-propionsaeure 23690-80-4 C15H17NO2 243.305 2-(1,2,3,4-四氢咔唑-9-基)乙酸 1,2,3,4-Tetrahydro-9-carbazolyl-essigsaeure 21144-93-4 C14H15NO2 229.279 1,2,3,4-四氢-9-甲基-4H-咔唑酮 9-methyl-1,2,3,9-tetrahydro-4H-carbazol-4-one 27387-31-1 C13H13NO 199.252 —— N,N-diethyl-2-[2-(1,2,3,4-tetrahydrocarbazol-9-yl)ethoxy]ethanamine —— C20H30N2O 314.471 —— 9-[2-(1H-tetrazol-5-yl)ethyl]-2,3,4,9-tetrahydro-1H-carbazole 872407-32-4 C15H17N5 267.333 —— N-(4-nitrophenyl)-2,3,4,9-tetrahydro-1H-carbazol-1-amine 1446264-37-4 C18H17N3O2 307.352 —— N-(4-phenylphenyl)-2,3,4,9-tetrahydro-1H-carbazol-1-amine 1541202-79-2 C24H22N2 338.452 —— 4-[2-[2-(1,2,3,4-Tetrahydrocarbazol-9-yl)ethoxy]ethyl]morpholine —— C20H28N2O2 328.455 —— 9-(4-chlorophenyl)-2,3,4,9-tetrahydro-1H-carbazole 55542-91-1 C18H16ClN 281.785 —— 1,3-bis(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 343805-70-9 C27H30N2O 398.548 —— 9-(2-(2-(1-Piperidinyl)ethoxy)ethyl)-2,3,4,9-tetrahydro-1H-carbazole —— C21H30N2O 326.482 —— 1,2,3,4-tetrahydrocarbazole-9-carboxamide 67242-61-9 C13H14N2O 214.267 9-乙酰基-2,3,4,9-四氢-1H-咔唑 N-acetyl-1,2,3,4-tetrahydrocarbazole 13815-69-5 C14H15NO 213.279 —— 9-(4-methoxyphenyl)-2,3,4,9-tetrahydro-1H-carbazole 55542-90-0 C19H19NO 277.366 —— N-oxiranylmethyl-1,2,3,4-tetrahydrocarbazole 77233-17-1 C15H17NO 227.306 —— phenyl (4-(2,3,4,9-tetrahydro-1H-carbazol-1-ylamino)phenyl)methanone 1446264-51-2 C25H22N2O 366.462 —— 1-(butylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-08-4 C19H28N2O 300.444 —— 9-(3-methoxyphenyl)-2,3,4,9-tetrahydro-1H-carbazole 64678-63-3 C19H19NO 277.366 —— 1-(1-pyrrolidinyl)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-12-0 C19H26N2O 298.428 —— 1-(cyclopentylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-09-5 C20H28N2O 312.455 2-氯-1-(1,2,3,4-四氢-9H-咔唑-9-基)乙酮 2-chloro-1-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)ethanone 43170-54-3 C14H14ClNO 247.724 —— 1-(1-piperidinyl)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 5679-70-9 C20H28N2O 312.455 —— 1-(benzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 300590-63-0 C22H26N2O 334.461 —— 1-(hexahydro-1H-azepin-1-yl)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-13-1 C21H30N2O 326.482 —— 1-(phenylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-10-8 C21H24N2O 320.434 —— 1,2,3,4-tetrahydrocarbazol-9-carboxylic acid ethyl ester 33568-45-5 C15H17NO2 243.305 —— 1-(4-methylbenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-16-4 C23H28N2O 348.488 —— 3-((E)-3-(5,6,7,8-tetrahydrocarbazol-9-yl)prop-1-enyl)oxazolidin-2-one 1213231-04-9 C18H20N2O2 296.369 —— 6-nitro-9-propyl-2,3,4,9-tetrahydro-1H-carbazole 1427185-18-9 C15H18N2O2 258.32 —— N-cyclohexyl-2-(1,2,3,4-tetrahydrocarbazol-9-yl)acetamide —— C20H26N2O 310.439 —— allyl 3,4-dihydro-1H-carbazole-9(2H)-carboxylate 1423039-17-1 C16H17NO2 255.316 —— 2,2-dimethyl-1-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)propan-1-one 78839-82-4 C17H21NO 255.36 —— 1-(3-methylbenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-15-3 C23H28N2O 348.488 —— 1-(4-chlorobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-30-2 C22H25ClN2O 368.906 —— 1-[(3-pyridylmethyl)amino]-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-38-0 C21H25N3O 335.449 —— 1-(4-bromobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-33-5 C22H25BrN2O 413.357 —— 1,2,3,4-tetrahydrocarbazol-9-carboxylic acid tert-butyl ester 98598-88-0 C17H21NO2 271.359 —— N-trifluoroacetyl-1,2,3,4-tetrahydrocarbazole 79096-94-9 C14H12F3NO 267.251 —— 1-(2-methylbenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-14-2 C23H28N2O 348.488 —— 9-[2-methoxy-3-(1-piperidinyl)propyl]-2,3,4,9-tetrahydro-1H-carbazole 1346265-07-3 C21H30N2O 326.482 —— N-(11-bromoundecanoyl)-1,2,3,4-tetrahydro-9H-carbazole 156609-55-1 C23H32BrNO 418.417 —— 5-oxo-5-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)pentanoic acid 156609-57-3 C17H19NO3 285.343 —— 1-(3-bromobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-32-4 C22H25BrN2O 413.357 —— 1-(3-chlorobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-29-9 C22H25ClN2O 368.906 苯基(1,2,3,4-四氢咔唑-9-基)甲酮 9-Benzoyl-1,2,3,4-tetrahydro-carbazol 13815-70-8 C19H17NO 275.35 —— 1-(2-bromobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-31-3 C22H25BrN2O 413.357 —— benzyl 3,4-dihydro-1H-carbazole-9(2H)-carboxylate 1197055-30-3 C20H19NO2 305.376 —— 1-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-3-[4-(trifluoromethyl)benzylamino]-2-propanol 1346265-36-8 C23H25F3N2O 402.46 —— 1-(4-methoxybenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-21-1 C23H28N2O2 364.488 —— 9-trans-cinnamoyl-1,2,3,4-tetrahydro-carbazole 102478-79-5 C21H19NO 301.388 —— 1-(2-chlorobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-28-8 C22H25ClN2O 368.906 —— 9-(4-methoxybenzyl)-1-methyl-1,2,3,4-tetrahydro-9H-carbazole 1313809-66-3 C21H23NO 305.42 —— 1-(2,6-difluorobenzylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-43-7 C22H24F2N2O 370.442 —— 1-(1-methyl-1-phenylethylamino)-3-(1,2,3,4-tetrahydro-9H-carbazol-9-yl)-2-propanol 1346265-11-9 C24H30N2O 362.515 - 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
反应信息
-
作为反应物:描述:参考文献:名称:Nakazaki; Isoe, Nippon Kagaku Zasshi, 1955, vol. 76, p. 1159,1167摘要:DOI:
-
作为产物:描述:参考文献:名称:费歇尔合成困难的新方法:在受控的微波辐射下在三甘醇中使用氯化锌催化剂。摘要:[反应:请参见文字]。三乙二醇与催化量的氯化锌(ZnCl2 / TEG)的应用被描述为一种难于进行费歇尔合成的新型高效反应介质,可导致敏感的吲哚。3-乙酰基-1-甲基硫代环烷基[c]吡啶苯基和对甲氧基苯基hydr转化为2-(2-吡啶基)吲哚和5-甲氧基-2-(2-吡啶基)吲哚,这是我们总的合成子sempervirine型生物碱的合成,是在受控的微波辐射下,在TEG中的干燥氯化锌溶液(0.16 M)中进行的。该方案以优异的收率通过苯乙酮或苯乙酮从苯乙酮或环己酮生产吲哚。DOI:10.1021/ol052255t
-
作为试剂:描述:呋喃 、 间氨基苯甲酸 在 tetrafluoroboric acid 、 sodium nitrite 、 1,2,3,4-四氢咔唑 作用下, 以 水 、 丙酮 、 二甲基亚砜 为溶剂, 反应 15.0h, 生成 3-(2-呋喃基)苯甲酸参考文献:名称:咔唑基电子给体受体(EDA)催化合成联芳基和芳基-杂芳基化合物摘要:描述了一种高度区域选择性的,基于咔唑的电子给体受体(EDA)催化的联芳基和芳基-杂芳基化合物的合成。筛选了各种吲哚和咔唑衍生物进行均相芳香取代(HAS)反应。四氢咔唑(THC)对于HAS转化非常有效,并通过重氮盐与富电子的四氢咔唑之间的络合物形成进行。紫外-可见光谱技术已用于确认复合物的形成。在原位发现即使催化量也能生成的EDA络合物对于单电子转移(SET)过程非常有效,并且没有任何光活化作用。联芳基化合物,2-苯基呋喃,2-苯基噻吩和2-苯基吡咯以及生物活性化合物(例如丹特林和canagliflozin)的合成产率中等至优异。DOI:10.1039/d0ob00282h
文献信息
-
[EN] METHODS OF TREATMENT OF AMYLOIDOSIS USING ASPARTYL-PROTEASE INIHIBITORS<br/>[FR] PROCEDES DE TRAITEMENT D'AMYLOIDOSE UTILISANT DES INHIBITEURS DE PROTEASE ASPARTYLE申请人:ELAN PHARM INC公开号:WO2005070407A1公开(公告)日:2005-08-04The invention relates to acetyl 2-hydroxy-1,3-diaminospirocyclohexanes and derivatives thereof that are useful in treating diseases, disorders, and conditions associated with amyloidosis. Amyloidosis refers to a collection of diseases, disorders, and conditions associated with abnormal deposition of A-beta protein.
-
Methods of treatment of amyloidosis using bi-aryl aspartyl protease inhibitors申请人:John Varghese公开号:US20060014737A1公开(公告)日:2006-01-19The invention relates to novel compounds and methods of treating diseases, disorders, and conditions associated with amyloidosis. Amyloidosis refers to a collection of diseases, disorders, and conditions associated with abnormal deposition of A-beta protein.这项发明涉及新型化合物和治疗与淀粉样变性相关的疾病、紊乱和症状的方法。淀粉样变性指与A-beta蛋白异常沉积相关的一系列疾病、紊乱和症状。
-
One-pot synthesis of polysubstituted indoles from aliphatic nitro compounds under mild conditions作者:Christopher A. Simoneau、Alexis M. Strohl、Bruce GanemDOI:10.1016/j.tetlet.2007.01.031日期:2007.3Polysubstituted indoles can be prepared directly from functionalized nitroalkanes under very mildly acidic conditions in a simple, one-pot, two-stage procedure.
-
TROPAN COMPOUND申请人:ONO PHARMACEUTICAL CO., LTD.公开号:EP1785421A1公开(公告)日:2007-05-16The conventional anticholinergic drugs for administration through inhalation have been considered to have the possibility of aggravating dysuria associated with prostatic hyperplasia mediated by blood, and it has been demanded that the conventional anticholinergic drugs for administration through inhalation will have to show reduced side effects or adverse ractions. The present invention relates to a compound represented by the general formula (I): (wherein A represents; and R1, R2, R3 and R1 each a hydrogen atom or a substituent; R5 is a substituent; X- is an anion;the symbol: denotes an exo-form or endo-form, or their mixture), its salt or solvation product thereof. They are useful as a prophylactic and/or therapeutic agent with reduced side effects or adverse reactions for the diseases mediated by the muscarinic receptor.
-
Enantioconvergent Alkylations of Amines by Alkyl Electrophiles: Copper-Catalyzed Nucleophilic Substitutions of Racemic α-Halolactams by Indoles作者:Agnieszka Bartoszewicz、Carson D. Matier、Gregory C. FuDOI:10.1021/jacs.9b07875日期:2019.9.18classic SN2 reaction of an amine with an alkyl electrophile, both with respect to reactivity and to enantioselectivity. In this study, we describe the development of a user-friendly method (reaction at room temperature, with commercially available catalyst components) for the enantioconvergent nucleophilic substitution of racemic secondary alkyl halides (α-iodolactams) by indoles. Mechanistic studies are
表征谱图
-
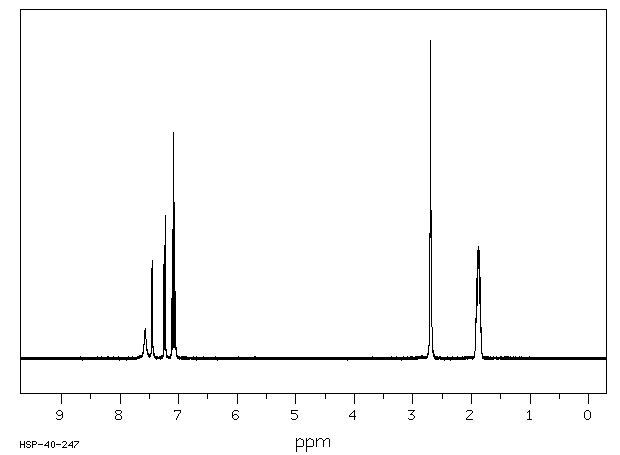
氢谱1HNMR
-
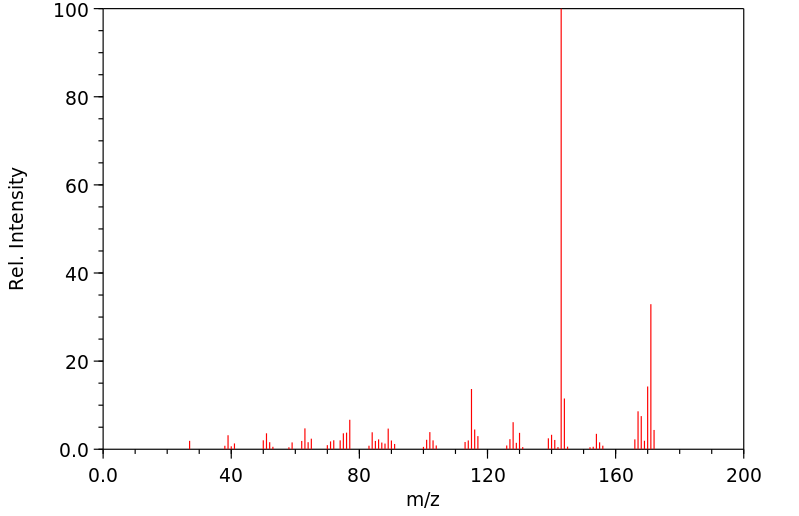
质谱MS
-
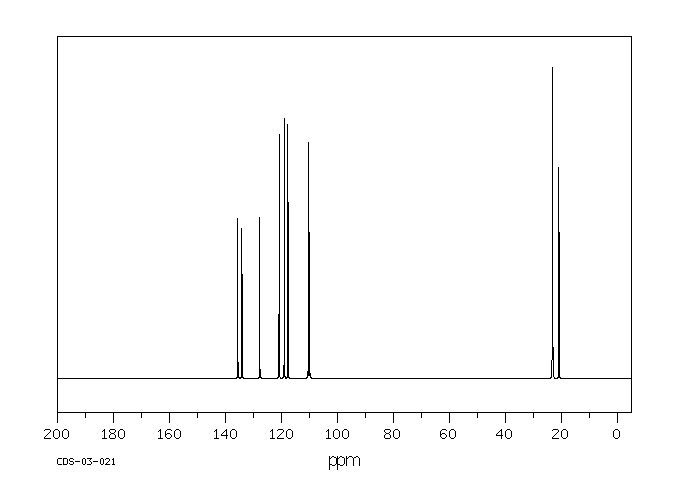
碳谱13CNMR
-
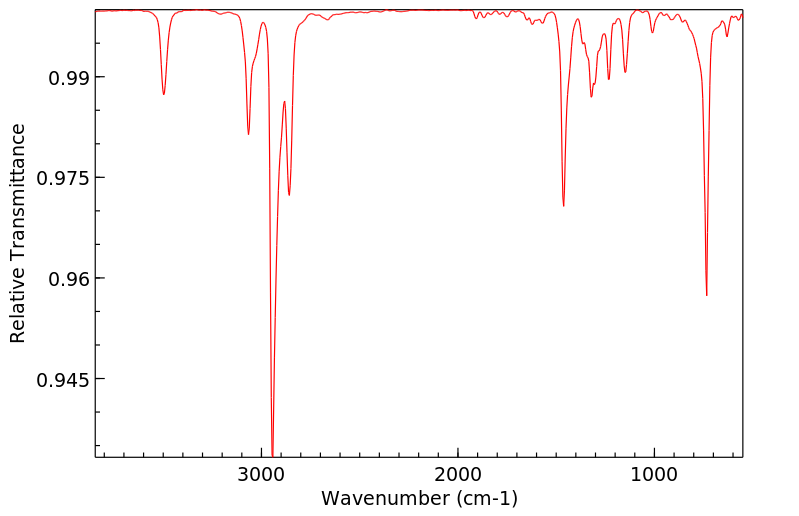
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(Z)-3-[[[2,4-二甲基-3-(乙氧羰基)吡咯-5-基]亚甲基]吲哚-2--2-
(S)-(-)-5'-苄氧基苯基卡维地洛
(R)-(+)-5'-苄氧基卡维地洛
(R)-卡洛芬
(N-(Boc)-2-吲哚基)二甲基硅烷醇钠
(E)-2-氰基-3-(5-(2-辛基-7-(4-(对甲苯基)-1,2,3,3a,4,8b-六氢环戊[b]吲哚-7-基)-2H-苯并[d][1,2,3]三唑-4-基)噻吩-2-基)丙烯酸
(4aS,9bR)-6-溴-2,3,4,4a,5,9b-六氢-1H-吡啶并[4,3-B]吲哚
(3Z)-3-(1H-咪唑-5-基亚甲基)-5-甲氧基-1H-吲哚-2-酮
(3Z)-3-[[[4-(二甲基氨基)苯基]亚甲基]-1H-吲哚-2-酮
(3R)-(-)-3-(1-甲基吲哚-3-基)丁酸甲酯
(3-氯-4,5-二氢-1,2-恶唑-5-基)(1,3-二氧代-1,3-二氢-2H-异吲哚-2-基)乙酸
齐多美辛
鸭脚树叶碱
鸭脚木碱,鸡骨常山碱
鲜麦得新糖
高氯酸1,1’-二(十六烷基)-3,3,3’,3’-四甲基吲哚碳菁
马鲁司特
马鞭草(VERBENAOFFICINALIS)提取物
马来酸阿洛司琼
马来酸替加色罗
顺式-ent-他达拉非
顺式-1,3,4,4a,5,9b-六氢-2H-吡啶并[4,3-b]吲哚-2-甲酸乙酯
顺式-(+-)-3,4-二氢-8-氯-4'-甲基-4-(甲基氨基)-螺(苯并(cd)吲哚-5(1H),2'(5'H)-呋喃)-5'-酮
靛青二磺酸二钾盐
靛藍四磺酸
靛红联二甲酚
靛红磺酸钠
靛红磺酸
靛红乙烯硫代缩酮
靛红-7-甲酸甲酯
靛红-5-磺酸钠
靛红-5-磺酸
靛红-5-硫酸钠盐二水
靛红-5-甲酸甲酯
靛红
靛玉红衍生物E804
靛玉红3'-单肟5-磺酸
靛玉红-3'-单肟
靛玉红
靛噻
青色素3联己酸染料,钾盐
雷马曲班
雷莫司琼杂质13
雷莫司琼杂质12
雷莫司琼杂质
雷替尼卜定
雄甾-1,4-二烯-3,17-二酮
阿霉素的代谢产物盐酸盐
阿贝卡尔
阿西美辛杂质3