1,5-二碘戊烷 | 628-77-3
中文名称
1,5-二碘戊烷
中文别名
1,5-二碘代戊烷
英文名称
1,5-diiodopentane
英文别名
diiodopentane
CAS
628-77-3
化学式
C5H10I2
mdl
MFCD00001101
分子量
323.943
InChiKey
IAEOYUUPFYJXHN-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:9°C
-
沸点:101-102 °C/3 mmHg (lit.)
-
密度:2.177 g/mL at 25 °C (lit.)
-
闪点:>110°C
-
稳定性/保质期:
远离氧化物,避光。
计算性质
-
辛醇/水分配系数(LogP):4.1
-
重原子数:7
-
可旋转键数:4
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
TSCA:Yes
-
危险等级:6.1(b)
-
安全说明:S26,S37/39
-
危险品运输编号:2810
-
危险类别:6.1(b)
-
危险品标志:Xi
-
危险类别码:R36/37/38
-
RTECS号:SA0425000
-
包装等级:III
-
WGK Germany:3
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:请将存放在密封容器内,并置于阴凉、干燥处。存储地点需远离氧化剂,务必避光保存。
SDS
1,5-Diiodopentane (stabilized with Copper chip) Revision number: 6
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 1,5-Diiodopentane (stabilized with Copper chip)
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Skin corrosion/irritation Category 2
Category 2A
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Warning
Hazard statements Causes skin irritation
Causes serious eye irritation
Precautionary statements:
Wash hands thoroughly after handling.
[Prevention]
Wear protective gloves/eye protection/face protection.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
[Response]
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: 1,5-Diiodopentane (stabilized with Copper chip)
Percent: >98.0%(GC)
CAS Number: 628-77-3
Synonyms: Pentamethylene Diiodide (stabilized with Copper chip)
C5H10I2
Chemical Formula:
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Section 4. FIRST AID MEASURES
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Unsuitable extinguishing Solid streams of water
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
A small amount of copper chip is added as a stabilizer. Separation such as filtration
should be taken as necessary.
Conditions for safe storage, including any
incompatibilities
Keep container tightly closed. Store in a cool and dark place.
Storage conditions:
Store under inert gas.
Store away from incompatible materials such as oxidizing agents.
Light-sensitive, Air-sensitive
Comply with laws.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Slightly pale yellow - Reddish yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:9°C (Freezing point)
Boiling point/range: 102°C/0.4kPa
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: 2.18
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen Iodide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
RTECS Number: SA0425000
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
No data available
Crustacea:
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
No data available
Henry's Law
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 1,5-Diiodopentane (stabilized with Copper chip)
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Skin corrosion/irritation Category 2
Category 2A
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Warning
Hazard statements Causes skin irritation
Causes serious eye irritation
Precautionary statements:
Wash hands thoroughly after handling.
[Prevention]
Wear protective gloves/eye protection/face protection.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
[Response]
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: 1,5-Diiodopentane (stabilized with Copper chip)
Percent: >98.0%(GC)
CAS Number: 628-77-3
Synonyms: Pentamethylene Diiodide (stabilized with Copper chip)
C5H10I2
Chemical Formula:
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Section 4. FIRST AID MEASURES
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Unsuitable extinguishing Solid streams of water
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
A small amount of copper chip is added as a stabilizer. Separation such as filtration
should be taken as necessary.
Conditions for safe storage, including any
incompatibilities
Keep container tightly closed. Store in a cool and dark place.
Storage conditions:
Store under inert gas.
Store away from incompatible materials such as oxidizing agents.
Light-sensitive, Air-sensitive
Comply with laws.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Slightly pale yellow - Reddish yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:9°C (Freezing point)
Boiling point/range: 102°C/0.4kPa
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: 2.18
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen Iodide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
RTECS Number: SA0425000
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
No data available
Crustacea:
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
No data available
Henry's Law
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
反应信息
-
作为反应物:参考文献:名称:Synthesis of Primary Amines via Alkylation of the Sodium Salt of Trifluoroacetamide: An Alternative to the Gabriel Synthesis摘要:DOI:10.1055/s-1984-31028
-
作为产物:参考文献:名称:酸催化有机加成和取代反应的强大工具†摘要:已经开发出一种用于酸催化反应的新型绿色化学工具。多功能工具基于干固体材料向质子提供质子(H +)的能力,同时使用亲核试剂(例如NaI)。该方法可以在20–50°C的温度下进行以下反应:在R 2 C CH 2系统(R≠H)中将碘或醇选择性加成更多取代的碳,酯化反应,例如游离脂肪酸与甲醇的反应,以及在更高的温度下(60–100°C):游离脂肪酸与受阻醇(异丙醇)的酯化反应,向C中添加碘C键,含杂环的氧的打开,在其他官能团或仲醇基存在下伯羟基被碘选择性取代,醇与腈(R-CN)酯化,脂肪酸甘油三酸酯酯交换成生物柴油和糖的次级部分上伯羟基(–CH 2 OH)的选择性衍生化,而没有任何保护。大多数反应也由重复使用的Dowex®阳离子交换树脂进行。DOI:10.1039/c4ra17321j
文献信息
-
双链连接的细胞毒性药物偶联物申请人:杭州多禧生物科技有限公司公开号:CN110621673A公开(公告)日:2019-12-27本发明涉及以双链连接体连接的细胞毒性分子与细胞结合分子的偶联物,如结构式(I)所示.本发明还提供了以双链连接制备细胞毒性药物/分子与细胞结合剂的偶联物的特定方式。它还涉及偶联物在治疗癌症,或自身免疫疾病或传染病中的应用。
-
[EN] CROSS-LINKED PYRROLOBENZODIAZEPINE DIMER (PBD) DERIVATIVE AND ITS CONJUGATES<br/>[FR] DÉRIVÉ DE DIMÈRE DE PYRROLOBENZODIAZÉPINE RÉTICULÉ (PBD) ET SES CONJUGUÉS申请人:HANGZHOU DAC BIOTECH CO LTD公开号:WO2020006722A1公开(公告)日:2020-01-09A novel cross-linked cytotoxic agents, pyrrolobenzo-diazepine dimer (PBD) derivatives, and their conjugates to a cell-binding molecule, a method for preparation of the conjugates and the therapeutic use of the conjugates.
-
[EN] CONJUGATION LINKERS CONTAINING 2,3-DIAMINOSUCCINYL GROUP<br/>[FR] LIEURS DE CONJUGAISON CONTENANT UN GROUPE 2,3-DIAMINOSUCCINYLE申请人:HANGZHOU DAC BIOTECH CO LTD公开号:WO2020073345A1公开(公告)日:2020-04-16Provided is a conjugate of a cytotoxic drug/molecule to a cell-binding molecule with a bis-linker (adual-linker) containing a 2, 3-diaminosuccinyl group. It also relates to preparation of the conjugate of a cytotoxic drug/molecule to a cell-binding molecule with the bis-linker, particularly when the drug having functional groups of amino, hydroxyl, diamino, amino-hydroxyl, dihydroxyl, carboxyl, hydrazine, aldehyde and thiol for conjugation with the bis-linker in a specific manner, as well as the therapeutic use of the conjugates.
-
新規な連結体及び生体分子と薬物との特異的共役におけるその使用申请人:ハンジョウ ディーエーシー バイオテック シーオー.,エルティディ.HANGZHOU DAC BIOTECH CO.,LTD.公开号:JP2021073185A公开(公告)日:2021-05-13【課題】連結体の製造方法及び均一な共役体の製造におけるそのような連結体の使用方法、並びに癌、感染症、及び自己免疫疾患の治療における共役体の適用方法に関する。【解決手段】細胞結合分子上のチオールの対に特異的に架橋連結することにより、連結体あたり2以上の化合物/細胞毒性剤を細胞結合分子と共役させるための、2,3−ジ置換コハク酸基又は2−モノ置換若しくは2,3−ジ置換フマル酸又はマレイン酸(トランス(E)−又はシス(Z)−ブテン二酸)基を含む新規な連結体。上記連結体は下記一般式で例示される。【選択図】なし
-
Copper bis(oxazolines) as catalysts for stereoselective aziridination of styrenes with N-tosyloxycarbamates作者:Hélène Lebel、Michaël Parmentier、Olivier Leogane、Karen Ross、Cédric SpitzDOI:10.1016/j.tet.2012.02.044日期:2012.4styrenes. Subsequent ring-opening reactions of styrene-derived aziridines at the benzylic position were observed with various oxygen and nitrogen nucleophiles under Lewis acid catalysis affording the corresponding products with complete inversion of stereochemistry. The strategy was used to synthesize the β-blocker, (R)-nifenalol.
表征谱图
-
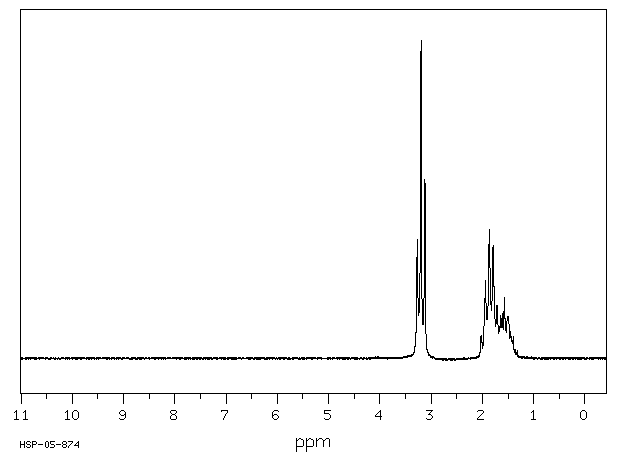
氢谱1HNMR
-
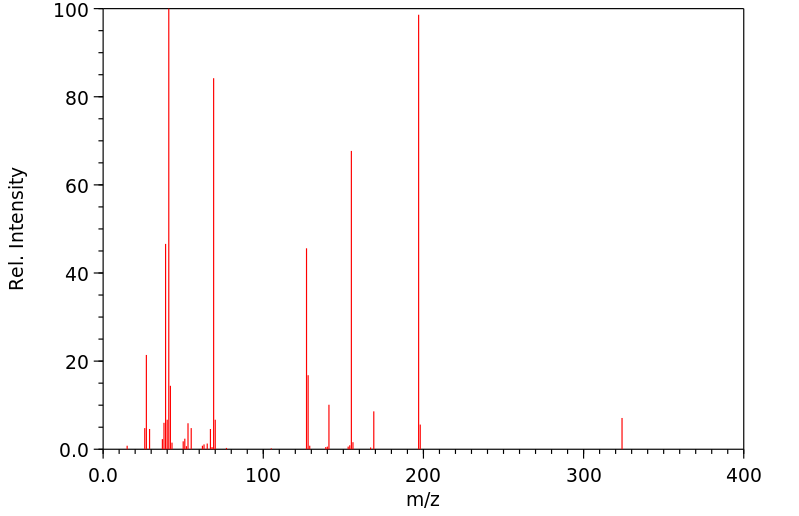
质谱MS
-
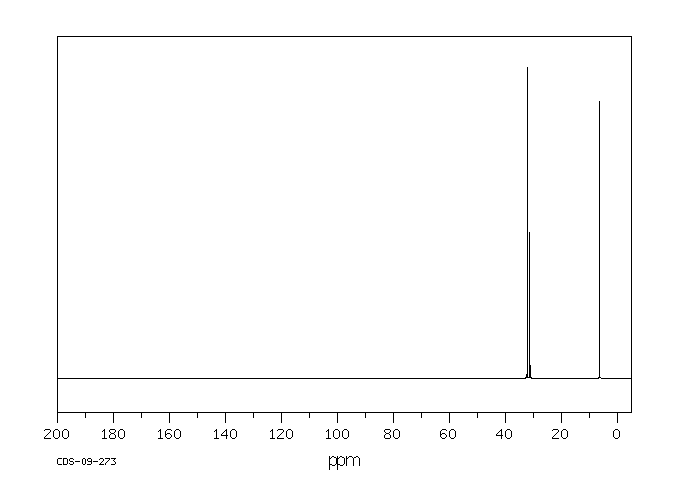
碳谱13CNMR
-
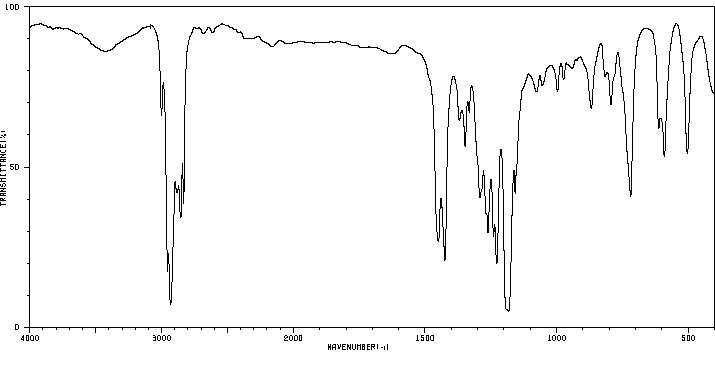
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
胍,N-[3-(氨基甲基)-5-甲基苯基]-N'-乙基-
碘甲烷
碘甲基环辛烷
碘甲基环戊烷
碘环庚烷
碘环十二烷
碘环丁烷
碘十六烷
碘代环戊烷
碘代正辛烷-D2
碘代异丁烷
碘代叔丁烷
碘代丙烷-D7
碘代丙烷-D3
碘代丙烷-D2
碘代丙烷-D2
碘乙烷-d<
碘乙烷-D1
碘乙烷-2-13C
碘乙烷-2,2,2-d3
碘乙烷-1-13C
碘乙烷-1,1-d2
碘乙烷(1,2-13C2)
碘乙烷
碘丁烷-D9
碘(碘甲氧基)甲烷
甲基碘化钙
环辛烷,1-氟-2-碘-,反-
环戊二烯并[1,3]环丙烯并[1,2]环庚烯-2(1H)-酮,八氢-3a,5,5-三甲基-,(3aR,3bR,8aS)-rel-
环丙基碘
无花果蛋白酶来源于无花果树乳胶
新戊氧基
新戊基碘
抗-8-碘-1,5-二甲基二环<3.2.1>辛烷
抗-8-碘-1,5-二甲基二环<3.2.1>辛烷
异戊基碘
异丁基锰(II)碘化物
反式-4-己烯基碘
十氢-2-(碘甲基)-萘
十四烷基碘化物
十五氟碘庚烷
十九氟-9-碘壬烷
全氟辛基碘烷
全氟碘代丁烷
全氟异戊基碘
全氟异庚基碘化物
全氟异壬基碘
全氟异十一烷基碘化物
全氟己基碘烷
全氟叔丁基碘化物