4'-溴苯丙酮 | 10342-83-3
中文名称
4'-溴苯丙酮
中文别名
对溴苯丙酮;4"-溴苯丙酮;对丙酰基溴苯;4-溴代苯丙酮
英文名称
4'-Bromopropiophenone
英文别名
1-(4-bromo-phenyl)-propan-1-one;p-bromopropiophenone;4’-bromopropiophenone;4′-bromopropiophenone;1-(4-bromophenyl)propan-1-one
CAS
10342-83-3
化学式
C9H9BrO
mdl
MFCD00000106
分子量
213.074
InChiKey
UOMOSYFPKGQIKI-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:45-47 °C(lit.)
-
沸点:138-140 °C14 mm Hg(lit.)
-
密度:1.3841 (rough estimate)
-
闪点:>230 °F
-
溶解度:可溶于乙腈(少许)、氯仿(少许)、甲醇
-
稳定性/保质期:
遵照规格使用和储存则不会分解。
计算性质
-
辛醇/水分配系数(LogP):3.1
-
重原子数:11
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.222
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
危险品标志:Xi
-
安全说明:S26,S37/39
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:2914399090
-
危险性防范说明:P261,P280,P305+P351+P338
-
危险性描述:H302,H315,H319,H332,H335
-
储存条件:请将密封于阴凉干燥处。
SDS
| Name: | 4 -Bromopropiophenone 97% Material Safety Data Sheet |
| Synonym: | None Known |
| CAS: | 10342-83-3 |
Synonym:None Known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 10342-83-3 | 4'-Bromopropiophenone | 97% | 233-745-7 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Runoff from fire control or dilution water may cause pollution.
Extinguishing Media:
Use water spray to cool fire-exposed containers. Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 10342-83-3: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: moist, white to off-white
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 138 - 140 deg C @ 14.00mmHg
Freezing/Melting Point: 45.00 - 47.00 deg C
Autoignition Temperature: Not applicable.
Flash Point: > 110 deg C (> 230.00 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C9H9BrO
Molecular Weight: 213.07
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, reducing agents, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents, reducing agents, strong acids, strong bases.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen bromide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 10342-83-3 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
4'-Bromopropiophenone - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 10342-83-3: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 10342-83-3 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 10342-83-3 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
化学性质:白色结晶体,熔点为45-47℃。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-溴苯乙酮 para-bromoacetophenone 99-90-1 C8H7BrO 199.047 2,4’-二溴苯丙酮 2-bromo-1-(4-bromophenyl)propan-1-one 38786-67-3 C9H8Br2O 291.97 4-溴-1-(2-羟甲基)丙酰苯 4-bromo-1-(2-hydroxymethyl)propionyl benzene 92207-34-6 C10H11BrO2 243.1 3-(4-溴苯甲酰)丙酸 4-(4-bromophenyl)-4-oxo-butyric acid 6340-79-0 C10H9BrO3 257.084 4-丙基溴苯 4-bromopropylbenzene 588-93-2 C9H11Br 199.09 对溴苯甲醛 4-bromo-benzaldehyde 1122-91-4 C7H5BrO 185.02 4-溴-alpha-乙基苄醇 (+/-)-1-(4-bromophenyl)propanol 4489-22-9 C9H11BrO 215.09 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 1-(4-溴苯基)-2-甲基丙烷-1-酮 4'-bromoisobutyrophenone 49660-93-7 C10H11BrO 227.101 3-溴-1-(4-溴苯基)丙烷-1-酮 3-bromo-1-(4-bromophenyl)propan-1-one 33994-13-7 C9H8Br2O 291.97 1-(4-溴苯基)-2,2-二甲基丙烷-1-酮 1-(4-bromophenyl)-2,2-dimethylpropan-1-one 30314-45-5 C11H13BrO 241.128 2,4’-二溴苯丙酮 2-bromo-1-(4-bromophenyl)propan-1-one 38786-67-3 C9H8Br2O 291.97 1-(4-溴苯基)-1,2-丙二酮 1-(4-bromophenyl)-1,2-propanedione 10557-20-7 C9H7BrO2 227.057 1-(4-溴苯基)丙-2-烯-1-酮 1-(4-bromophenyl)-2-methylprop-2-en-1-one 42071-66-9 C10H9BrO 225.085 4-溴-1-(2-羟甲基)丙酰苯 4-bromo-1-(2-hydroxymethyl)propionyl benzene 92207-34-6 C10H11BrO2 243.1 1-(4-溴-苯基)-2-氯-丙-1-酮 1-(4-Bromophenyl)-2-chloropropan-1-one 87010-95-5 C9H8BrClO 247.519 —— 1-(4-bromophenyl)-2-hydroxypropan-1-one 145070-49-1 C9H9BrO2 229.073 —— (R)-1-(4-bromophenyl)-2-hydroxypropan-1-one —— C9H9BrO2 229.073 —— (S)-1-(4-bromophenyl)-2-chloropropan-1-one 87010-95-5 C9H8BrClO 247.519 苯丙酮 1-phenyl-propan-1-one 93-55-0 C9H10O 134.178 —— 1-(4-bromophenyl)prop-2-en-1-one 22731-70-0 C9H7BrO 211.058 —— 1-(4-bromophenyl)-3-methoxy-2-methylpropan-1-one 49661-00-9 C11H13BrO2 257.127 4-溴甲卡西酮 4-bromomethcathinone 486459-03-4 C10H12BrNO 242.115 —— 1-(4-bromophenyl)-2-(hydroxyimino)propan-1-one 56472-73-2 C9H8BrNO2 242.072 4-丙基溴苯 4-bromopropylbenzene 588-93-2 C9H11Br 199.09 —— 2-azido-1-(4-bromophenyl)propan-1-one 875289-85-3 C9H8BrN3O 254.086 4-碘苯丙酮 1-(4-iodophenyl)propan-1-one 31970-26-0 C9H9IO 260.074 4-羟基苯丙酮 4-hydroxypropiophenone 70-70-2 C9H10O2 150.177 4-胺基丙苯酮 1-(4-aminophenyl)-1-propanone 70-69-9 C9H11NO 149.192 —— 1-bromo-4-neopentylbenzene 51991-28-7 C11H15Br 227.144 —— 1-(4-vinylphenyl)propan-1-one 7646-72-2 C11H12O 160.216 1-(4-环丙基苯基)丙烷-1-酮 1-(4-cyclopropylphenyl)propan-1-one 30170-62-8 C12H14O 174.243 4-氰基苯丙酮 4-propionylbenzonitrile 52129-98-3 C10H9NO 159.188 —— 4-(4-bromo-phenyl)-3-methyl-4-oxo-cis-crotonic acid 35513-28-1 C11H9BrO3 269.095 —— 4-(4-bromophenyl)-3-methyl-4-oxobut-2-enoic acid 35504-94-0 C11H9BrO3 269.095 4-丙酰基苯甲酰氯 p-Propionylbenzoyl Chloride 153929-32-9 C10H9ClO2 196.633 —— 1-(4-(difluoromethyl)phenyl)propan-1-one —— C10H10F2O 184.186 —— 4'-Br-2-Me-3-piperidinopropiophenon 3644-58-4 C15H20BrNO 310.234 对甲氧基苯丙酮 4-Methoxypropiophenone 121-97-1 C10H12O2 164.204 4-丙酰苯甲酸 4-carboxypropiophenone 4219-55-0 C10H10O3 178.188 (1R)-1-(4-溴苯基)-1-丙醇 (R)-1-(4-Bromophenyl)-1-propanol 112777-66-9 C9H11BrO 215.09 —— (S)-1-(4-bromophenyl)propan-1-ol —— C9H11BrO 215.09 4-溴-alpha-乙基苄醇 (+/-)-1-(4-bromophenyl)propanol 4489-22-9 C9H11BrO 215.09 —— (E)-1-(4-bromophenyl)-2-propanone oxime 104749-39-5 C9H10BrNO 228.088 4-溴查尔酮 (E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one 2403-27-2 C15H11BrO 287.156 —— 2-(4-bromophenyl)butanal —— C10H11BrO 227.101 1-(4-溴苯基)-1-丙胺 1-(4'-bromophenyl)propylamine 74877-09-1 C9H12BrN 214.105 —— 1-bromo-4-(1-bromopropyl)benzene 1094335-60-0 C9H10Br2 277.986 1-溴-4-丁-1-烯-2-基苯 1-bromo-4-(but-1-en-2-yl)benzene 42427-51-0 C10H11Br 211.101 —— 4-Propionyl-diphenylamin 23600-84-2 C15H15NO 225.29 4-丙酰联苯 1-biphenyl-4-yl-propan-1-one 37940-57-1 C15H14O 210.276 —— 4,4′-dipropanoylbiphenyl 4392-77-2 C18H18O2 266.34 —— 1-(4'-ethylbiphenyl-4-yl)propan-1-one 96187-90-5 C17H18O 238.329 —— 1-(4'-methylbiphenyl-4-yl)propan-1-one 96187-83-6 C16H16O 224.302 - 1
- 2
- 3
- 4
- 5
反应信息
-
作为反应物:描述:4'-溴苯丙酮 在 palladium diacetate sodium tetrahydroborate 、 sodium carbonate 、 三苯基膦 作用下, 以 丙醇 、 乙醇 为溶剂, 反应 9.0h, 生成 1-([1,1'-biphenyl]-4-yl)propan-1-ol参考文献:名称:南极假丝酵母脂肪酶B催化的1-联芳基-和1-(吡啶基苯基)烷基-1-醇的动力学拆分摘要:南极假丝酵母的脂肪酶B (CAL-B)催化一些1-联芳基-2-基-,-3-基和-4-基乙醇和-丙-1-醇的高度对映选择性(E > 200)酯交换,以及1-(o-,m-和对-吡啶基苯基)乙醇6含乙酸乙烯酯,在所有情况下均应遵守Kazlauskas规则。间位和对位取代的底物在数小时内转化(转化率范围为23-50%),丙-1-醇衍生物的反应速率比乙醇衍生物的反应速率慢。邻位酯交换-取代的醇用了几天,并伴有化学酶促副反应:形成了另一种由半缩醛衍生的乙酸酯,其在6和乙醛之间是由乙酸乙烯酯形成的。在乙酸异丙烯酯作为酰基供体的情况下,该副反应被抑制,酯交换反应的转化度在十天后为20-40%(E > 200)。还证明了(R)-6p作为配体在将二乙基锌不对称加成到苯甲醛中的有用性。DOI:10.1002/adsc.200404330
-
作为产物:参考文献:名称:烷基苯的双氧化/溴化摘要:在溴化钠和Oxone的存在下,一系列烷基苯衍生物被溴化和/或氧化,最多可官能化四个CH键。DOI:10.1016/j.tetlet.2016.01.062
文献信息
-
One-Pot Transformation of Ketoximes into Optically Active Alcohols and Amines by Sequential Action of Laccases and Ketoreductases or ω-Transaminases作者:Raquel S. Correia Cordeiro、Nicolás Ríos-Lombardía、Francisco Morís、Robert Kourist、Javier González-SabínDOI:10.1002/cctc.201801900日期:2019.2.20An enzymatic one‐pot process for asymmetric transformation of prochiral ketoximes into alcohols or amines was developed by sequential coupling of a laccase‐catalyzed deoximation either with a ketone reduction (ketoreductase, KRED) or bioamination (ω‐transaminase, ω‐TA) in aqueous medium. An accurate selection of biocatalysts provided the corresponding products in excellent enantiomeric excesses and
-
Application of Silicon-Initiated Water Splitting for the Reduction of Organic Substrates作者:Ashot Gevorgyan、Satenik Mkrtchyan、Tatevik Grigoryan、Viktor O. IaroshenkoDOI:10.1002/cplu.201800131日期:2018.5several important classes of organic compounds is described. It is found that the reductive water splitting can be promoted by several metalloids among which silicon shows the best efficiency. The developed methodologies were applied for the reduction of nitro compounds, N-oxides, sulfoxides, alkenes, alkynes, hydrodehalogenation as well as for the gram-scale synthesis of several substrates of industrial
-
From a Sequential to a Concurrent Reaction in Aqueous Medium: Ruthenium-Catalyzed Allylic Alcohol Isomerization and Asymmetric Bioreduction作者:Nicolás Ríos-Lombardía、Cristian Vidal、Elisa Liardo、Francisco Morís、Joaquín García-Álvarez、Javier González-SabínDOI:10.1002/anie.201601840日期:2016.7.18The ruthenium‐catalyzed redox isomerization of allylic alcohols was successfully coupled with the enantioselective enzymatic ketone reduction (mediated by KREDs) in a concurrent process in aqueous medium. The overall transformation, formally the asymmetric reduction of allylic alcohols, took place with excellent conversions and enantioselectivities, under mild reaction conditions, employing commercially
-
Copper catalyzed oxygen assisted C(CNOH)–C(alkyl) bond cleavage: a facile conversion of aryl/aralkyl/vinyl ketones to aromatic acids作者:Pochampalli Sathyanarayana、Owk Ravi、Prathap Reddy Muktapuram、Surendar Reddy BathulaDOI:10.1039/c5ob01569c日期:——A novel copper-catalyzed aerobic oxidative C(NOH)–C(alkyl) bond cleavage reaction of aryl/aralkyl/vinyl ketones for the synthesis of aromatic/acrylic acids is described. A series of ketones having aryl/aralkyl/vinyl at the one end and methyl to any higher alkyl at the other end can be selectively cleaved and converted into the corresponding acids via oxime intermediates.
-
α-羟基酮化合物的廉价高效合成方法申请人:北京大学公开号:CN104710256B公开(公告)日:2017-03-22本发明公开了一种α‑羟基酮化合物的廉价高效合成方法。该合成方法包括:在常压条件下,以碘单质、N‑溴代丁二酰亚胺、溴化铜、溴单质、溴化氢、N‑碘代丁二酰亚胺或碘化氢作为催化剂,以亚砜为氧化剂,以水或亚砜为羟基源,以亚砜,乙酸乙酯、N,N‑二甲基甲酰胺、乙腈、甲苯、1,4‑二氧六环、1,2‑二氯乙烷、四氢呋喃或H2O为溶剂,与羰基化合物混合于10‑120℃温度下进行氧化羟基化反应即可将羰基化合物高选择性的转化为α‑羟基酮化合物。本发明方法与传统合成方法相比具有操作简单,收率高,条件简单,易于纯化,废弃物排放量少,反应设备简单,易于工业化生产等诸多优点。本发明的方法具有广泛地适用性,能够用于多种α‑羟基酮化合物的合成。
表征谱图
-
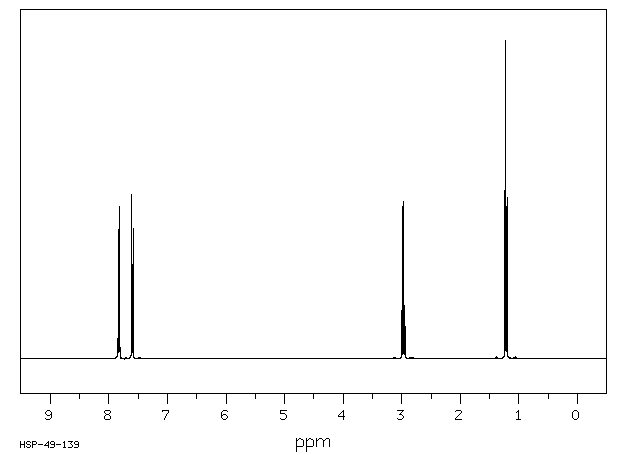
氢谱1HNMR
-
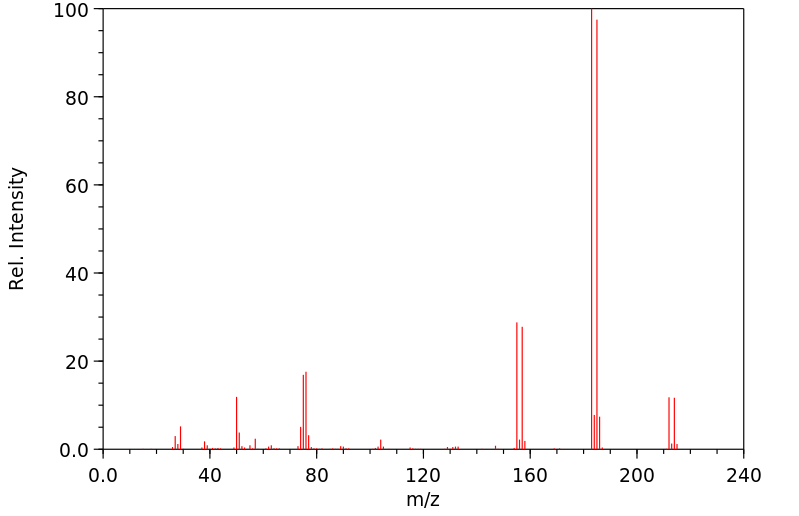
质谱MS
-
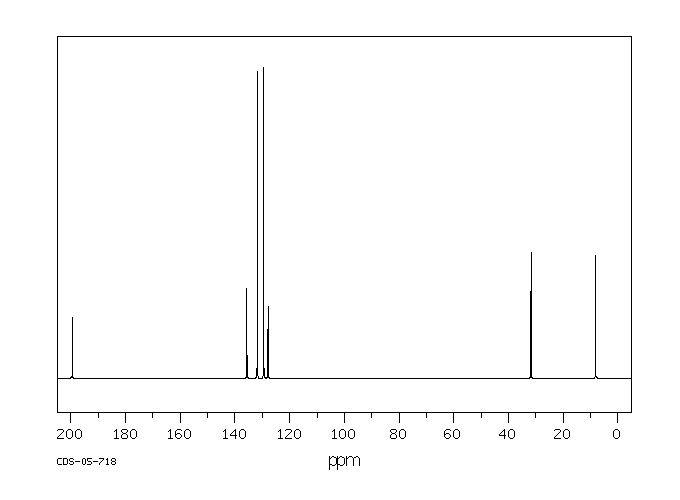
碳谱13CNMR
-
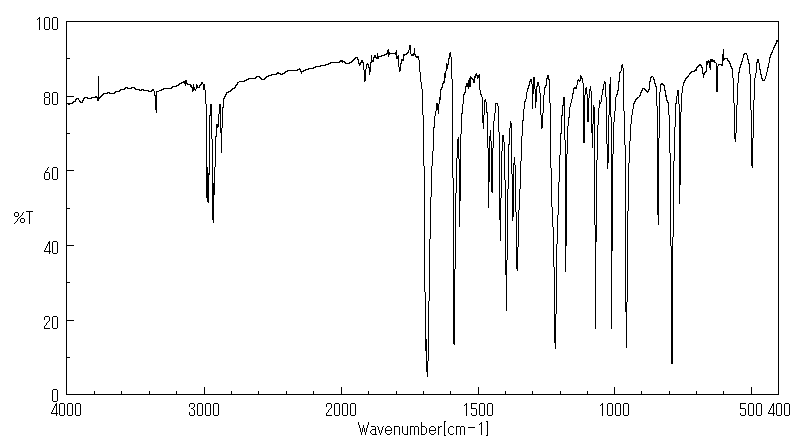
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷