4-(2-溴乙基)苯乙酮 | 40422-73-9
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:111-112 °C(Press: 0.3 Torr)
-
密度:1.360±0.06 g/cm3(Predicted)
-
溶解度:乙腈(微溶)。氯仿(微溶)
计算性质
-
辛醇/水分配系数(LogP):2.8
-
重原子数:12
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2914700090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319
-
储存条件:存放条件:室温、密封、干燥。
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 乙基溴苯 1-phenyl-2-bromoethane 103-63-9 C8H9Br 185.063 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 1,1'-(1,2-乙二基二-4,1-亚苯基)二-乙酮 4,4'-diacetylbibenzyl 793-06-6 C18H18O2 266.34 1-(4-乙烯基-苯基)-乙酮 1-(4-vinylphenyl)ethanone 10537-63-0 C10H10O 146.189 —— 3-(4-acetylphenyl)propanenitrile 158613-21-9 C11H11NO 173.214 —— p-<2-Dimethylamino-aethyl>-acetophenon 91562-68-4 C12H17NO 191.273 4-(2-溴乙基)苯甲酸 4-(2-bromoethyl)benzoic acid 52062-92-7 C9H9BrO2 229.073 3-(4-乙酰基苯基)丙酸 3-(4-acetylphenyl)propanoic acid 39105-51-6 C11H12O3 192.214 4-(2-溴乙基)苯甲酸甲酯 methyl 4-(2-bromoethyl)benzoate 136333-97-6 C10H11BrO2 243.1 —— 4-(2-bromoethyl)styrene 2499-63-0 C10H11Br 211.101 2-(4-乙酰基苯基)丙酸 2-[4-(acetyl)phenyl] propanoic acid 41387-29-5 C11H12O3 192.214 4-(1-羟乙基)苯乙基溴 4-(1-hydroxyethyl)phenethyl bromide 53085-77-1 C10H13BrO 229.117
反应信息
-
作为反应物:描述:4-(2-溴乙基)苯乙酮 在 三甲基氯硅烷 、 potassium tert-butylate 、 N,N-二异丙基乙胺 、 三氟乙酸 作用下, 以 四氢呋喃 、 二氯甲烷 、 2,2,2-三氟乙醇 、 N,N-二甲基乙酰胺 、 二甲基亚砜 为溶剂, 反应 5.0h, 生成 艾乐替尼参考文献:名称:METHOD FOR PREPARING ALECTINIB摘要:一种制备阿雷替尼(Alectinib, I)的方法,包括以下制备步骤:将6-氰基-1H-吲哚-3-甲酸酯和4-乙基-3-(4-吗啡啉-4-基哌啶-1-基)-α,α-二甲基苄醇进行缩合、水解和环化反应,以制备阿雷替尼(I)。该制备方法具有原料易得、工艺简单、经济环保且适合工业生产的特点。公开号:US20170247352A1
-
作为产物:描述:参考文献:名称:OXIMES: I. THE SYNTHESIS OF SOME SUBSTITUTED 2-OXIMINOACETOPHENONES摘要:一些p-(ω′-二甲氨基烷基)-和p-(ω′-二甲氨氧基)-2-氧代肟苯酮及其碘化物的制备已被描述。没有找到合成短链(C0, C1, C2) p-(ω′-二甲氨基烷基)-2-氧代肟苯酮的通用方法。较长链化合物(C3, C4)是通过一种看似通用的方法制备的。 ω-苯基-1-卤代烷烃经过弗里德尔-克拉夫斯酰化反应生成p-(ω′-卤代烷基)-苯乙酮。这些化合物被转化为二甲氨基化合物,随后生成2-氧代肟苯酮。没有找到制备p-(2′-二甲氨基乙基)-2-氧代肟苯酮的方法。使用混合的α,ω-二卤代烷烃和p-羟基苯乙酮完成了p-(ω′-卤代氧基)-苯乙酮的合成。从这些反应物中首先将p-(ω′-卤代氧基)-苯乙酮转化为肟,然后转化为二甲氨基化合物。成功合成了p-(ω′-二甲氨氧乙氧基、-丙氧基、-丁氧基和-戊氧基)-2-氧代肟苯酮。p-(二甲氨基甲氧基)-2-氧代肟苯酮的合成未成功。DOI:10.1139/v62-233
文献信息
-
Effect of cyclodextrin on elimination reactions作者:Luis Viola、Rita H de RossiDOI:10.1139/v99-085日期:1999.6.1
The reaction of 1-bromo-2-X-2-(Y-phenyl) ethane derivatives (1: X = Y = H; 2: X = Ph, Y = H; 3: X = H, Y = 4-Ac; 4: X = H, Y = 3-NO2; 5: X = H, Y = 4-NO2; 6: X = H, Y = 3-Me; 7: X = H, Y = 4-Me) in basic solution was studied, and in most cases, only the elimination product is formed. Only (2-bromo-1-phenylethyl)benzene, 2, yielded significant substitution product, and this yield decreased with the concentration of HO-. Addition of cyclodextrin (β-CD) diminished (about half for 0.02 M cyclodextrin concentration) the reaction rate of all substrates but 4 and 5. In the latter two cases, the rate rises. The observed rate-constant value at 0.5 M NaOH is 6.78 × 10-4 s-1 (at 40°C) and 1.80 × 10-3 s-1 (at 25°C) for 4 and 5, respectively. Under the same reaction conditions but with 0.01 M β-CD, the corresponding rates were 7.70 × 10-4 s-1 and 5.20 × 10-3 s-1. The elimination yield for 2 increased from 64 to 98% when the β-CD changed from zero to 0.02 M at 0.5 M NaHO. Also, there was an increase in the relative elimination products of 20-40% for compounds 6 and 7. The Hammet ρ values were 1.3 and 2.3 for the reaction in pure solvent and in the presence of β-cyclodextrin, indicating an increase in the negative character of the transition state for the reactions in the latter conditions. The results are interpreted in terms of the formation of an inclusion complex whose structure depends on the substrate.Key words: cyclodextrin, elimination reactions, inhibition, catalysis.
1-溴-2-X-2-(Y-苯基)乙烷衍生物(1:X = Y = H;2:X = Ph,Y = H;3:X = H,Y = 4-Ac;4:X = H,Y = 3-NO2;5:X = H,Y = 4-NO2;6:X = H,Y = 3-Me;7:X = H,Y = 4-Me)在碱性溶液中的反应进行了研究,在大多数情况下,只形成消除产物。只有(2-溴-1-苯基乙基)苯,2,产生了显著的取代产物,这种产量随着HO-浓度的减少而减少。环糊精(β-CD)的添加减少了所有底物的反应速率(对于0.02 M环糊精浓度减少了约一半),但对于4和5而言,速率却上升了。在后两种情况下,速率增加。在0.5 M NaOH下观察到的速率常数值分别为6.78 × 10-4 s-1(在40°C下)和1.80 × 10-3 s-1(在25°C下)对于4和5。在相同的反应条件下,但使用0.01 M β-CD,相应的速率分别为7.70 × 10-4 s-1和5.20 × 10-3 s-1。当β-CD从零变为0.02 M时,2的消除产量从64%增加到98%。此外,化合物6和7的相对消除产物增加了20-40%。在纯溶剂和β-环糊精存在的情况下,Hammet ρ值分别为1.3和2.3,表明在后一种条件下反应的过渡态的负性特征增加。结果根据形成取决于底物的包含复合物的结构进行解释。关键词:环糊精,消除反应,抑制,催化。 -
Piperazine-substituted aryl and aralkyl carboxylic acids useful for申请人:Boehringer Mannheim GmbH公开号:US04616086A1公开(公告)日:1986-10-07The present invention is concerned with new carboxylic acid derivatives, with processes for the preparation thereof and with pharmaceutical compositions for lipid depression and thrombocyte aggregation, containing them, and to methods for treating infirmaties caused by excess lipids or thrombocyte aggregation. The new carboxylic acid derivatives according to the present invention are compounds of the general formula: ##STR1## wherein A is a valency bond or a lower alkylene chain, B is a valency bond or a saturated or unsaturated lower alkylene chain, R is hydrogen, an alkyl group which can be substituted by hydroxyl, carboxyl, sulphonic acid or optionally substituted phenoxy group, or R is an aralkyl radical, the aryl moiety of which can be substituted and the alkyl moiety of which is optionally unsaturated and can contain up to 4 carbon atoms, or R is a phenacyl radical, the phenyl moiety optionally substituted, or R is an acyl radical derived from aliphatic, araliphatic or aromatic carboxylic or sulphonic acid, or R is an aryl radical optionally substituted with the proviso that when A is a valency bond, R cannot be hydrogen, methyl, ethyl, hydroxyethyl, benzyl or phenyl, and the physiologically acceptable salts, esters and amides thereof.本发明涉及新的羧酸衍生物,涉及其制备方法以及用于脂质抑制和血小板聚集的药物组合物,其中包含它们,并且涉及用于治疗由于脂质过多或血小板聚集引起的疾病的方法。根据本发明的新羧酸衍生物是符合以下一般式的化合物:##STR1## 其中A是一个价键或较低的烷基链,B是一个价键或饱和或不饱和的较低烷基链,R是氢,一种可以被羟基,羧基,磺酸基或可选择地取代的苯氧基团取代的烷基团,或者R是一种芳基烷基基团,其芳基部分可以被取代,烷基部分可以是可选择地不饱和的并且可以含有多达4个碳原子,或者R是一种苯乙酰基基团,苯基部分可选择地取代,或者R是由脂肪,芳基脂肪或芳香族羧酸或磺酸衍生的酰基基团,或者R是一种芳基基团,可选择地取代,但当A是一个价键时,R不能是氢,甲基,乙基,羟乙基,苄基或苯基,以及其生理上可接受的盐,酯和酰胺。
-
Preparation of Alkyl Indium Reagents by Iodine-Catalyzed Direct Indium Insertion and Their Applications in Cross-Coupling Reactions作者:Man-Ling Zhi、Bing-Zhi Chen、Wei Deng、Xue-Qiang Chu、Teck-Peng Loh、Zhi-Liang ShenDOI:10.1021/acs.joc.9b00204日期:2019.3.1means of an iodine-catalyzed direct indium insertion into alkyl iodide in THF is reported. The thus-generated alkyl indium reagents effectively underwent Pd-catalyzed cross-coupling reactions with various aryl halides, exhibiting good compatibility to a variety of sensitive functional groups. By replacing THF with DMA and using 0.75 equiv of iodine, less reactive alkyl bromide could be used as substrate
-
Bis(phenylsulfonyl)methane mediated synthesis of olefins <i>via</i> a halogen elimination and double bond migration作者:Jing Jiang、Ying Hu、Xin Cai、Liudi Wang、Yanwei Hu、Shaohua Chen、Shilei Zhang、Yinan ZhangDOI:10.1039/c8ob00033f日期:——An effective dehydrochlorination of bis(phenylsulfonyl)alkane to prepare alkene building blocks is developed. The elimination together with double bond migration results in a variety of β,γ-unsaturated bis(phenylsulfonyl)olefins in good yields with only E geometry. The following chemical diversification represents an easy and straightforward access to a series of alkene building blocks.
-
Novel 17beta-hydroxysteroid dehydrogenase type I inhibitors申请人:Messinger Josef公开号:US20050192263A1公开(公告)日:2005-09-013,15-substituted estrone compounds which act as inhibitors of 17β-hydroxysteroid dehydrogenase type I (17β-HSD1), salts thereof, pharmaceutical preparations containing such compounds, processes for preparing such compounds, and therapeutic uses of such compounds, particularly in the treatment or inhibition of steroid hormone dependent diseases or disorders, such as steroid hormone dependent diseases or disorders requiring the inhibition of 17β-hydroxysteroid dehydrogenase type I enzymes and/or requiring the lowering of the endogenous 17β-estradiol concentration, as well as the general use of selective 17β-hydroxysteroid dehydrogenase type 1 inhibitors which possess in addition no or only pure antagonistic binding affinities to the estrogen receptor for the treatment or inhibition of benign gynecological disorders, particularly endometriosis.
表征谱图
-
氢谱1HNMR
-
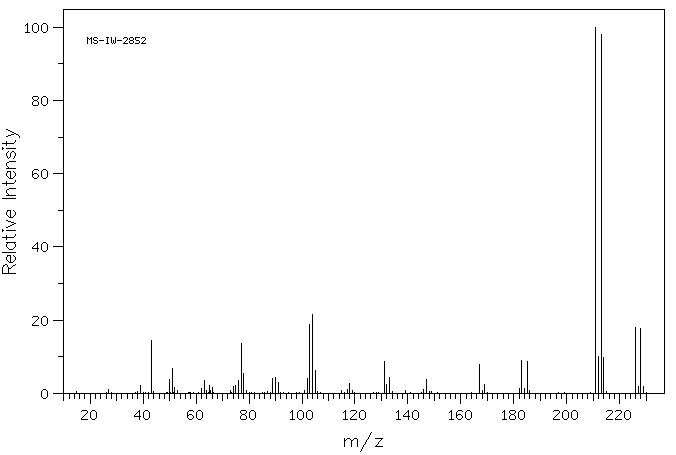
质谱MS
-
碳谱13CNMR
-
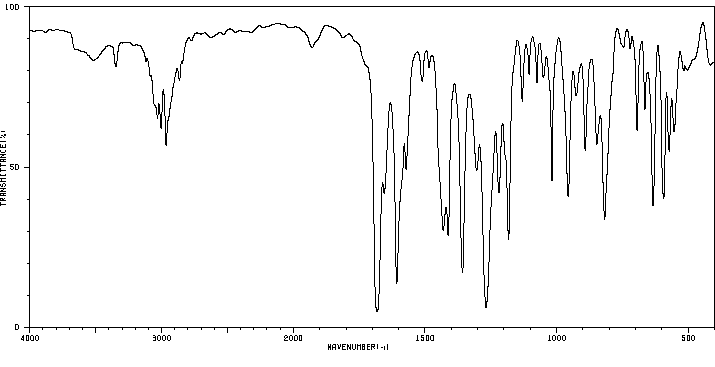
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息