2-甲基吲哚-3-乙酸 | 1912-43-2
物质功能分类
中文名称
2-甲基吲哚-3-乙酸
中文别名
2-甲基-3-吲哚乙酸
英文名称
2-methyl-3-indoleacetic acid
英文别名
2-methylindole-3-acetic acid;2-(2-methyl-1H-indol-3-yl)acetic acid;(2-methylindol-3-yl)acetic acid
CAS
1912-43-2
化学式
C11H11NO2
mdl
MFCD00075006
分子量
189.214
InChiKey
QJNNHJVSQUUHHE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:205 °C (dec.) (lit.)
-
沸点:418.2±30.0 °C(Predicted)
-
密度:1.299±0.06 g/cm3(Predicted)
-
溶解度:可溶于DMSO(少许)、甲醇(少许)
-
稳定性/保质期:
如果按照规定使用和储存,则不会分解,不存在已知的危险反应。应避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):1.9
-
重原子数:14
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.181
-
拓扑面积:53.1
-
氢给体数:2
-
氢受体数:2
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
WGK Germany:3
-
海关编码:2933990090
-
危险类别:IRRITANT
-
安全说明:S22,S24/25
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:请将贮藏器密封保存,在阴凉、干燥处储存,并确保工作环境有良好的通风或排气设施。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 2-Methylindole-3-acetic acid
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2-Methylindole-3-acetic acid
CAS number: 1912-43-2
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C11H11NO2
Molecular weight: 189.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 2-Methylindole-3-acetic acid
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2-Methylindole-3-acetic acid
CAS number: 1912-43-2
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C11H11NO2
Molecular weight: 189.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
2-甲基吲哚-3-乙酸是一种常用的植物生长激素。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-甲基-3-吲哚乙酸乙酯 ethyl (2-methylindol-3-yl)acetate 21909-49-9 C13H15NO2 217.268 —— (2-methyl-indol-3-yl)-glyoxylic acid 62454-47-1 C11H9NO3 203.197 2-(2-甲基-1H-吲哚-3-基)乙腈 2-(2-methyl-1H-indol-3-yl)acetonitrile 4071-16-3 C11H10N2 170.214 —— (2-methyl-3-indolyl)glyoxyloyl chloride 22980-10-5 C11H8ClNO2 221.643 2-氯-1-(2-甲基-1H-吲哚-3-基)-乙酮 2-chloro-1-(2-methyl-1H-indol-3-yl)ethan-1-one 38693-08-2 C11H10ClNO 207.659 2-甲基吲哚-3-甲醛 2-methyl-1H-indole-3-carbaldehyde 5416-80-8 C10H9NO 159.188 2-甲基-3-N,N-二甲氨基甲基吲哚 2-methylgramine 37125-92-1 C12H16N2 188.272 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 (2-甲基-吲哚-3-基)-乙酸甲酯 methyl 2-(2-methyl-1H-indol-3-yl)acetate 78564-10-0 C12H13NO2 203.241 2-甲基-3-吲哚乙酸乙酯 ethyl (2-methylindol-3-yl)acetate 21909-49-9 C13H15NO2 217.268 2-甲基-1H-吲哚-3-乙醇 2-(2-methyl-1H-indol-3-yl)ethanol 56895-60-4 C11H13NO 175.23 —— 2-(2-methyl-1H-indol-3-yl)acetaldehyde 54765-13-8 C11H11NO 173.214 —— 2-formylindol-3-ylacetic acid 118361-82-3 C11H9NO3 203.197 —— benzyl 2-(2-methyl-1H-indol-3-yl)acetate 502607-41-2 C18H17NO2 279.338 —— 2-(2-methyl-1H-indol-3-yl)acetamide 58360-14-8 C11H12N2O 188.229 —— 2-methylindole-3-acetic acid chloride 194017-63-5 C11H10ClNO 207.659 3-吲哚乙酸 3-indolacetic acid 87-51-4 C10H9NO2 175.187 (2-甲基-吲哚-3-基)-乙酸酰肼 2-(2-methyl-1H-indol-3-yl)acetohydrazide 21909-51-3 C11H13N3O 203.244 1,2-二甲基吲哚-3-乙酸 1,2-dimethylindole-3-acetic acid 2597-28-6 C12H13NO2 203.241 —— N-benzyl-2-(2-methyl-1H-indol-3-yl)acetamide 1241355-28-1 C18H18N2O 278.354 —— 2-(1-Ethyl-2-methylindol-3-yl)acetic acid —— C13H15NO2 217.268 —— 2-(2-Methyl-3-indolyl)essigsaeure-morpholid 88114-72-1 C15H18N2O2 258.32 2,3-二甲基吲哚 2,3-dimethylindole 91-55-4 C10H11N 145.204 —— [2-Methyl-1-(3-oxo-1-buten-1-yl)-1H-indol-3-yl]acetic acid 88114-69-6 C15H15NO3 257.289 —— 1-imidazol-1-yl-2-(2-methyl-1H-indol-3-yl)ethanone 1017683-50-9 C14H13N3O 239.277 —— N-benzyl-2-methylindole-3-acetic acid 85620-08-2 C18H17NO2 279.338 1-[[2-(2-甲基-1H-吲哚-3-基)乙酰基]氨基]-3-苯基硫脲 1-(2-methylindolyl-3-acetyl)-4-phenylthiosemicarbazide 54648-89-4 C18H18N4OS 338.433 —— 2-<1-(3-Oxo-1-butenyl)-2-methyl-3-indolyl>essigsaeure-methylester 88114-71-0 C16H17NO3 271.316 帕比司他 panobinostat 404950-80-7 C21H23N3O2 349.433 —— 2-methyl-1-(phenylmethyl)-1H-indole-3-acetic acid ethyl ester 97945-26-1 C19H19NO2 293.365 1-[[2-(2-甲基-1H-吲哚-3-基)乙酰基]氨基]-3-(4-甲基苯基)硫脲 1-(2-methylindolyl-3-acetyl)-4-p-tolylthiosemicarbazide 54648-92-9 C19H20N4OS 352.46 —— dimethyl (1,2-dimethyl-1H-indol-3-yl)malonate —— C15H17NO4 275.304 —— 2-(1-Benzyl-2-methylindol-3-yl)acetaldehyde 85620-11-7 C18H17NO 263.339 —— 2-[2-Methyl-1-[(2-methylpropan-2-yl)oxycarbonyl]indol-3-yl]acetic acid 790262-56-5 C16H19NO4 289.331 —— methyl 2-[2-methyl-1-(4-nitrobenzyl)-1H-indol-3-yl]acetate —— C19H18N2O4 338.363 2-(2-甲基-1-苯基甲氧基羰基吲哚-3-基)乙酸 2-(1-(benzyloxycarbonyl)-2-methyl-1H-indol-3-yl)acetic acid 924635-06-3 C19H17NO4 323.348 待美达新 (1-benzoyl-2-methyl-indol-3-yl)-acetic acid 16401-80-2 C18H15NO3 293.322 —— 2-[2-methyl-1-(3-oxobut-1-enyl)indol-3-yl]acetamide 88114-70-9 C15H16N2O2 256.304 2-[1-(4-氯苯甲酰基)-2-甲基吲哚-3-基]乙酸 2-(1-(4-chlorobenzoyl)-2-methyl-1H-indol-3-yl)acetic acid 16390-26-4 C18H14ClNO3 327.767 —— bis(2-methyl-1H-indol-3-yl)methane 61995-50-4 C19H18N2 274.365 —— (E)-3-(4-{(S)-1-[2-(2-methyl-1H-indol-3-yl)-acetyl]-pyrrolidin-2-yl}-phenyl)-acrylic acid methyl ester 1189153-37-4 C25H26N2O3 402.493 - 1
- 2
- 3
- 4
反应信息
-
作为反应物:参考文献:名称:[EN] PROCESS FOR THE PREPARATION OF PANOBINOSTAT
[FR] PROCÉDÉ DE PRÉPARATION DE PANOBINOSTAT摘要:本发明涉及一种用于制备帕诺比诺斯他及其中间体的新工艺。公开号:WO2021170719A1 -
作为产物:描述:(2-methyl-indol-3-yl)-glyoxylic acid 在 sodium tetrahydroborate 作用下, 以 四氢呋喃 、 甲醇 为溶剂, 反应 18.0h, 生成 2-甲基吲哚-3-乙酸参考文献:名称:合成吲哚-3-乙酸的简便方法摘要:从相应的吲哚开始,通过吲哚-3-乙醛酸合成吲哚-3-乙酸,然后与对甲苯磺酰肼形成,然后用硼氢化钠还原hydr。DOI:10.1016/s0040-4039(00)76815-8
文献信息
-
MACROCYCLIC COMPOUNDS AND THEIR USE AS KINASE INHIBITORS申请人:Combs Andrew Paul公开号:US20090286778A1公开(公告)日:2009-11-19The present invention relates to macrocyclic compounds of Formula I: or pharmaceutically acceptable salts thereof or quaternary ammonium salts thereof wherein constituent members are provided hereinwith, as well as their compositions and methods of use, which are JAK/ALK inhibitors useful in the treatment of JAK/ALK-associated diseases including, for example, inflammatory and autoimmune disorders, as well as cancer.
-
CHEMICAL SUBSTANCES WHICH INHIBIT THE ENZYMATIC ACTIVITY OF HUMAN KALLIKREIN-RELATED PEPTIDASE 6 (KLK6)申请人:Deutsches Krebsforschungszentrum公开号:EP3305781A1公开(公告)日:2018-04-11The invention relates to compounds which are suitable for the treatment of a disease associated with kallikrein-like peptidase 6 overexpression and to pharmaceutical compositions containing such compounds. The invention further relates to a kit of parts comprising such compounds or pharmaceutical compositions.这项发明涉及适用于治疗与kallikrein样肽酶6过度表达相关疾病的化合物,以及含有这些化合物的药物组合物。该发明还涉及包括这些化合物或药物组合物的配套工具包。
-
Green Esterification of Carboxylic Acids Promoted by <i>tert</i> ‐Butyl Nitrite作者:Yonggao Zheng、Yanwei Zhao、Suyan Tao、Xingxing Li、Xionglve Cheng、Gangzhong Jiang、Xiaobing WanDOI:10.1002/ejoc.202100326日期:2021.5.14TBN‐catalyzed green esterification of carboxylic acids has been developed, which features a broad range of substrates and excellent functional groups tolerance. The mechanistic study confirmed that the nitrous acid formed in situ in the system is the actual catalyst for this transformation.
-
Hypoglycemic imidazoline compounds申请人:ELI LILLY AND COMPANY公开号:EP1266897A3公开(公告)日:2003-12-03This invention relates to certain novel imidazoline compounds and analogues thereof, to their use for the treatment of diabetes, diabetic complications, metabolic disorders, or related diseases where impaired glucose disposal is present, to pharmaceutical compositions comprising them, and to processes for their preparation.The compounds have the following formula: whereinX is -O-, -S-, or -NR5-;R5 is hydrogen, C1-8 alkyl, or an amino protecting group;R4 isY is -O-, -S-, or -NR8-;Y' is -O- or -S-;
-
Chemical profiling of HIV-1 capsid-targeting antiviral PF74作者:Lei Wang、Mary C. Casey、Sanjeev Kumar V. Vernekar、Ha T. Do、Rajkumar Lalji Sahani、Karen A. Kirby、Haijuan Du、Atsuko Hachiya、Huanchun Zhang、Philip R. Tedbury、Jiashu Xie、Stefan G. Sarafianos、Zhengqiang WangDOI:10.1016/j.ejmech.2020.112427日期:2020.8herein the design, synthesis, and evaluation of a large number of PF74 analogs aiming to provide a comprehensive chemical profiling of PF74 and advance the understanding on its detailed binding mechanism and pharmacophore. The analogs, containing structural variations mainly in the aniline domain and/or the indole domain, were assayed for their effect on stability of CA hexamers, antiviral activity,HIV-1 的衣壳蛋白 (CA) 通过组装成功能性衣壳核心、控制脱壳和进入核的动力学以及与各种宿主因子相互作用,在病毒复制周期的多个步骤中发挥重要作用。靶向 CA 是一种有吸引力但尚未开发的抗病毒方法。在所有已知的靶向 CA 的小分子化学型中,拟肽 PF74 特别有趣,因为它与一些重要宿主因子使用的相同口袋结合,从而产生非常理想的抗病毒表型。然而,PF74 的进一步开发需要了解其药效团并减轻其较差的代谢稳定性。我们在此报告设计、合成、和评估大量 PF74 类似物,旨在提供 PF74 的全面化学分析,并促进对其详细结合机制和药效团的理解。分析了主要在苯胺结构域和/或吲哚结构域中包含结构变异的类似物对 CA 六聚体稳定性、抗病毒活性和细胞毒性的影响。还测试了选定的类似物在肝微粒体中的代谢稳定性,单独或在 CYP3A 抑制剂存在的情况下。总的来说,我们的研究确定了重要的药效团元素并揭示了 PF74
表征谱图
-
氢谱1HNMR
-
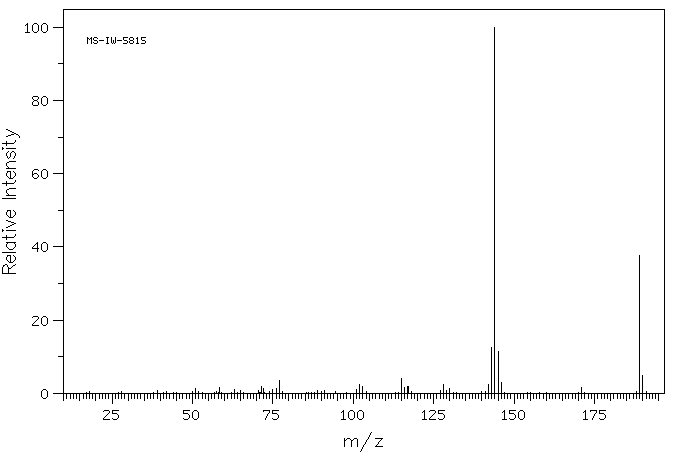
质谱MS
-
碳谱13CNMR
-
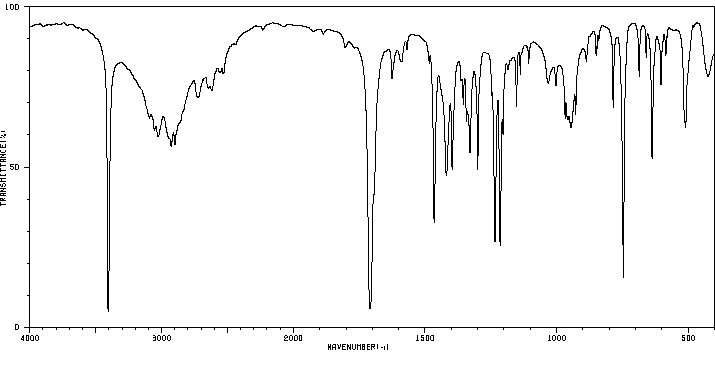
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(Z)-3-[[[2,4-二甲基-3-(乙氧羰基)吡咯-5-基]亚甲基]吲哚-2--2-
(S)-(-)-5'-苄氧基苯基卡维地洛
(R)-(+)-5'-苄氧基卡维地洛
(R)-卡洛芬
(N-(Boc)-2-吲哚基)二甲基硅烷醇钠
(E)-2-氰基-3-(5-(2-辛基-7-(4-(对甲苯基)-1,2,3,3a,4,8b-六氢环戊[b]吲哚-7-基)-2H-苯并[d][1,2,3]三唑-4-基)噻吩-2-基)丙烯酸
(4aS,9bR)-6-溴-2,3,4,4a,5,9b-六氢-1H-吡啶并[4,3-B]吲哚
(3Z)-3-(1H-咪唑-5-基亚甲基)-5-甲氧基-1H-吲哚-2-酮
(3Z)-3-[[[4-(二甲基氨基)苯基]亚甲基]-1H-吲哚-2-酮
(3R)-(-)-3-(1-甲基吲哚-3-基)丁酸甲酯
(3-氯-4,5-二氢-1,2-恶唑-5-基)(1,3-二氧代-1,3-二氢-2H-异吲哚-2-基)乙酸
齐多美辛
鸭脚树叶碱
鸭脚木碱,鸡骨常山碱
鲜麦得新糖
高氯酸1,1’-二(十六烷基)-3,3,3’,3’-四甲基吲哚碳菁
马鲁司特
马鞭草(VERBENAOFFICINALIS)提取物
马来酸阿洛司琼
马来酸替加色罗
顺式-ent-他达拉非
顺式-1,3,4,4a,5,9b-六氢-2H-吡啶并[4,3-b]吲哚-2-甲酸乙酯
顺式-(+-)-3,4-二氢-8-氯-4'-甲基-4-(甲基氨基)-螺(苯并(cd)吲哚-5(1H),2'(5'H)-呋喃)-5'-酮
靛青二磺酸二钾盐
靛藍四磺酸
靛红联二甲酚
靛红磺酸钠
靛红磺酸
靛红乙烯硫代缩酮
靛红-7-甲酸甲酯
靛红-5-磺酸钠
靛红-5-磺酸
靛红-5-硫酸钠盐二水
靛红-5-甲酸甲酯
靛红
靛玉红衍生物E804
靛玉红3'-单肟5-磺酸
靛玉红-3'-单肟
靛玉红
靛噻
青色素3联己酸染料,钾盐
雷马曲班
雷莫司琼杂质13
雷莫司琼杂质12
雷莫司琼杂质
雷替尼卜定
雄甾-1,4-二烯-3,17-二酮
阿霉素的代谢产物盐酸盐
阿贝卡尔
阿西美辛杂质3