间氨基苯磺酰胺 | 98-18-0
中文名称
间氨基苯磺酰胺
中文别名
3-氨基苯磺酰胺;3-氨基苯磺胺;3-氨基苯-1-磺酰胺
英文名称
m-aminobenzenesulfonamide
英文别名
metanilamide;3-aminobenzenesulfonamide;3-aminobenzene-1-sulfonamide
CAS
98-18-0
化学式
C6H8N2O2S
mdl
MFCD00035781
分子量
172.208
InChiKey
JPVKCHIPRSQDKL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:139 °C
-
沸点:420.7±47.0 °C(Predicted)
-
密度:1.303 (estimate)
-
稳定性/保质期:
避免与氧化剂接触。
计算性质
-
辛醇/水分配系数(LogP):-0.4
-
重原子数:11
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:94.6
-
氢给体数:2
-
氢受体数:4
安全信息
-
TSCA:Yes
-
危险品标志:Xi
-
安全说明:S24/25
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:2935009090
-
危险品运输编号:NONH for all modes of transport
-
RTECS号:OY2200000
-
储存条件:储存于阴凉、通风的库房,远离火种、热源,防止阳光直射,保持容器密封。应与氧化剂分开存放,切忌混储,并配备相应品种和数量的消防器材。储区应备有泄漏应急处理设备和合适的收容材料。
SDS
| Name: | 3-Aminobenzene-1-sulfonamide 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 98-18-0 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 98-18-0 | 3-Aminobenzene-1-sulfonamide | 97% | 202-646-0 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Not available.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
May cause respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 98-18-0: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: brown
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 140 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C6H8N2O2S
Molecular Weight: 172
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials, exposure to air.
Incompatibilities with Other Materials:
Acids, oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, oxides of nitrogen, oxides of sulfur, carbon dioxide, ammonia.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 98-18-0: OY2200000 LD50/LC50:
Not available.
Carcinogenicity:
3-Aminobenzene-1-sulfonamide - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 98-18-0: 1
Canada
CAS# 98-18-0 is listed on Canada's NDSL List.
CAS# 98-18-0 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 98-18-0 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
制备步骤
步骤a:3-硝基苯基磺酰胺的合成
将4.71克(20毫摩尔)3-硝基苯基磺酰氯溶解于20毫升乙腈中,然后向其中加入饱和碳酸铵的浓氨水(20毫升),并剧烈搅拌1小时。室温下反应完成后,蒸发掉乙腈,用20毫升冷水稀释残余物,产生沉淀。滤出沉淀,并用水(共两次,每次5毫升)和乙醚洗涤后减压干燥。得到3-硝基苯基磺酰胺3.5克,产率80%。
步骤b:间氨基苯磺酰胺的合成将3.5克(16毫摩尔)3-硝基苯甲磺酰胺溶解在含0.5克阮内镍的20毫升甲醇中,在50℃及70PSI氢压下进行4小时的氢化反应。过滤出催化剂,用温热甲醇洗涤后合并滤液并蒸发,最终得到间氨基苯磺酰胺2.83克,产率95%。
用途用作有机合成中间体
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3-硝基苯磺酰胺 3-nitrophenylsulfonamide 121-52-8 C6H6N2O4S 202.191 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 3-(二甲基氨基)苯磺酰胺 3-Dimethylamino-benzol-sulfonamid-(1) 63935-19-3 C8H12N2O2S 200.261 3-异硫氰基苯磺酰胺 3-isothiocyanatobenzenesulfonamide 23165-62-0 C7H6N2O2S2 214.269 3-叠氮苯磺酰胺 3-azidobenzenesulfonamide 43101-06-0 C6H6N4O2S 198.205 —— 3-isocyanato-benzenesulfonamide 900164-97-8 C7H6N2O3S 198.202 —— 4-Amino-3'-sulfamoyl-azobenzol 92848-84-5 C12H12N4O2S 276.319 —— 3-amino-N-[(dimethylamino)methylidene]benzenesulfonamide 405167-72-8 C9H13N3O2S 227.287 —— (3-sulfamoyl-phenyl)-urea 15466-75-8 C7H9N3O3S 215.233 —— 3-thioureidobenzenesulfonamide 5657-42-1 C7H9N3O2S2 231.299 —— 3-acetylamino-benzenesulfonic acid amide 23010-42-6 C8H10N2O3S 214.245 —— 3-(4'-hydroxyphenyl)diazenylbenzenesulfonamide 1190756-64-9 C12H11N3O3S 277.304 —— 5-amino-2-bromobenzenesulfonamide 100367-51-9 C6H7BrN2O2S 251.104 —— N-acetyl-3-amino-benzenosulfonamide 5661-46-1 C8H10N2O3S 214.245 —— 3-methanesulfonylamino-benzenesulfonamide 117662-92-7 C7H10N2O4S2 250.299 —— 3-(3-Phenylureido)benzenesulfonamide —— C13H13N3O3S 291.331 —— 3-(3-phenylselenoureido)benzenesulfonamide —— C13H13N3O2SSe 354.291 —— N-methyl-N'-(3-sulfamoyl-phenyl)-thiourea —— C8H11N3O2S2 245.326 —— 3-(benzylamino)benzenesulfonamide 1019540-71-6 C13H14N2O2S 262.332 2-氯-N-(3-氨磺酰-苯基)-乙酰胺 2-chloro-N-(3-sulfamoylphenyl)acetamide 135202-69-6 C8H9ClN2O3S 248.69 —— 3-(4'-dimethylaminophenyl)diazenylbenzenesulfonamide 93569-44-9 C14H16N4O2S 304.373 —— 3,3'-((1,4-phenylenebis(methanylylidene))bis(azanylylidene))dibenzenesulfonamide —— C20H18N4O4S2 442.519 —— 3-(Pyrimidin-5-ylamino)benzenesulfonamide 1294519-77-9 C10H10N4O2S 250.281 —— N-phenyl-sulfanilic acid amide 6786-93-2 C12H12N2O2S 248.305 N-苄基-3-氨基苯磺酰胺 3-amino-N-benzylbenzenesulfonamide 303780-52-1 C13H14N2O2S 262.332 —— 3-benzenesulfonylamino-benzenesulfonic acid amide —— C12H12N2O4S2 312.37 —— 2-(dimethylamino)-N-(3-sulfamoyl-phenyl)acetamide 848051-96-7 C10H15N3O3S 257.313 —— N-allyl-N'-(3-sulfamoyl-phenyl)-thiourea —— C10H13N3O2S2 271.364 —— 3-(3-(4'-fluorophenyl)ureido)benzenesulfonamide 185149-49-9 C13H12FN3O3S 309.321 —— tert-butyl 3-sulfamoylphenylcarbamate 259537-18-3 C11H16N2O4S 272.325 —— N-[(dimethylamino)methylidene]-3-azidobenzenesulfonamide 1383120-67-9 C9H11N5O2S 253.285 —— 3-sulfanilylamino-benzenesulfonic acid amide 16840-26-9 C12H13N3O4S2 327.385 —— 3-((4-isopropylbenzyl)amino)benzenesulfonamide —— C16H20N2O2S 304.413 —— 3,3'-((1,2-phenylenebis(methanylylidene))bis(azanylylidene))dibenzenesulfonamide —— C20H18N4O4S2 442.519 —— 3-(toluene-4-sulfonylamino)-benzenesulfonic acid amide —— C13H14N2O4S2 326.397 - 1
- 2
- 3
- 4
反应信息
-
作为反应物:参考文献:名称:356. benz-1:2:4-thiadiazine 1:1-dioxide的衍生物摘要:DOI:10.1039/jr9500001760
-
作为产物:参考文献:名称:发现了一系列5,11-二氢-6 H-苯并[ e ]嘧啶[5,4- b ] [1,4]二氮杂-6-酮类作为选择性PI3K-δ/γ抑制剂摘要:PI3K-δ和PI3K-γ的双重抑制是血液恶性肿瘤的既定治疗策略。报道的选择性靶向PI3K-δ/γ的分子在化学上是相似的,并且基于异喹啉-1(2 H)-one或喹唑啉-4(3 H)-one支架。在这里,我们报告了基于5,11-二氢-6 H-苯并[ e ]嘧啶[5,4- b ] [1,4]二氮杂-6-的一系列有效的,选择性的PI3K-δ/γ抑制剂一种支架具有可比的生化效能和对PI3K信号传导的细胞作用。我们设想这些分子将为开发下一代PI3K-δ/γ靶向治疗剂提供有用的线索。DOI:10.1021/acsmedchemlett.6b00209
文献信息
-
Compositions for Treatment of Cystic Fibrosis and Other Chronic Diseases申请人:Vertex Pharmaceuticals Incorporated公开号:US20150231142A1公开(公告)日:2015-08-20The present invention relates to pharmaceutical compositions comprising an inhibitor of epithelial sodium channel activity in combination with at least one ABC Transporter modulator compound of Formula A, Formula B, Formula C, or Formula D. The invention also relates to pharmaceutical formulations thereof, and to methods of using such compositions in the treatment of CFTR mediated diseases, particularly cystic fibrosis using the pharmaceutical combination compositions.
-
[EN] THIOPHENE DERIVATIVES FOR THE TREATMENT OF DISORDERS CAUSED BY IGE<br/>[FR] DÉRIVÉS DE THIOPHÈNE POUR LE TRAITEMENT DE TROUBLES PROVOQUÉS PAR IGE申请人:UCB BIOPHARMA SRL公开号:WO2019243550A1公开(公告)日:2019-12-26Thiophene derivatives of formula (I) and a pharmaceutically acceptable salt thereof are provided. These compounds have utility for the treatment or prevention of disorders caused by IgE, such as allergy, type 1 hypersensitivity or familiar sinus inflammation.
-
COMPOSITIONS FOR TREATMENT OF CYSTIC FIBROSIS AND OTHER CHRONIC DISEASES申请人:Van Goor Fredrick F.公开号:US20110098311A1公开(公告)日:2011-04-28The present invention relates to pharmaceutical compositions comprising an inhibitor of epithelial sodium channel activity in combination with at least one ABC Transporter modulator compound of Formula A, Formula B, Formula C, or Formula D. The invention also relates to pharmaceutical formulations thereof, and to methods of using such compositions in the treatment of CFTR mediated diseases, particularly cystic fibrosis using the pharmaceutical combination compositions.
-
N,N’-双取代芳基硫脲衍生物及其合成方法 和应用
-
[EN] PYRROLE COMPOUNDS FOR THE TREATMENT OF PROSTAGLANDIN MEDIATED DISEASES<br/>[FR] COMPOSES PYRROLIQUES DESTINES AU TRAITEMENT DE MALADIES INDUITES PAR PROSTAGLANDINE申请人:GLAXO GROUP LTD公开号:WO2003101959A1公开(公告)日:2003-12-11Compounds of formula (I) or a pharmaceutically acceptable derivative thereof: wherein A, R1, R2a, R2b, Rx, R8, and R9 are as defined in the specification, a process for the preparation of such compounds, pharmaceutical compositions comprising such compounds and the use of such compounds in medicine, in particular their use in the treatment of prostaglandin mediated diseases such as pain, inflammatory, immunological, bone, neurodegenerative or renal disorder.
表征谱图
-
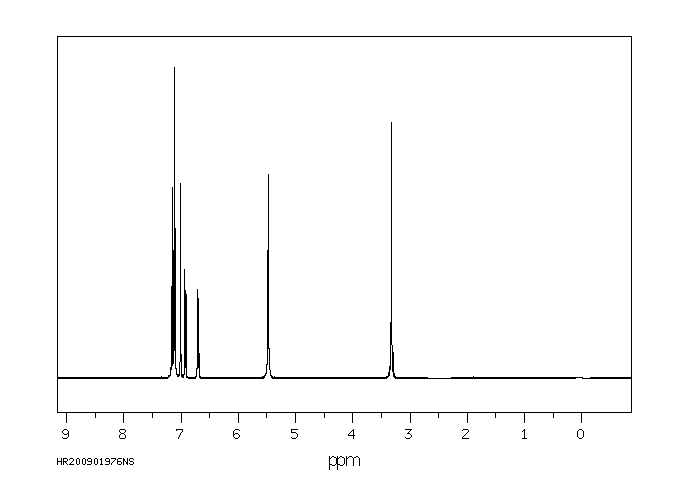
氢谱1HNMR
-
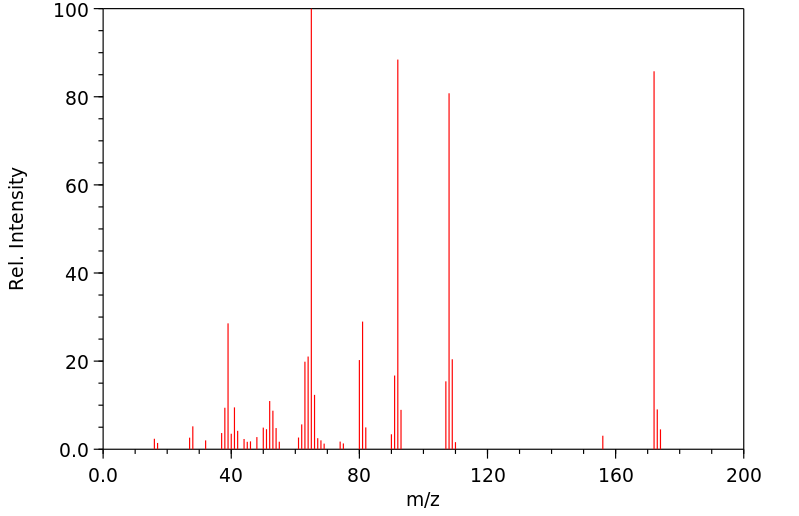
质谱MS
-
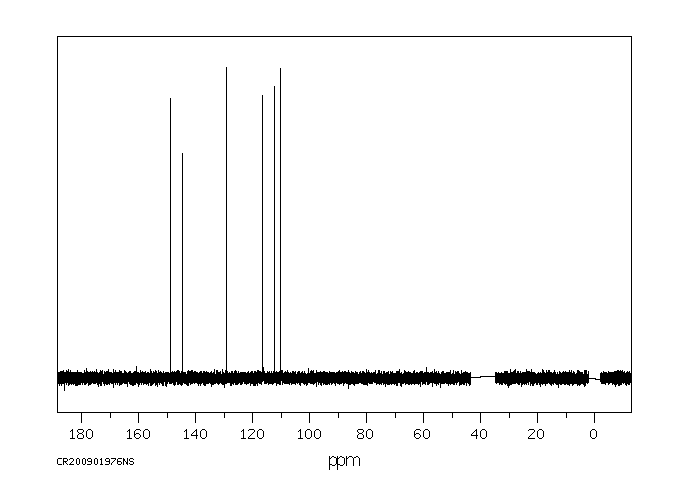
碳谱13CNMR
-
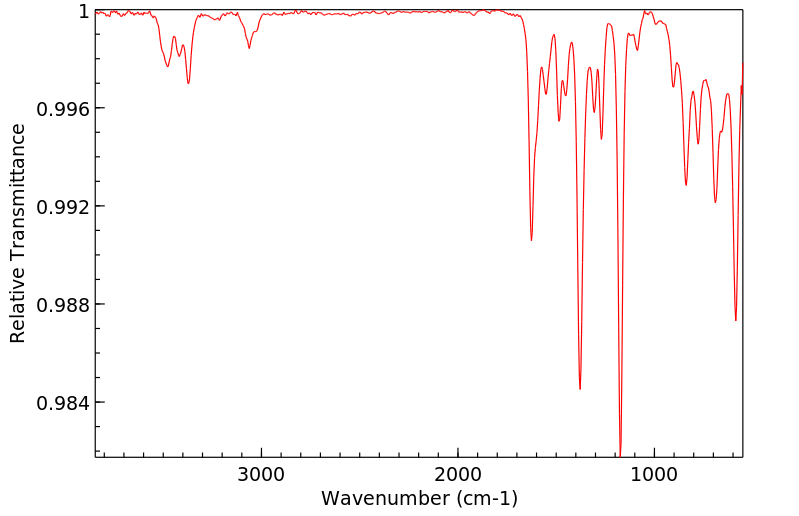
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫