2-碘代甲苯 | 615-37-2
中文名称
2-碘代甲苯
中文别名
2-甲基碘苯;邻碘甲苯;2-碘甲苯
英文名称
2-Iodotoluene
英文别名
ortho-methylphenyl iodide;1-iodo-2-methylbenzene;2-methyliodobenzene;o-iodotoluene;1-iodo-2-toluene
CAS
615-37-2
化学式
C7H7I
mdl
——
分子量
218.037
InChiKey
RINOYHWVBUKAQE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:11.27°C (estimate)
-
沸点:211 °C(lit.)
-
密度:1.713 g/mL at 25 °C(lit.)
-
闪点:194 °F
-
溶解度:氯仿(微溶)、甲醇(微溶)
-
蒸汽压力:0.45 mmHg
-
保留指数:1122.4;1150;1150;1149
-
稳定性/保质期:
性质与稳定性: 在常温常压下,不会分解产生其他物质。
计算性质
-
辛醇/水分配系数(LogP):2.9
-
重原子数:8
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.14
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
TSCA:T
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S26,S36,S37
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:29036990
-
危险品运输编号:UN 2810
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:贮存: 将密器密封后,放入密封的主容器中,并存放在阴凉、干燥的地方。
SDS
| Name: | 2-Iodotoluene 98% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 615-37-2 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 615-37-2 | 2-Iodotoluene | 98 | 210-422-9 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation. May cause chemical conjunctivitis and corneal damage.
Skin:
May cause irritation and dermatitis. May cause cyanosis of the extremities.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated. Aspiration may lead to pulmonary edema. Inhalation at high concentrations may cause CNS depression and asphixiation.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Use water spray to keep fire-exposed containers cool. Combustible liquid.
Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Containers may explode when heated.
Extinguishing Media:
Use water spray to cool fire-exposed containers. Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Clean up spills immediately, observing precautions in the Protective Equipment section. Remove all sources of ignition. Use a spark-proof tool. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use only in a well-ventilated area. Avoid contact with eyes, skin, and clothing. Empty containers retain product residue, (liquid and/or vapor), and can be dangerous. Keep container tightly closed. Keep away from heat, sparks and flame. Avoid ingestion and inhalation. Do not pressurize, cut, weld, braze, solder, drill, grind, or expose empty containers to heat, sparks or open flames.
Storage:
Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate general or local explosion-proof ventilation to keep airborne levels to acceptable levels.
Exposure Limits CAS# 615-37-2: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear yellow
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 211 deg C @ 760.00 mmHg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not applicable.
Flash Point: 90 deg C ( 194.00 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.7130 g/cm3
Molecular Formula: C7H7I
Molecular Weight: 218.04
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, ignition sources, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen iodide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 615-37-2 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-Iodotoluene - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 615-37-2: No information available.
Canada
CAS# 615-37-2 is listed on Canada's NDSL List.
CAS# 615-37-2 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 615-37-2 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
用途
2-碘代甲苯是一种碘代芳烃类化合物,卤代芳烃化合物如碘代芳烃类化合物是重要的化工原料和中间体。以常见的碘苯为例,碘苯相比溴苯和氯苯具有更高的化学活性,在催化C-C键合成及C-杂原子键合反应中应用更广泛。
制备 方法一在装有磁搅拌子的圆底烧瓶中加入0.2 mL氧化亚铜/NH₃催化体系,135.8 mg邻甲基苯硼酸,0.830 g碘化钾以及2 mL水。室温下开放体系反应24小时后,向其中加入1.5 mL 2 mol/L的氢氧化钠溶液并用二氯甲烷萃取三次(每次2 mL),合并后的有机相经浓缩得到2-碘代甲苯176 mg,产率为81%。
方法二精确称取邻溴甲苯(42.8 mg, 0.25 mmol)、四甲基碘化铵(150.79 mg, 0.75 mmol)、氧化亚铜(3.6 mg, 0.025 mmol)和L-脯氨酸(5.8 mg, 0.05 mmol),依次加入到25 mL的Schlenk瓶中,加入精制过的乙醇(1.5 mL),置于110℃油浴中反应30小时。反应结束后,通过硅胶柱过滤至10 mL容量瓶,并用乙酸乙酯将溶液体积定量至指定刻度。使用气相色谱进行分析,2-碘代甲苯的产率为28%。
用途主要作为生物制药中的医药中间体。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-碘苄醇 2-iodobenzyl alcohol 5159-41-1 C7H7IO 234.036 2-碘苯甲基溴 2-Iodobenzyl bromide 40400-13-3 C7H6BrI 296.933 2-碘苄基碘 2-iodobenzyliodide 4622-38-2 C7H6I2 343.934 邻碘氯苄 2-(chloromethyl)iodobenzene 59473-45-9 C7H6ClI 252.482 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2,4-diiodo-toluene 32704-08-8 C7H6I2 343.934 1,3,5-三碘-2-甲基苯 2,4,6-triiodo-toluene 36994-79-3 C7H5I3 469.83 2-碘苯甲醛 2-iodobenzaldehyde 26260-02-6 C7H5IO 232.021 2-碘苄醇 2-iodobenzyl alcohol 5159-41-1 C7H7IO 234.036 2-碘苯甲基溴 2-Iodobenzyl bromide 40400-13-3 C7H6BrI 296.933 2-碘氰基苯 2-iodobenzonitrile 4387-36-4 C7H4IN 229.02 2-碘苄胺 2-iodobenzylamine 39959-51-8 C7H8IN 233.052 —— iodosyl-o-toluene 64297-59-2 C7H7IO 234.036 5-溴-2-碘甲苯 2-methyl-4-bromoiodobenzene 116632-39-4 C7H6BrI 296.933 2-碘异丙基苯 2-isopropyliodobenzene 19099-54-8 C9H11I 246.091 2-碘苯基乙腈 2-(2-iodophenyl)acetonitrile 40400-15-5 C8H6IN 243.047 2-碘d二苯基甲烷 1-benzyl-2-iodobenzene 35444-93-0 C13H11I 294.135 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:涉及碳-杂原子双键的 Heck 型反应。铑(I)-催化的芳基卤化物与N-吡唑醛亚胺的偶联摘要:DOI:10.1021/ja003306e
-
作为产物:参考文献:名称:在有电子转移催化剂存在或不存在的情况下,干燥的Arenediazonium o-Benzenedisulfonimides的卤代重氮化作用。制备芳基氯化物,溴化物和碘化物的简单通用程序。摘要:该论文报告了一项广泛研究的结果,该研究旨在通过将干燥的芳基重氮邻苯二磺酰亚胺1与卤化四烷基铵2进行卤代重氮化来制备芳基氯化物3(19例),溴化物4(19例)和碘化物5(9例)。在无水乙腈中,在室温(约20℃)下,在铜粉存在下,在60℃或室温下,在无催化剂的条件下进行。在最佳条件下,收率从良好到优异(60个反应,产率61-94%),只有少数例外(8个反应,产率51-55%)。总是从反应中回收大量的邻苯二磺酰亚胺(7),然后可以将其重新用于制备盐1。DOI:10.1021/jo9819537
-
作为试剂:描述:1,3,5-三甲基环己烷 在 十二羰基三钌 、 2-碘代甲苯 、 N,N'-1,2-tetrakis(4-fluorophenyl)ethane-1,2-diimine 、 caesium carbonate 作用下, 以 氯苯 为溶剂, 反应 24.0h, 以17%的产率得到1,3-二甲基-5-亚甲基环己烷参考文献:名称:通过分子间氢原子转移机制的钌催化脱氢摘要:烷烃直接脱氢是获得有价值的烯烃产品的最有效方法之一。尽管已经设计了几种催化剂来促进这种转变,但不幸的是它们在精细化学合成中的应用有限。在这里,我们报告了一种使用钌催化剂催化烷烃分子间脱氢的概念新颖的策略。氧化还原活性配体和空间位阻芳基自由基中间体的组合释放了这种新策略。重要的是,已经进行了机理研究,为进一步开发这种新型催化脱氢系统提供了概念框架。DOI:10.1002/anie.202015837
文献信息
-
Nickel(II) N‐Heterocyclic Carbene Complexes: Versatile Catalysts for C–C, C–S and C–N Coupling Reactions作者:Lourdes Benítez Junquera、Francys E. Fernández、M. Carmen Puerta、Pedro ValergaDOI:10.1002/ejic.201700057日期:2017.5.18A variety of Ni(II) complexes with a wide range of electronic and steric properties, bearing picolyl-imidazolidene ligands (a-g) and Cp (2a-f) or Cp* (3a,c,g) groups, have been synthesised and characterised using NMR and single crystal X-ray crystallography. The complexes have been used as precatalysts for a wide range of catalytic transformations most likely involving a Ni0/NiII catalytic cycle. In
-
Microwave-Assisted Simple and Efficient Ligand Free Copper Nanoparticle Catalyzed Aryl-Sulfur Bond Formation作者:Brindaban C. Ranu、Amit Saha、Ranjan JanaDOI:10.1002/adsc.200700289日期:2007.12.10A new protocol for the coupling of aryl iodides with thiophenols and alkanethiols catalyzed by copper nanoparticles under ligand-free condition has been developed. A variety of functionalized aryl sulfides are prepared in excellent yields under microwave irradiation for 5–7 min. A plausible radical mechanism has been suggested.
-
Pd(II)-Catalyzed Direct γ-C(sp<sup>3</sup>)-H Arylation between Free β<sup>2</sup>-Amino Esters and β<sup>3</sup>-Amino Esters and Aryl Iodides Using a Catalytic Transient Directing Group作者:Zhaohui Wang、Yangjie Fu、Qiyu Zhang、Hong Liu、Jiang WangDOI:10.1021/acs.joc.0c00115日期:2020.6.19Pd(II)-catalyzed direct γ-C(sp3)-H arylation coupling with free β2-amino esters and β3-amino esters using a commercially available catalytic transient directing group has been developed. This approach features high efficiency, broad substrate tolerance, easily accessible starting materials, and mild reaction conditions.
-
Combining Transition Metal Catalysis with Radical Chemistry: Dramatic Acceleration of Palladium-Catalyzed CH Arylation with Diaryliodonium Salts作者:Sharon R. Neufeldt、Melanie S. SanfordDOI:10.1002/adsc.201200738日期:2012.12.14This paper describes a photoredox palladium/iridium-catalyzed C-H arylation with diaryliodonium reagents. Details of the reaction optimization, substrate scope, and mechanism are presented along with a comparison to a related method in which aryldiazonium salts are used in place of diaryliodonium reagents. The unprecedentedly mild reaction conditions (25 masculineC in methanol), the requirement for
-
Two Stereoinduction Events in One C−H Activation Step: A Route towards Terphenyl Ligands with Two Atropisomeric Axes作者:Quentin Dherbassy、Jean‐Pierre Djukic、Joanna Wencel‐Delord、Françoise ColobertDOI:10.1002/anie.201801130日期:2018.4.16scaffolds—ortho‐orientated terphenyls presenting two atropisomeric Ar–Ar axes. These unusual structures were built up by using the C−H activation approach, and remarkably, both chiral axes were controlled with excellent stereoselectivity in a single transformation. During the reaction, not only does atroposelective functionalization of a biaryl precursor occur to establish one stereogenic axis, but an
表征谱图
-
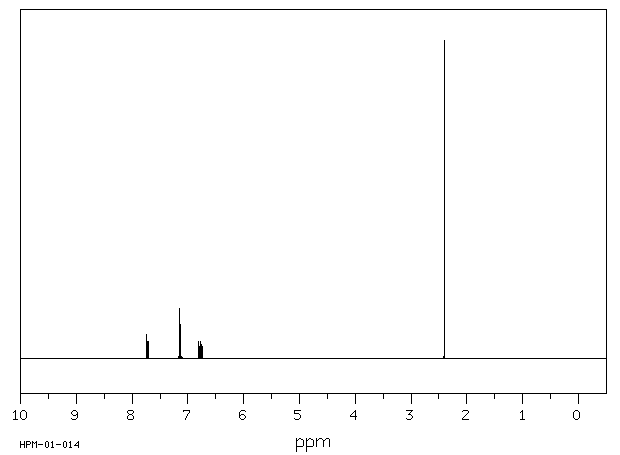
氢谱1HNMR
-
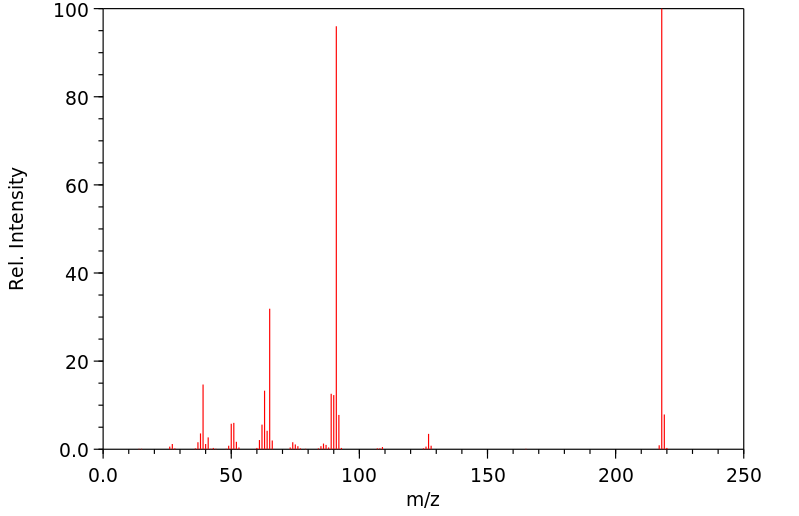
质谱MS
-
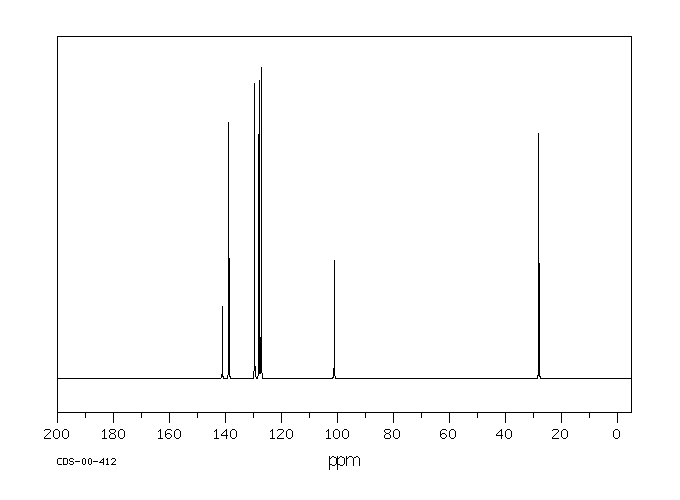
碳谱13CNMR
-
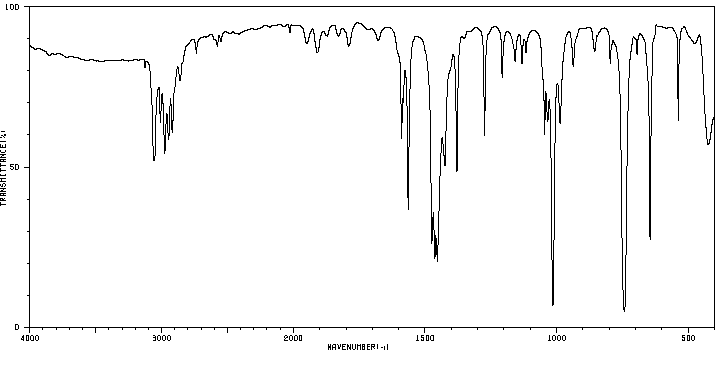
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫