5-氯-2-硝基三氟甲苯 | 118-83-2
中文名称
5-氯-2-硝基三氟甲苯
中文别名
2-硝基-5-氯三氟甲苯;2-三氟甲基-4-氯-硝基苯;4-氯-1-硝基-2-三氟甲基苯;5-氯-2-硝基三氟甲基苯;5-氯-2-硝基三氟苯
英文名称
5-chloro-2-nitrotrifluoromethylbenzene
英文别名
5-Chloro-2-nitrobenzotrifluoride;4-chloro-1-nitro-2-(trifluoromethyl)benzene
CAS
118-83-2
化学式
C7H3ClF3NO2
mdl
MFCD00007298
分子量
225.555
InChiKey
CFPIGEXZPWTNOR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:21 °C
-
沸点:222-224 °C
-
密度:1.526 g/mL at 25 °C(lit.)
-
闪点:217 °F
-
LogP:3.42 at 25℃
-
溶解度:4.43e-04 M
-
稳定性/保质期:
避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):3.8
-
重原子数:14
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.142
-
拓扑面积:45.8
-
氢给体数:0
-
氢受体数:5
安全信息
-
危险等级:6.1
-
危险品标志:Xn
-
安全说明:S26,S28,S36/37/39,S36/39
-
危险类别码:R20/21/22,R36/37/38
-
WGK Germany:2
-
海关编码:2904909090
-
危险品运输编号:2810
-
危险类别:6.1
-
包装等级:III
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:密封于阴凉、干燥处保存。确保工作间有良好的通风设施,并远离氧化剂。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 4-Chloro-1-nitro-2-(trifluoromethyl)benzene
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
Section 3. Composition/information on ingredients.
Ingredient name: 4-Chloro-1-nitro-2-(trifluoromethyl)benzene
CAS number: 118-83-2
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
Eye contact:
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
No data
Boiling point:
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C7H3ClF3NO2
Molecular weight: 225.6
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, hydrogen chloride, hydrogen fluoride.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN2810 Class: 6.1 Packing group: III
Proper shipping name: TOXIC, LIQUIDS, ORGANIC, N.O.S. OR TOXIC, LIQUIDS, ORGANIC, N.O.S. INHALA-
TION HAZARD, PACKING GROUP I, ZONE A OR B (4-Chloro-1-nitro-2-(trifluoromethyl)benzene)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 4-Chloro-1-nitro-2-(trifluoromethyl)benzene
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
Section 3. Composition/information on ingredients.
Ingredient name: 4-Chloro-1-nitro-2-(trifluoromethyl)benzene
CAS number: 118-83-2
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
Eye contact:
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
No data
Boiling point:
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C7H3ClF3NO2
Molecular weight: 225.6
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, hydrogen chloride, hydrogen fluoride.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN2810 Class: 6.1 Packing group: III
Proper shipping name: TOXIC, LIQUIDS, ORGANIC, N.O.S. OR TOXIC, LIQUIDS, ORGANIC, N.O.S. INHALA-
TION HAZARD, PACKING GROUP I, ZONE A OR B (4-Chloro-1-nitro-2-(trifluoromethyl)benzene)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 5-氨基-2-硝基三氟甲苯 4-nitro-3-(trifluoromethyl)benzeneamine 393-11-3 C7H5F3N2O2 206.124 —— 2-(trifluoromethyl)nitrosobenzene —— C7H4F3NO 175.11 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 5-氨基-2-硝基三氟甲苯 4-nitro-3-(trifluoromethyl)benzeneamine 393-11-3 C7H5F3N2O2 206.124 3-三氟甲基-4-硝基苯酚 3-trifluoromethyl-4-nitrophenol 88-30-2 C7H4F3NO3 207.109 4-硝基-3-(三氟甲基)苯硫酚 4-Nitro-3-trifluormethylthiophenol 39234-82-7 C7H4F3NO2S 223.175 2-氨基-5-氯三氟甲苯 4-chloro-2-(trifluoromethyl)aniline 445-03-4 C7H5ClF3N 195.572 4-硝基-3-(三氟甲基)苯腈 4-nitro-3-(trifluoromethyl)benzonitrile 320-36-5 C8H3F3N2O2 216.119 —— N-methyl-4'-nitro-3'-(trifluoromethyl)aniline 52807-22-4 C8H7F3N2O2 220.151 3-三氟甲基-4-硝基苯甲醚 4-methoxy-1-nitro-2-(trifluoromethyl)benzene 344-39-8 C8H6F3NO3 221.136 —— methyl(4-nitro-3-(trifluoromethyl)phenyl)sulfane 54998-75-3 C8H6F3NO2S 237.202 5-二甲氨-2-硝基三氟甲苯 N,N-dimethyl-4-nitro-3-(trifluoromethyl)aniline 41512-62-3 C9H9F3N2O2 234.178 1-硝基-4-[4-硝基-3-(三氟甲基)苯氧基]-2-(三氟甲基)苯 3,3'-bis(trifluoromethyl)-4,4'-dinitrodiphenyl ether 145854-47-3 C14H6F6N2O5 396.202 —— 2-[4-Nitro-3-(trifluoromethyl)phenyl]acetonitrile 176689-23-9 C9H5F3N2O2 230.146 —— 2-nitro-5-ethylthiobenzotrifluoride 68706-73-0 C9H8F3NO2S 251.229 —— N-[4-nitro-3-(trifluoromethyl)phenyl]propane-1,3-diamine 1032995-12-2 C10H12F3N3O2 263.219 —— (4-nitro-3-(trifluoromethyl)phenyl)pent-4-yn-1-ylamine —— C12H11F3N2O2 272.227 —— 5-(4-nitro-3-(trifluoromethyl)phenylamino)pentan-1-ol —— C12H15F3N2O3 292.258 4,4'-二硝基-3,3'-双(三氟甲基)联苯 4,4'-dinitro-3,3'-bis-trifluoromethyl-biphenyl 363-95-1 C14H6F6N2O4 380.203 —— 2-nitro-5-phenylthiobenzotrifluoride 59431-99-1 C13H8F3NO2S 299.273 —— di-(3-trifluoromethyl-4-nitro-phenyl) disulfide 27006-08-2 C14H6F6N2O4S2 444.335 4-氯-2-三氟甲基异氰酸酯 4-chloro-2-trifluoromethylphenylisocyanate 16588-69-5 C8H3ClF3NO 221.566 —— N-Cyclohexyl-4-nitro-3-(trifluoromethyl)aniline 61587-18-6 C13H15F3N2O2 288.27 N-(4-乙氧基苯基)-4-硝基-3-(三氟甲基)苯胺 N-(4-Ethoxyphenyl)-4-nitro-3-(trifluoromethyl)aniline 61587-16-4 C15H13F3N2O3 326.275 - 1
- 2
- 3
反应信息
-
作为反应物:描述:5-氯-2-硝基三氟甲苯 在 sodium hydroxide 、 苄基三乙基氯化铵 、 乙硫醇 作用下, 以 二氯甲烷 、 水 为溶剂, 生成 2-nitro-5-ethylthiobenzotrifluoride参考文献:名称:4-Alkylthio-2-trifluoromethylalkanesulfonanilides and derivatives thereof摘要:在对位被烷基硫醚、烷基亚砜或烷基砜基取代的2-三氟甲基烷磺酰苯胺及其农业可接受的盐是有用的除草剂和植物生长调节剂。公开号:US04341901A1
-
作为产物:描述:参考文献:名称:635.有机氟化合物。第一部分:三氟化苯的羟基衍生物摘要:DOI:10.1039/jr9490003016
文献信息
-
Synthesis of sulfilimines申请人:Monsanto Company公开号:US04578514A1公开(公告)日:1986-03-25Processes are disclosed for preparation of N-aryl-S,S-dihydrocarbylsulfilimines by reaction of phenylisocyanate compounds with hydrocarbyl sulfoxides. The sulfilimines can be rearranged to ortho-thioalkylene anilines and the reactions can be employed in a route for converting nitrobenzene compounds to ortho-thioalkylene anilines, which are useful intermediates for preparation of herbicidal compounds.
-
Preparation and Biological Activity of 2-(4-(Thiazol-2-yl)phenyl)propionic Acid Derivatives Inhibiting Cyclooxygenase.作者:Youichiro NAITO、Tomokazu GOTO、Fumihiko AKAHOSHI、Shiniciro ONO、Haruko YOSHITOMI、Tadashi OKANO、Naoki SUGIYAMA、Shunichi ABE、Syuichi HANADA、Mitsuru HIRATA、Masahiro WATANABE、Chikara FUKAYA、Kazumasa YOKOYAMA、Toshio FUJITADOI:10.1248/cpb.39.2323日期:——A series of 2-[4-(thiazol-2-yl)phenyl]propionic acids substituted at various positions were prepared by the reaction of diethyl 2-methyl-2-(4-thiocarbamoylphenyl)malonates with alpha-bromoaldehyde diethyl acetals or alpha-haloketones followed by hydrolysis of esters. The inhibition of prostaglandin H synthetase (cyclooxygenase) was assayed by use of an enzyme preparation from guinea pig polymorphonuclear通过使2-甲基-2-(4-硫代氨基甲酰基苯基)丙二酸二乙酯与α-溴醛二乙缩醛或α反应制备一系列在各个位置取代的2- [4-(噻唑-2-基)苯基]丙酸。 -卤代酮,然后酯水解。通过使用来自豚鼠多形核白细胞的酶制剂测定前列腺素H合成酶(环加氧酶)的抑制作用。这些化合物的结构活性关系的研究表明,苯环3位(R1)处的卤素和噻唑环4位(R2)和/或5位(R3)处的甲基被卤素取代是有利的抑制活性。在R2位带有大的烷基或极性官能团的化合物是弱抑制剂。测试了有效的环氧合酶抑制剂减轻角叉菜胶诱导的大鼠爪炎症的能力。这些衍生物由于对环加氧酶的强抑制作用而具有很强的抗炎活性,除了一些例外,包括那些在R1处带有硫代甲基的衍生物。
-
Tertiary aminoacids
-
A Homogeneous Method for the Conveniently Scalable Palladium- and Nickel-Catalyzed Cyanation of Aryl Halides作者:Finn Burg、Julian Egger、Johannes Deutsch、Nicolas GuimondDOI:10.1021/acs.oprd.6b00229日期:2016.8.19Homogeneous conditions for the palladium-catalyzed cyanation of aryl halides were developed. This new system features a broad scope of aryl chlorides and bromides, uses 2-propanol or 1-butanol as solvent, and is readily scalable. The same conditions can also provide simple benzonitriles using the recently developed (TMEDA)NiCl(o-tolyl) precatalyst in conjunction with 1,1′-bis(diphenylphosphino)ferrocene
-
<i>Para</i>-Selective Halogenation of Nitrosoarenes with Copper(II) Halides作者:Angela van der Werf、Nicklas SelanderDOI:10.1021/acs.orglett.5b03198日期:2015.12.18The para-selective direct bromination and chlorination of nitrosoarenes with copper(II) bromide and chloride is reported. Under mild reaction conditions, a range of halogenated arylnitroso compounds are obtained in moderate to good yields with high regioselectivity. Additionally, the versatility of the method is demonstrated by the development of a one-pot procedure to obtain the corresponding para-halogenated
表征谱图
-
氢谱1HNMR
-
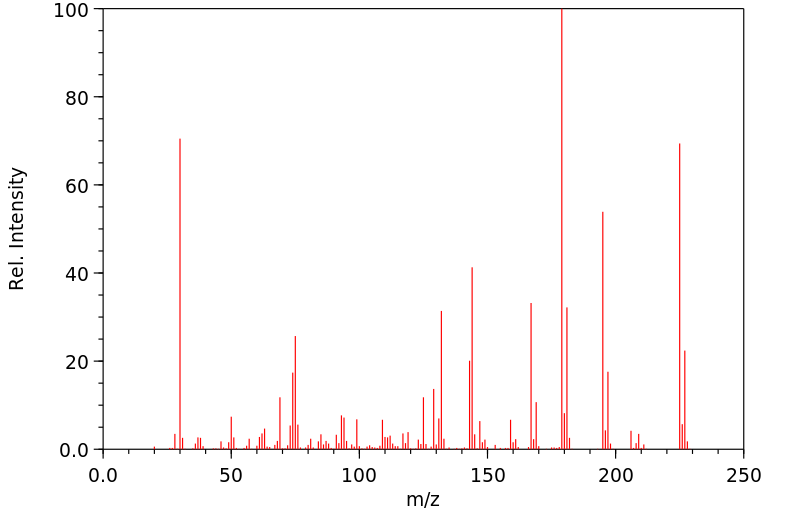
质谱MS
-
碳谱13CNMR
-
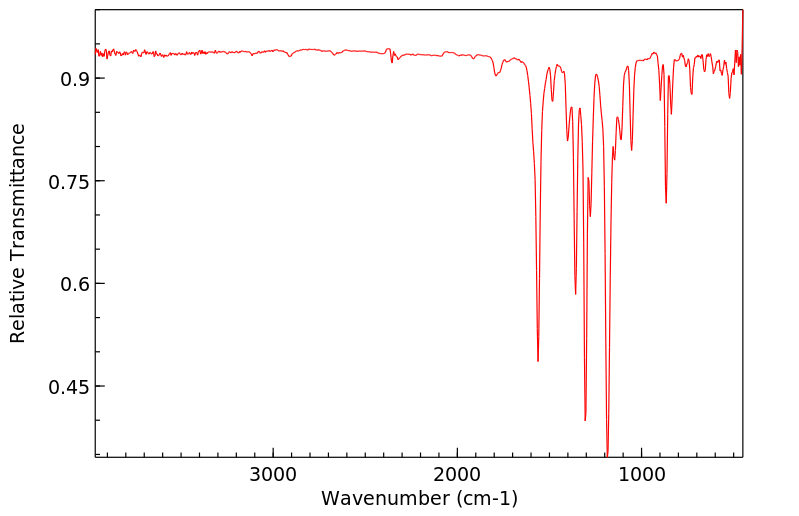
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫