乙酰水杨酸甲酯 | 580-02-9
中文名称
乙酰水杨酸甲酯
中文别名
乙酰柳酸甲酯;甲基阿斯匹林;2-(乙酰氧基)苯甲酸甲酯
英文名称
methyl acetylsalicylate
英文别名
methyl 2-acetoxybenzoate;aspirin methyl ester;5-acetylsalicylic acid methyl ester;methyl 2-acetyloxybenzoate
CAS
580-02-9
化学式
C10H10O4
mdl
MFCD00014978
分子量
194.187
InChiKey
ONWPLBKWMAUFGZ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:50°C
-
沸点:bp9 134-136°
-
密度:1.2534 (rough estimate)
-
溶解度:溶于甲醇
-
保留指数:1454
计算性质
-
辛醇/水分配系数(LogP):1.5
-
重原子数:14
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.2
-
拓扑面积:52.6
-
氢给体数:0
-
氢受体数:4
安全信息
-
危险品标志:F
-
WGK Germany:3
-
海关编码:2918229000
-
危险类别:3
-
安全说明:S15,S16,S29,S33
-
危险类别码:R11
-
包装等级:II
-
危险品运输编号:UN 1993 3/PG 2
-
危险性防范说明:P233,P260,P261,P264,P271,P280,P302+P352,P304,P304+P340,P305+P351+P338,P312,P321,P332+P313,P337+P313,P340,P362,P403,P403+P233,P405,P501
-
危险性描述:H315,H319,H335
-
储存条件:室温且干燥环境中使用。
SDS
乙酰水杨酸甲酯 修改号码:5
模块 1. 化学品
产品名称: Methyl Acetylsalicylate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害 未分类
健康危害 未分类
环境危害 未分类
GHS标签元素
图标或危害标志 无
信号词 无信号词
危险描述 无
防范说明 无
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 乙酰水杨酸甲酯
百分比: >98.0%(GC)
CAS编码: 580-02-9
俗名: 2-Acetoxybenzoic Acid Methyl Ester , Methyl 2-Acetoxybenzoate ,
Acetylsalicylic Acid Methyl Ester
分子式: C10H10O4
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,雾状水,二氧化碳
乙酰水杨酸甲酯 修改号码:5
模块 5. 消防措施
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:如果安全,移去可移动容器。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 清扫收集粉尘,封入密闭容器。注意切勿分散。附着物或收集物应该立即根据合适的
法律法规处置。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止粉尘扩散。处理后彻底清洗双手
和脸。
注意事项: 如果粉尘或浮质产生,使用局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗处。
远离不相容的材料比如氧化剂存放。
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统,操作人员切勿直接接触。同时安装淋浴器和洗
眼器。
个人防护用品
呼吸系统防护: 防尘面具。依据当地和政府法规。
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
固体
外形(20°C):
外观: 晶体-粉末
颜色: 白色类白色
气味: 无资料
pH: 无数据资料
熔点:
50°C
沸点/沸程 136 °C/1.2kPa
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 无资料
溶解度:
[水] 无资料
[其他溶剂]
溶于: 甲醇
乙酰水杨酸甲酯 修改号码:5
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在可燃溶剂中溶解混合,在装有后燃和洗涤装置的化学焚烧炉中
焚烧。废弃处置时请遵守国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 与联合国分类标准不一致
UN编号: 未列明
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
乙酰水杨酸甲酯 修改号码:5
模块16 - 其他信息
N/A
模块 1. 化学品
产品名称: Methyl Acetylsalicylate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害 未分类
健康危害 未分类
环境危害 未分类
GHS标签元素
图标或危害标志 无
信号词 无信号词
危险描述 无
防范说明 无
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 乙酰水杨酸甲酯
百分比: >98.0%(GC)
CAS编码: 580-02-9
俗名: 2-Acetoxybenzoic Acid Methyl Ester , Methyl 2-Acetoxybenzoate ,
Acetylsalicylic Acid Methyl Ester
分子式: C10H10O4
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,雾状水,二氧化碳
乙酰水杨酸甲酯 修改号码:5
模块 5. 消防措施
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:如果安全,移去可移动容器。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 清扫收集粉尘,封入密闭容器。注意切勿分散。附着物或收集物应该立即根据合适的
法律法规处置。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止粉尘扩散。处理后彻底清洗双手
和脸。
注意事项: 如果粉尘或浮质产生,使用局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗处。
远离不相容的材料比如氧化剂存放。
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统,操作人员切勿直接接触。同时安装淋浴器和洗
眼器。
个人防护用品
呼吸系统防护: 防尘面具。依据当地和政府法规。
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
固体
外形(20°C):
外观: 晶体-粉末
颜色: 白色类白色
气味: 无资料
pH: 无数据资料
熔点:
50°C
沸点/沸程 136 °C/1.2kPa
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 无资料
溶解度:
[水] 无资料
[其他溶剂]
溶于: 甲醇
乙酰水杨酸甲酯 修改号码:5
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在可燃溶剂中溶解混合,在装有后燃和洗涤装置的化学焚烧炉中
焚烧。废弃处置时请遵守国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 与联合国分类标准不一致
UN编号: 未列明
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
乙酰水杨酸甲酯 修改号码:5
模块16 - 其他信息
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 阿司匹林 aspirin 50-78-2 C9H8O4 180.16 2-甲氧基苯甲酸甲酯 2-methoxybenzoic acid methyl ester 606-45-1 C9H10O3 166.177 水杨酸甲酯 methyl salicylate 119-36-8 C8H8O3 152.15 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 阿司匹林 aspirin 50-78-2 C9H8O4 180.16 水杨酸甲酯 methyl salicylate 119-36-8 C8H8O3 152.15 邻乙酰水杨酰氯 O-acetylsalicyloyl chloride 5538-51-2 C9H7ClO3 198.606
反应信息
-
作为反应物:参考文献:名称:二苯脲衍生物,用于对抗耐甲氧西林和万古霉素的金黄色葡萄球菌。摘要:一类新型的二苯基脲被鉴定为具有抗菌谱的新型抗菌支架,该抗菌谱包括高度耐药的葡萄球菌分离株,即耐甲氧西林和耐万古霉素的金黄色葡萄球菌(MRSA和VRSA)。从在一侧带有氨基胍官能团且在另一环上带有正丁基部分的铅化合物3开始,制备了几种类似物。考虑到药代动力学参数是结构优化的关键因素,对亲脂性侧链的结构-活性-关系(SAR)进行了严格检查,从而发现了环庚氧基类似物21n作为潜在的候选药物。与万古霉素和利奈唑胺相比,该化合物具有几个显着优势,包括针对MRSA的快速杀伤动力学以及靶向并减轻MRSA在免疫细胞(巨噬细胞)内部的负担的能力。此外,使用秀丽隐杆线虫动物模型在体内证实了21n的有效抗MRSA活性。本研究为进一步开发二苯基脲化合物作为潜在的治疗剂提供了基础,以解决细菌对抗生素的耐药性迅速发展的挑战。DOI:10.1016/j.ejmech.2017.02.044
-
作为产物:参考文献:名称:Lapin,H.; Horeau,A., Gazzetta Chimica Italiana, 1963, vol. 93, p. 451 - 454摘要:DOI:
文献信息
-
Cleavage of Carboxylic Esters by Aluminum and Iodine作者:Dayong Sang、Huaxin Yue、Yang Fu、Juan TianDOI:10.1021/acs.joc.1c00034日期:2021.3.5A one-pot procedure for deprotecting carboxylic esters under nonhydrolytic conditions is described. Typical alkyl carboxylates are readily deblocked to the carboxylic acids by the action of aluminum powder and iodine in anhydrous acetonitrile. Cleavage of lactones affords the corresponding ω-iodoalkylcarboxylic acids. Aryl acetylates undergo deacetylation with the participation of the neighboring group
-
Evaluation of glycolamide esters and various other esters of aspirin as true aspirin prodrugs作者:Niels Moerk Nielsen、Hans BundgaardDOI:10.1021/jm00123a040日期:1989.3aryl esters of acetylsalicylic acid (aspirin) were synthesized and evaluated as potential prodrug forms of aspirin. N,N-Disubstituted glycolamide esters were found to be rapidly hydrolyzed in human plasma, resulting in the formation of aspirin as well as the corresponding salicylate esters. These in turn hydrolyzed rapidly to salicylic acid. The largest amount of aspirin formed from the esters were合成了一系列乙酰基水杨酸(阿司匹林)的乙醇酰胺,乙醇酸酯,(酰氧基)甲基,烷基和芳基酯,并将其评估为阿司匹林的潜在前药形式。N,N-二取代的乙醇酰胺酯被发现在人血浆中迅速水解,导致形成阿司匹林以及相应的水杨酸酯。这些反过来又迅速水解成水杨酸。在N,N-二甲基-和N,N-二乙基甘醇酰胺酯的情况下,由这些酯形成的最大量的阿司匹林分别为50和55%。用N,N-二甲基-和N,N-二乙基乙醇酰胺酯在血液中获得了相似的结果。未取代的和单取代的乙二酰胺酰胺酯以及大多数其他先前被认为是阿司匹林前体酯的酯已显示出仅水解为相应的水杨酸酯。测定了酯的亲脂性参数和水溶性,并讨论了有利于酯前药水解但以脱乙酰基为代价产生水杨酸酯的结构因素。关于阿司匹林的一些N,N-二取代的乙醇酰胺酯的特性,突出了它们作为潜在的阿司匹林前药的用途。
-
The importance of ester and alkoxy type functionalities for the chemo- and enantio-recognition of substrates by hydrolysis with Candida rugosa lipase作者:Francesca Bellezza、Antonio Cipiciani、Gabriele Cruciani、Francesco FringuelliDOI:10.1039/b005512n日期:——Racemic esters of 1-phenyl- and 1-(pyridyl)ethyl acetates 1a–c (R = Me, Ph) were subjected to hydrolysis in water in the presence of Candida rugosa lipase (CRL). 1-Pyridylethyl acetates (1, R = Me) are hydrolyzed by crude CRL (C-CRL) and isopropanol (isopropyl alcohol) treated CRL (PT-CRL) at very low rates, and the enantiorecognition was disappointing. By using 1-pyridylethyl benzoates (1, R = Ph) the results differed greatly: hydrolysis occurred much faster, and the enantiorecognition was good for 3- and 4-pyridyl derivatives and excellent for 2-pyridyl derivative. Analogous results were obtained by submitting the 1-phenylethanol esters 4 to enzymatic hydrolysis under the same experimental conditions. The hydrolysis of methyl o-acetoxybenzoates 7 (R = Me) gave quantitatively the deacetylated methyl o-hydroxybenzoates 9 (R = Me) by using either C-CRL or PT-CRL. A complete reversed selectivity is observed in the hydrolysis of phenyl o-acetoxybenzoates 7 (R = Ph) catalyzed by PT-CRL. Molecular modeling studies, aimed at probing the substrate specificity and the enantioselectivity of enzyme in terms of its three-dimensional structure is reported.1-苯基和1-(吡啶基)乙酸酯的消旋酯1a–c(R=甲基,苯基)在皱褶假丝酵母脂酶(CRL)存在下水解。1-吡啶基乙酸酯(1,R=甲基)在粗制CRL(C-CRL)和异丙醇(异丙基酒精)处理过的CRL(PT-CRL)作用下的水解速率非常低,且对映体识别令人失望。使用1-吡啶基苯甲酸酯(1,R=苯基)时结果大不相同:水解速率快得多,且对映体识别对于3-和4-吡啶衍生物良好,对于2-吡啶衍生物非常优异。在相同实验条件下进行1-苯乙醇酯4的酶促水解也得到了类似的结果。甲基邻乙酰氧基苯甲酸酯7(R=甲基)的水解在C-CRL或PT-CRL的作用下完全转化为脱乙酰化的甲基邻羟基苯甲酸酯9(R=甲基)。在PT-CRL催化的苯基邻乙酰氧基苯甲酸酯7(R=苯基)的水解中,观察到了完全相反的选择性。报道了旨在探究酶的三维结构对底物特异性和对映选择性的分子建模研究。
-
Enzymatic hydrolyses of acetoxy- and phenethylbenzoates by Candida cylindracea lipase作者:Antonio Cipiciani、Francesco Fringuelli、Anna Maria ScappiniDOI:10.1016/0040-4020(96)00519-4日期:1996.7The lipase from Candida cylindracea (CCL) deacetyles rapidly and selectively all three regioisomer methyl acetoxybenzoates. In the enzymatic hydrolyses of analogous aryl acetoxybenzoates, the difference of reactivity between the acetoxy and benzoyloxy functionalities is reduced and a methoxy group in meta position of the aryl group reverses the reactivity order making the compounds aspirin or aspirin-like
-
HYDROGEN SULFIDE DERIVATIVES OF NON-STEROIDAL ANTI-INFLAMMATORY DRUGS申请人:WALLACE JOHN公开号:US20080004245A1公开(公告)日:2008-01-03The present invention relates to derivatives of non-steroidal anti-inflammatory drugs (NSAIDs) having improved anti-inflammatory properties useful in the treatment of inflammation, pain and fever. More particularly, NSAIDs are derivatized with a hydrogen sulfide (H 2 S) releasing moiety to produce novel anti-inflammatory compounds having reduced side effects.
表征谱图
-
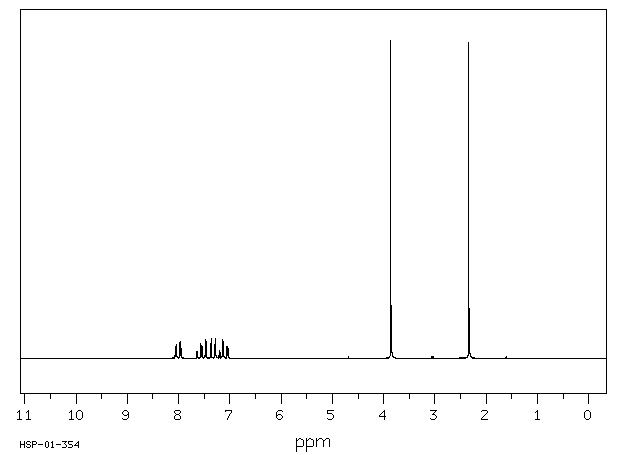
氢谱1HNMR
-
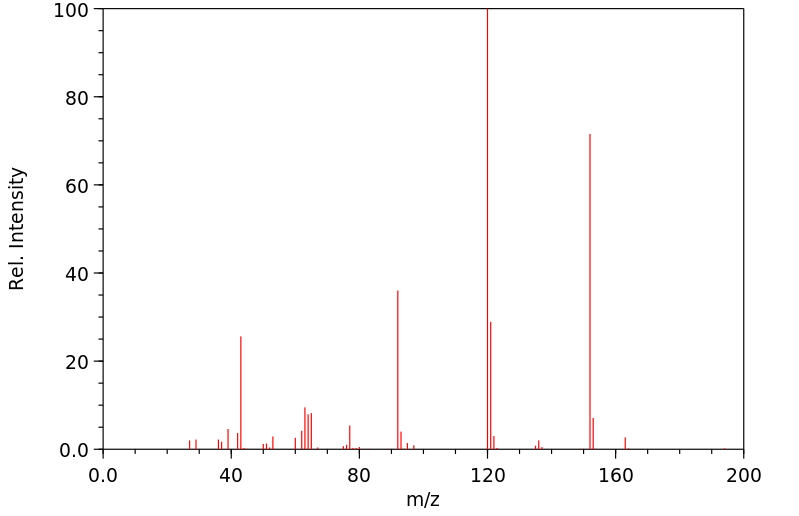
质谱MS
-
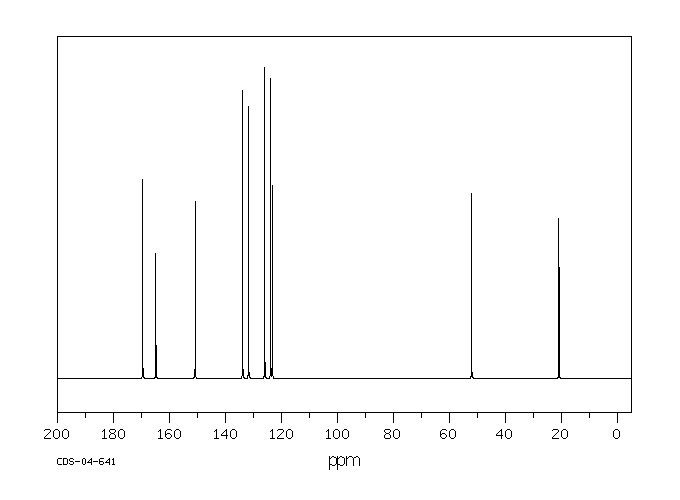
碳谱13CNMR
-
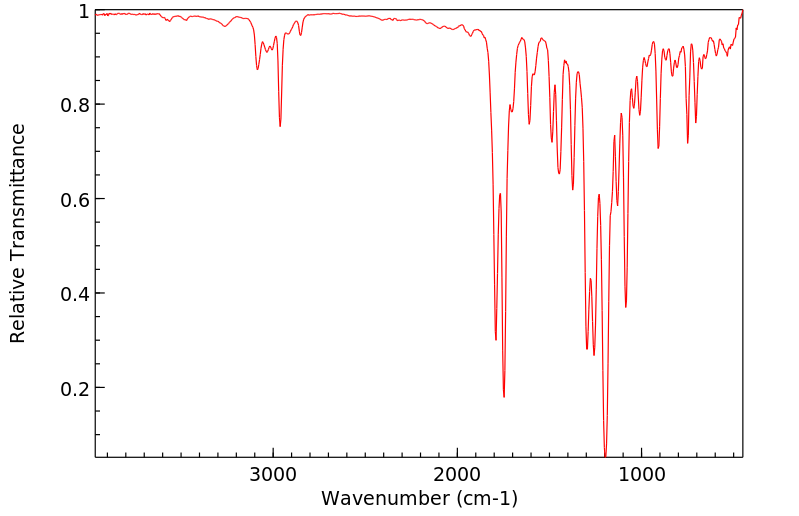
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫