6-氯己酰氯 | 19347-73-0
中文名称
6-氯己酰氯
中文别名
——
英文名称
6-chlorohexanoyl chloride
英文别名
——
CAS
19347-73-0
化学式
C6H10Cl2O
mdl
MFCD00041436
分子量
169.051
InChiKey
WZILXAPNPKMOSA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:90°C 7mm
-
密度:1.400 g/mL at 25 °C (lit.)
-
闪点:>230 °F
-
稳定性/保质期:
在常温常压下稳定,应避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):2.4
-
重原子数:9
-
可旋转键数:5
-
环数:0.0
-
sp3杂化的碳原子比例:0.833
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
危险等级:8
-
危险品标志:C
-
安全说明:S26,S36/37/39,S45
-
危险类别码:R34
-
WGK Germany:3
-
海关编码:2915900090
-
包装等级:II
-
危险品运输编号:UN 3265 8/PG 2
-
危险类别:8
-
危险性防范说明:P301+P330+P331,P303+P361+P353,P363,P304+P340,P310,P321,P260,P264,P280,P305+P351+P338,P405,P501
-
危险性描述:H314
-
储存条件:常温下应密闭避光保存,并保持通风和干燥。
SDS
Section 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE
Product identifiers
Product name : 6-Chlorohexanoyl chloride
CAS-No. : 19347-73-0
Relevant identified uses of the substance or mixture and uses advised against
Identified uses : Laboratory chemicals, Manufacture of substances
Section 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
Classification according to Regulation (EC) No 1272/2008 [EU-GHS/CLP]
Skin corrosion (Category 1B)
Serious eye damage (Category 1)
Classification according to EU Directives 67/548/EEC or 1999/45/EC
Reacts violently with water. Causes burns.
Label elements
Labelling according Regulation (EC) No 1272/2008 [CLP]
Pictogram
Signal word Danger
Hazard statement(s)
H314 Causes severe skin burns and eye damage.
Precautionary statement(s)
P280 Wear protective gloves/ protective clothing/ eye protection/ face
protection.
P305 + P351 + P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove
contact lenses, if present and easy to do. Continue rinsing.
P310 Immediately call a POISON CENTER or doctor/ physician.
Supplemental Hazard information (EU)
EUH014 Reacts violently with water.
According to European Directive 67/548/EEC as amended.
Hazard symbol(s)
R-phrase(s)
R14 Reacts violently with water.
R34 Causes burns.
S-phrase(s)
S26 In case of contact with eyes, rinse immediately with plenty of water and
seek medical advice.
S36/37/39 Wear suitable protective clothing, gloves and eye/face protection.
S45 In case of accident or if you feel unwell, seek medical advice immediately
(show the label where possible).
Other hazards
Lachrymator.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
Formula : C6H10Cl2O
Molecular Weight : 169,05 g/mol
Component Concentration
6-Chlorohexanoyl chloride
CAS-No. 19347-73-0 -
EC-No. 242-979-9
Section 4. FIRST AID MEASURES
Description of first aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician.
In case of skin contact
Take off contaminated clothing and shoes immediately. Wash off with soap and plenty of water. Consult a
physician.
In case of eye contact
Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician.
If swallowed
Do NOT induce vomiting. Never give anything by mouth to an unconscious person. Rinse mouth with
water. Consult a physician.
Most important symptoms and effects, both acute and delayed
Material is extremely destructive to tissue of the mucous membranes and upper respiratory tract, eyes, and
skin., spasm, inflammation and edema of the larynx, spasm, inflammation and edema of the bronchi,
pneumonitis, pulmonary edema, burning sensation, Cough, wheezing, laryngitis, Shortness of breath,
Headache, Nausea
Indication of any immediate medical attention and special treatment needed
no data available
Section 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media
Dry powder
Special hazards arising from the substance or mixture
Carbon oxides
Advice for firefighters
Wear self contained breathing apparatus for fire fighting if necessary.
Further information
no data available
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid breathing vapors, mist or gas. Ensure adequate ventilation.
Evacuate personnel to safe areas.
Environmental precautions
Do not let product enter drains.
Methods and materials for containment and cleaning up
Soak up with inert absorbent material and dispose of as hazardous waste. Do not flush with water. Keep
in suitable, closed containers for disposal.
Reference to other sections
For disposal see section 13.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Avoid inhalation of vapour or mist.
Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Containers which are
opened must be carefully resealed and kept upright to prevent leakage.
Never allow product to get in contact with water during storage.
Specific end use(s)
no data available
Section 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Control parameters
Components with workplace control parameters
Exposure controls
Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and
at the end of workday.
Personal protective equipment
Eye/face protection
Tightly fitting safety goggles. Faceshield (8-inch minimum). Use equipment for eye protection
tested and approved under appropriate government standards such as NIOSH (US) or EN
166(EU).
Skin protection
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique
(without touching glove's outer surface) to avoid skin contact with this product. Dispose of
contaminated gloves after use in accordance with applicable laws and good laboratory practices.
Wash and dry hands.
The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and
the standard EN 374 derived from it.
Body Protection
Complete suit protecting against chemicals, Flame retardant protective clothing, The type of
protective equipment must be selected according to the concentration and amount of the
dangerous substance at the specific workplace.
Respiratory protection
Where risk assessment shows air-purifying respirators are appropriate use a full-face respirator
with multi-purpose combination (US) or type ABEK (EN 14387) respirator cartridges as a backup
to engineering controls. If the respirator is the sole means of protection, use a full-face supplied air
respirator. Use respirators and components tested and approved under appropriate government
standards such as NIOSH (US) or CEN (EU).
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
a) Appearance Form: clear, liquid
Colour: colourless
b) Odour no data available
c) Odour Threshold no data available
d) pH no data available
e) Melting point/freezing no data available
point
f) Initial boiling point and 101 °C at 8 hPa130 °C at 27 hPa
boiling range
g) Flash point 110 °C - closed cup
h) Evaporation rate no data available
i) Flammability (solid, gas) no data available
j) Upper/lower no data available
flammability or
explosive limits
k) Vapour pressure no data available
l) Vapour density no data available
m) Relative density 1,4 g/cm3 at 25 °C
n) Water solubility no data available
o) Partition coefficient: n- no data available
octanol/water
p) Auto-ignition no data available
temperature
q) Decomposition no data available
temperature
r) Viscosity no data available
s) Explosive properties no data available
t) Oxidizing properties no data available
Other safety information
no data available
Section 10. STABILITY AND REACTIVITY
Reactivity
no data available
Chemical stability
no data available
Possibility of hazardous reactions
Reacts violently with water.
Conditions to avoid
Exposure to moisture.
Incompatible materials
Strong oxidizing agentsWater, Alcohols, Oxidizing agents, Strong bases
Hazardous decomposition products
Other decomposition products - no data available
Section 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity
no data available
Skin corrosion/irritation
no data available
Serious eye damage/eye irritation
no data available
Respiratory or skin sensitization
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
IARC: No component of this product present at levels greater than or equal to 0.1% is identified as
probable, possible or confirmed human carcinogen by IARC.
Reproductive toxicity
no data available
Specific target organ toxicity - single exposure
no data available
Specific target organ toxicity - repeated exposure
no data available
Aspiration hazard
no data available
Potential health effects
Inhalation May be harmful if inhaled. Material is extremely destructive to the tissue of
the mucous membranes and upper respiratory tract.
Ingestion May be harmful if swallowed. Causes burns.
Skin May be harmful if absorbed through skin. Causes skin burns.
Eyes Causes eye burns.
Signs and Symptoms of Exposure
Material is extremely destructive to tissue of the mucous membranes and upper respiratory tract, eyes, and
skin., spasm, inflammation and edema of the larynx, spasm, inflammation and edema of the bronchi,
pneumonitis, pulmonary edema, burning sensation, Cough, wheezing, laryngitis, Shortness of breath,
Headache, Nausea
Additional Information
RTECS: Not available
Section 12. ECOLOGICAL INFORMATION
Toxicity
no data available
Persistence and degradability
no data available
Bioaccumulative potential
no data available
Mobility in soil
no data available
Results of PBT and vPvB assessment
no data available
Other adverse effects
no data available
Section 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company.
Contaminated packaging
Dispose of as unused product.
Section 14. TRANSPORT INFORMATION
UN number
ADR/RID: 3265 IMDG: 3265 IATA: 3265
UN proper shipping name
ADR/RID: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S. (6-Chlorohexanoyl chloride)
IMDG: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S. (6-Chlorohexanoyl chloride)
IATA: Corrosive liquid, acidic, organic, n.o.s. (6-Chlorohexanoyl chloride)
Transport hazard class(es)
ADR/RID: 8 IMDG: 8 IATA: 8
Packaging group
ADR/RID: II IMDG: II IATA: II
Environmental hazards
ADR/RID: no IMDG Marine Pollutant: no IATA: no
Special precautions for user
no data available
Section 15. REGULATORY INFORMATION
This safety datasheet complies with the requirements of Regulation (EC) No. 1907/2006.
Safety, health and environmental regulations/legislation specific for the substance or mixture
no data available
Chemical Safety Assessment
no data available
Section 16. OTHER INFORMATION
Further information
Copyright 2012 Co. LLC. License granted to make unlimited paper copies for internal use
only.
The above information is believed to be correct but does not purport to be all inclusive and shall be
used only as a guide. The information in this document is based on the present state of our knowledge
and is applicable to the product with regard to appropriate safety precautions. It does not represent any
guarantee of the properties of the product. Corporation and its Affiliates shall not be held
liable for any damage resulting from handling or from contact with the above product. See
and/or the reverse side of invoice or packing slip for additional terms and conditions of sale.
上下游信息
反应信息
-
作为反应物:参考文献:名称:维拉佐酮的合成研究摘要:以 4-氰基苯胺和 5-溴-2-羟基苯甲醛为起始原料,描述了一种合成维拉佐酮的新途径,总产率为 24%,纯度为 99%。首先,通过 4-氰基苯胺的重氮化合成中间体 (3-(4-氯丁基)-1H-indole-5-carbonitrile),然后用 6-氯己醛进行 Fischer 吲哚环化。随后,通过用哌嗪对 5-溴苯并呋喃-2-甲酰胺进行芳香族亲核取代,生成另一种中间体 5-(哌嗪-1-基) 苯并呋喃-2-甲酰胺。最后,通过用 Et3N/K2CO3 处理上述两个关键中间体的亲核取代获得维拉佐酮。与原始工艺相比,该路线避免了使用昂贵且有毒的试剂,并解决了安全、环境问题和高成本等问题。DOI:10.1177/1747519819893293
-
作为产物:参考文献:名称:合成和评估一些稳定的多底物加合物作为亚精胺合酶的特异性抑制剂。摘要:设计了一系列新的氨基丙基转移酶抑制剂,其中亲核氨基丙基受体与氨基丙基供体S-腺苷-1-(甲硫基)-3-丙胺(脱羧的S-腺苷甲硫氨酸)连接,形成“多底物加合物”。在当前情况下,已经合成了S-腺苷-1,8-二氨基-3-硫辛烷(2b)和相应的甲基thy盐(3b)。分析了几种这类化合物作为亚精胺合酶的抑制剂,发现2b和3b均为该酶的有效抑制剂。硫醚2b是迄今为止描述的最有效的亚精胺合酶抑制剂,几乎完全没有针对紧密相关的氨丙基转移酶精胺合酶的抑制活性。DOI:10.1021/jm00143a003
文献信息
-
1,3-Diketones from Acid Chlorides and Ketones: A Rapid and General One-Pot Synthesis of Pyrazoles作者:Stephen T. Heller、Swaminathan R. NatarajanDOI:10.1021/ol060570p日期:2006.6.1[reaction: see text] 1,3-Diketones were synthesized directly from ketones and acid chlorides and were then converted in situ into pyrazoles by the addition of hydrazine. This method is extremely fast, general, and chemoselective, allowing for the synthesis of previously inaccessible pyrazoles and synthetically demanding pyrazole-containing fused rings.
-
SENSITIVE OLIGONUCLEOTIDE SYNTHESIS USING SULFUR-BASED FUNCTIONS AS PROTECTING GROUPS AND LINKERS申请人:Fang Shiyue公开号:US20210032281A1公开(公告)日:2021-02-04Embodiments for the synthesis of sensitive oligonucleotides as well as insensitive oligonucleotides are provided. Sulfur-based groups are used for the protection of exo-amino groups of nucleobases, phosphate groups and 2′-OH groups, and as cleavable linker for linking oligonucleotides to a support. Oligonucleotide syntheses are achieved under typical conditions using phosphoramidite chemistry with important modifications. To prevent replacing sulfur-based protecting groups by acyl groups via cap-exchange, special capping agents are used. To retain hydrophobic tag to assist RP HPLC purification, special phosphoramidites are used in the last synthetic cycle. With the sulfur-based groups for protection and linking, oligonucleotide deprotection and cleavage are achieved via oxidation followed by beta-elimination under mild conditions. Therefore, besides for insensitive oligonucleotide synthesis, the embodiments of the invention are capable for the synthesis of oligonucleotide analogs containing sensitive functional groups that cannot survive the harsh conditions used in prior art oligonucleotide synthesis technologies.本发明提供了用于合成敏感寡核苷酸和不敏感寡核苷酸的实施方案。使用基于硫的基团来保护核苷酸碱基的exo-氨基团、磷酸基团和2′-OH基团,并且作为可裂解的连接剂将寡核苷酸连接到支持物上。寡核苷酸的合成是在典型条件下使用磷酰亚胺化学进行的,并进行了重要的修改。为了防止通过帽交换将基于硫的保护基团替换为酰基,使用了特殊的封端剂。为了保留亲脂性标签以协助反相高效液相色谱(RP HPLC)纯化,在最后的合成周期中使用了特殊的磷酰亚胺。使用基于硫的基团进行保护和连接,寡核苷酸的脱保护和裂解是通过氧化随后在温和条件下进行beta-消除实现的。因此,除了用于不敏感寡核苷酸的合成外,本发明的实施方案还能够合成包含在先前的寡核苷酸合成技术中使用的严酷条件下无法存活的敏感功能团的寡核苷酸类似物。
-
HIV INTEGRASE INHIBITORS申请人:ViiV Healthcare Company公开号:US20150225399A1公开(公告)日:2015-08-13The present invention features compounds that are HIV integrase inhibitors and therefore are useful in the inhibition of HIV replication, the prevention and/or treatment of infection by HIV, and in the treatment of AIDS and/or ARC.本发明具有作为HIV整合酶抑制剂的化合物,因此在抑制HIV复制、预防及/或治疗HIV感染以及治疗艾滋病及/或艾滋病相关综合症方面具有用途。
-
[EN] BROMODOMAIN INHIBITORS<br/>[FR] INHIBITEURS DE BROMODOMAINE申请人:ABBVIE INC公开号:WO2018068283A1公开(公告)日:2018-04-19Provided herein are compounds of formula (I) wherein R 1, Y, L 1, G 1, X 1, X 2, L 2, R 2, R 3, and R 4 have any of the values defined in the specification, and pharmaceutically acceptable salts thereof, which are useful as agents in the treatment of diseases and conditions, including inflammatory diseases, cancer, and AIDS. Also provided are pharmaceutical compositions comprising compounds of formula (I).本文提供了式(I)的化合物,其中R 1、Y、L 1、G 1、X 1、X 2、L 2、R 2、R 3和R 4具有规范中定义的任何值,以及其药学上可接受的盐,这些化合物在治疗疾病和病况中是有用的,包括炎症性疾病、癌症和艾滋病。还提供了包含式(I)化合物的药物组合物。
-
Chemoselective Intermolecular Cross-Enolate-Type Coupling of Amides作者:Daniel Kaiser、Christopher J. Teskey、Pauline Adler、Nuno MaulideDOI:10.1021/jacs.7b08813日期:2017.11.15A new approach for the synthesis of 1,4-dicarbonyl compounds is reported. Chemoselective activation of amide carbonyl functionality and subsequent umpolung viaN-oxide addition generates an electrophilic enolonium species that can be coupled with a wide range of nucleophilic enolates. The method conveys broad functional group tolerance on both components, does not suffer from formation of homocoupling
表征谱图
-
氢谱1HNMR
-
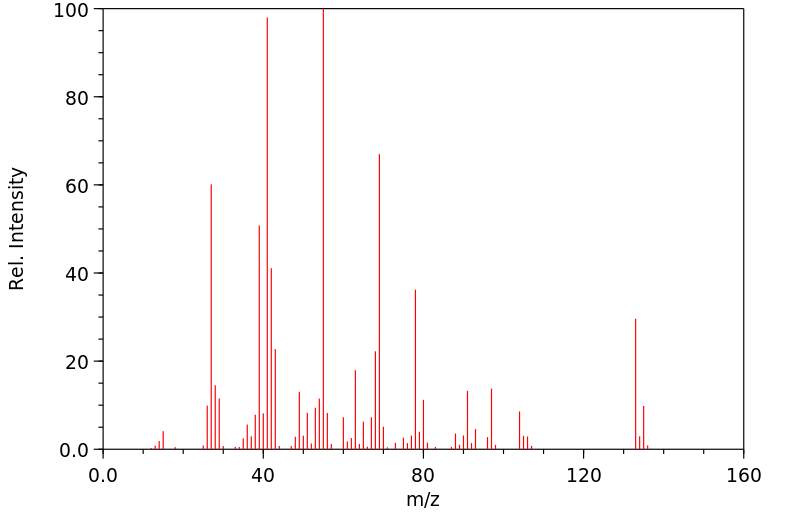
质谱MS
-
碳谱13CNMR
-
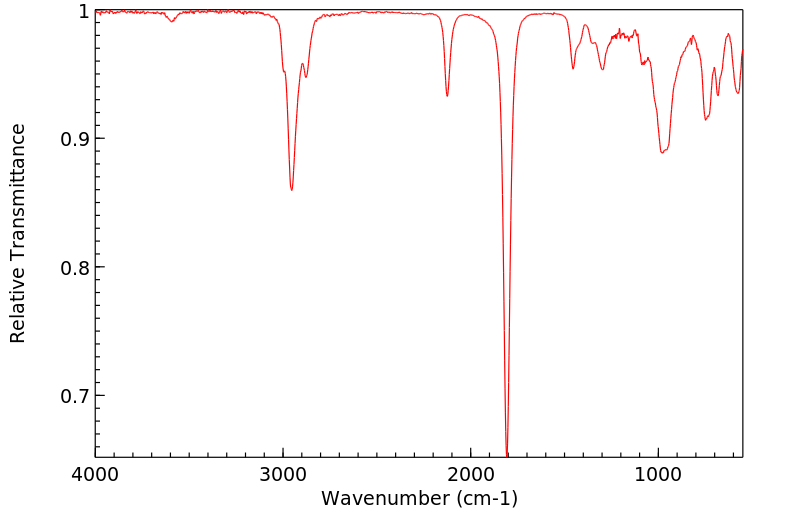
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-六氢-3a(1H)-并环戊二烯羰基氯化物
金刚烷酰氯
辛酰溴
辛酰氯
辛酰氟
辛-5t-烯酰氯
衣康酰氯
螺[3.5]壬烷-2-甲酰氯
螺[3.4]辛烷-2-甲酰氯
草酰溴
草酰氯
草酰氟
花生酰氯
花生四烯酰氯
肉豆蔻酰氯
肉豆蔻酰-1-13C氯
癸酰氯
癸基二酰二氯
癸二酰氯
異丁醯溴
甲酰氯
甲酰基溴化物
甲氧基乙酰氯
甲基丙烯酰氯
甲基丙烯酰氟
环辛烷羰基氯化物
环戊烷羰基溴
环戊烷羰基氟
环戊基甲酰氯
环戊基乙酰氯
环戊基乙酰氯
环庚烷羰酰氯
环己酰溴
环己甲酰氯
环己基乙酰氯
环己-3-烯-1-甲酰氯
环丙烷乙酰氯
环丙烷丙酰氯
环丁烷羰基碘化物
环丁烷羰基溴化物
环丁烷-1,2-二甲酰氯
环丁基甲酰氯
溴二氯乙酰氯
溴乙酰溴
溴乙酰氯
溴(二氟)乙酰氯
油酰氯
氰基乙酰溴
氯碘乙酰氯
氯氟乙酰氟