2,4,6-三异丙基苄氯 | 38580-86-8
中文名称
2,4,6-三异丙基苄氯
中文别名
2,4,6-三异丙基氯苄
英文名称
2,4,6-triisopropylbenzyl chloride
英文别名
1-(chloromethyl)-2,4,6-triisopropylbenzene;2-(chloromethyl)-1,3,5-tri(propan-2-yl)benzene
CAS
38580-86-8
化学式
C16H25Cl
mdl
MFCD03701106
分子量
252.827
InChiKey
CQHQLMOCMQMZPP-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:36 °C
-
沸点:126-128 °C(Press: 3.5 Torr)
-
密度:0.945±0.06 g/cm3(Predicted)
-
稳定性/保质期:
常规情况下不会分解,也没有危险的反应。
计算性质
-
辛醇/水分配系数(LogP):5.7
-
重原子数:17
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.625
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
海关编码:2903999090
-
危险品运输编号:UN 3261
-
储存条件:密封、阴凉、干燥处保存。
SDS
2,4,6-Triisopropylbenzyl Chloride Revision number: 5
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 2,4,6-Triisopropylbenzyl Chloride
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Category 1
Corrosive to metals
HEALTH HAZARDS
Category 1C
Skin corrosion/irritation
Serious eye damage/eye irritation Category 1
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements May be corrosive to metals
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep only in original container.
Do not breathe dust/fume/gas/mist/vapours/spray.
Wash hands thoroughly after handling.
Wear protective gloves/eye protection/face protection.
[Response] IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
breathing.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Absorb spillage to prevent material damage.
[Storage] Store locked up.
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
2,4,6-Triisopropylbenzyl Chloride
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: 2,4,6-Triisopropylbenzyl Chloride
Percent: >98.0%(GC)
CAS Number: 38580-86-8
Synonyms: 2-Chloromethyl-1,3,5-triisopropylbenzene
Chemical Formula: C16H25Cl
Section 4. FIRST AID MEASURES
Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Inhalation:
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Ingestion: Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
induce vomiting.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (P3 filter respirator for toxic particles). Keep
protective equipment and people away from and upwind of spill/leak. Entry to non-involved personnel should
emergency procedures: be controlled around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a closed system if possible. Use a local exhaust if dust or aerosol will be
generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Use corrosive resistant equipment.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a refrigerator.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Comply with laws. Keep only in original container.
Packaging material:
2,4,6-Triisopropylbenzyl Chloride
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator, self-contained breathing apparatus(SCBA), supplied air respirator,
etc. Use respirators approved under appropriate government standards and follow
local and national regulations.
Hand protection: Impervious gloves.
Safety goggles. A face-shield, if the situation requires.
Eye protection:
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Crystal- Powder
Form:
Colour: White - Very pale yellow
No data available
Odour:
pH: No data available
Melting point/freezing point:36°C
Boiling point/range: No data available
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: No data available
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen chloride
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
No data available
IARC =
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
2,4,6-Triisopropylbenzyl Chloride
Section 12. ECOLOGICAL INFORMATION
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 8: Corrosive.
UN-No: 3261
Proper shipping name: Corrosive solid, acidic, organic, n.o.s.
Packing group: III
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 2,4,6-Triisopropylbenzyl Chloride
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Category 1
Corrosive to metals
HEALTH HAZARDS
Category 1C
Skin corrosion/irritation
Serious eye damage/eye irritation Category 1
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements May be corrosive to metals
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep only in original container.
Do not breathe dust/fume/gas/mist/vapours/spray.
Wash hands thoroughly after handling.
Wear protective gloves/eye protection/face protection.
[Response] IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
breathing.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Absorb spillage to prevent material damage.
[Storage] Store locked up.
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
2,4,6-Triisopropylbenzyl Chloride
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: 2,4,6-Triisopropylbenzyl Chloride
Percent: >98.0%(GC)
CAS Number: 38580-86-8
Synonyms: 2-Chloromethyl-1,3,5-triisopropylbenzene
Chemical Formula: C16H25Cl
Section 4. FIRST AID MEASURES
Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Inhalation:
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Ingestion: Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
induce vomiting.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (P3 filter respirator for toxic particles). Keep
protective equipment and people away from and upwind of spill/leak. Entry to non-involved personnel should
emergency procedures: be controlled around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a closed system if possible. Use a local exhaust if dust or aerosol will be
generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Use corrosive resistant equipment.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a refrigerator.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Comply with laws. Keep only in original container.
Packaging material:
2,4,6-Triisopropylbenzyl Chloride
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator, self-contained breathing apparatus(SCBA), supplied air respirator,
etc. Use respirators approved under appropriate government standards and follow
local and national regulations.
Hand protection: Impervious gloves.
Safety goggles. A face-shield, if the situation requires.
Eye protection:
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Crystal- Powder
Form:
Colour: White - Very pale yellow
No data available
Odour:
pH: No data available
Melting point/freezing point:36°C
Boiling point/range: No data available
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: No data available
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen chloride
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
No data available
IARC =
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
2,4,6-Triisopropylbenzyl Chloride
Section 12. ECOLOGICAL INFORMATION
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 8: Corrosive.
UN-No: 3261
Proper shipping name: Corrosive solid, acidic, organic, n.o.s.
Packing group: III
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
自组装材料与接触印刷材料
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— (2,4,6-triisopropylphenyl)methanol 4276-88-4 C16H26O 234.382 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2.4.6-Tri-i-propyltoluol 6319-85-3 C16H26 218.382 —— 1,3,5-Triisopropyl-2,4-dimethylbenzol 57440-46-7 C17H28 232.409 —— 1-ethyl-2,4,6-tri-isopropylbenzene 81667-55-2 C17H28 232.409 —— 2,4,6-triisopropylbenzyl cyanide 4276-84-0 C17H25N 243.392 —— 3-Brom-2,4,6-triisopropylbenzylchlorid 57190-28-0 C16H24BrCl 331.724 —— (2,4,6-triisopropylphenyl)methanol 4276-88-4 C16H26O 234.382 —— 2,4,6-triisopropylbenzyl mercaptan 184898-33-7 C16H26S 250.448 —— 1,2-Bis-(2,4,6-triisopropyl-phenyl)-ethan 4276-86-2 C32H50 434.749 2,4,6-三异丙基苯乙酸 (2,4,6-Triisopropyl-phenyl)-essigsaeure 4276-85-1 C17H26O2 262.392
反应信息
-
作为反应物:描述:参考文献:名称:Rearrangement of 2,4,6-Triisopropylbenzyltrimethylammonium Ion by Sodium Amide to Form an exo-Methylenecyclohexadieneamine and its Reactions1摘要:DOI:10.1021/ja01577a051
-
作为产物:描述:参考文献:名称:Wagner, Peter J.; Meador, Michael A.; Zhou, Boli, Journal of the American Chemical Society, 1991, vol. 113, # 25, p. 9630 - 9639摘要:DOI:
文献信息
-
Chiral monodentate phosphine ligands for the enantioselective α- and γ-arylation of aldehydes作者:Ivan Franzoni、Laure Guénée、Clément MazetDOI:10.1016/j.tet.2014.02.079日期:2014.7phosphine ligands elaborated on the binepine scaffold is described. Their application in the Pd-catalyzed intramolecular asymmetric α-arylation of aldehydes and the intermolecular asymmetric γ-arylation of α,β-unsaturated aldehydes provides a mean of validating the design of these ligands. For the first reaction, excellent reactivities have been obtained while only modest enantioselectivities were measured
-
Ligand‐Facilitated Reductive Coupling of Benzyl Chlorides with Aryl Chlorides Catalyzed by Well‐Defined Heteroleptic Ni (II)‐NHC Complexes作者:Gusheng Lu、Ruipeng Li、Zhengwang Shen、Qinjia Wu、Hongmei SunDOI:10.1002/aoc.5741日期:2020.93‐bis(2,6‐diisopropylphenyl)imidazol‐2‐ylidene), complex 1 exhibited superior catalytic activity in the magnesium‐mediated reductive coupling of benzyl chlorides with aryl chlorides, featuring outstanding tolerance of both coupling partners with steric demand. This study discloses a ligand‐facilitated reductive coupling of benzyl chlorides with aryl chlorides, which provides a new and practical synthetic新型杂合Ni(II)配合物,带有高度受阻但具有柔性的IPr *配体,Ni(IPr *)(PPh 3)Br 2(1)和Ni(IPr *)(PCy 3)Br 2(2)(IPr * = 1,3-双(2,6-双(二苯甲基)-4-甲基苯基)咪唑-2-亚基可以轻松制备,产率分别为78%和89%。两者均通过元素分析和NMR光谱进行了表征,并对1进行了X射线晶体学分析。与2及其类似物相比,其空间要求较低的IPr配体(IPr = 1,3-二(2,6-二异丙基苯基)咪唑-2-亚基)复杂1在苄基氯与芳基氯的镁介导的还原偶联中表现出卓越的催化活性,这两种偶联伙伴均具有出色的空间耐受性。这项研究揭示了苄基氯与芳基氯的配体促进还原偶联,为二芳基甲烷的合成提供了一种新的实用合成工具。
-
Cyanobenzeneacetonitriles申请人:E. I. Du Pont de Nemours and Company公开号:US03962307A1公开(公告)日:1976-06-08Certain substituted cyanobenzeneacetonitriles, and their halobenzyl halide intemediates are effective preemergence and postemergence herbicides, which control undesired vegetation without injury to crops.
-
Ready Approach to Organophosphines from ArCl via Selective Cleavage of C–P Bonds by Sodium作者:Jingjing Ye、Jian-Qiu Zhang、Yuta Saga、Shun-ya Onozawa、Shu Kobayashi、Kazuhiko Sato、Norihisa Fukaya、Li-Biao HanDOI:10.1021/acs.organomet.0c00295日期:2020.7.27mechanism of sodium phosphide R2PNa and other alkali metal phosphides R2PM (M = Li and K) have been studied. R2PNa could be prepared, accurately and selectively, via the reactions of SD (sodium finely dispersed in mineral oil) with phosphinites R2POR′ and chlorophosphines R2PCl. R2PNa could also be prepared from triarylphosphines and diarylphosphines via the selective cleavage of C–P bonds. Na was superior研究了磷化钠R 2 PNa和其他碱金属磷化物R 2 PM(M = Li和K)的制备,应用和反应机理。R 2 PNa可以通过SD(精细分散在矿物油中的钠)与次膦酸酯R 2 POR'和氯膦R 2 PC1的反应准确而选择性地制备。R 2 PNa也可以通过选择性裂解C–P键由三芳基膦和二芳基膦制备。在这些反应中,Na优于Li和K。R 2 PNa与多种ArCl反应以有效产生R 2PAr。ArCl优于ArBr和ArI,因为它们只会降低产品收率。另外,Ph 2 PNa优于Ph 2 PLi和Ph 2 PK,因为Ph 2 PLi不产生与PhCl的偶联产物,而Ph 2 PK仅产生低产率的产物。ArCl苯环上的吸电子基团极大地促进了与R 2 PNa的反应,而烷基降低了反应活性。氯乙烯和烷基氯RC1也有效地反应。当t-BuCl没有产生相应的产物,金刚烷基卤化物可以高产率得到相应的膦。通过这种方法,由相应的氯化物制备了多种膦。还可以方便地从Ph
-
1,2,3-Triazolylidene ruthenium(<scp>ii</scp>)-cyclometalated complexes and olefin selective hydrogenation catalysis作者:Bidraha Bagh、Adam M. McKinty、Alan J. Lough、Douglas W. StephanDOI:10.1039/c4dt03156c日期:——RuHCl(PPh3)3 gave RuHCl(PPh3)2(PhCH2C2N2(NMe)Ph) (4a1) as the minor product and the cyclometalated complex RuCl(PPh3)2(PhCH2C2N2(NMe)C6H4) (4a2) as the major product. However, similar reaction with 3b selectively formed the cyclometalated complex RuCl(PPh3)2((C6H2iPr3)CH2C2N2(NMe)C6H4) (4b2). Similarly the silver(I) triazolylidenes 3a and 3b were reacted with RuHCl(H2)(PCy3)2; gave RuHCl(PCy3)2(PhCH2C2N2(NMe)Ph)银(I)1,2,3-三唑-5-亚烷基[[RCH 2 C 2 N 2(NMe)Ph)2 Ag] [AgCl 2 ](R = Ph 3a,C 6 H 2 iPr 3 3b,C 6 H 2 Me 3 3c)和[(PhCH 2 C 2 N 2(NMe)R)2 Ag] [AgCl 2 ](R = C 6 H 4 Me 3d,C 6 H 4 CF 3 3e)合成,然后分别用RuHCl(PPh 3)3和RuHCl(H 2)(PCy 3)2处理。3a与RuHCl(PPh 3)3反应生成次要产物RuHCl(PPh 3)2(PhCH 2 C 2 N 2(NMe)Ph)(4a 1)和环化金属络合物RuCl(PPh 3)2(PhCH 2 C 2 N 2(NMe)C 6 H 4)(4a 2)作为主要产品。但是,与3b的类似反应选择性地形成了环金属化的复合物RuCl(PPh 3)2((C 6 H 2 iPr 3)CH
表征谱图
-
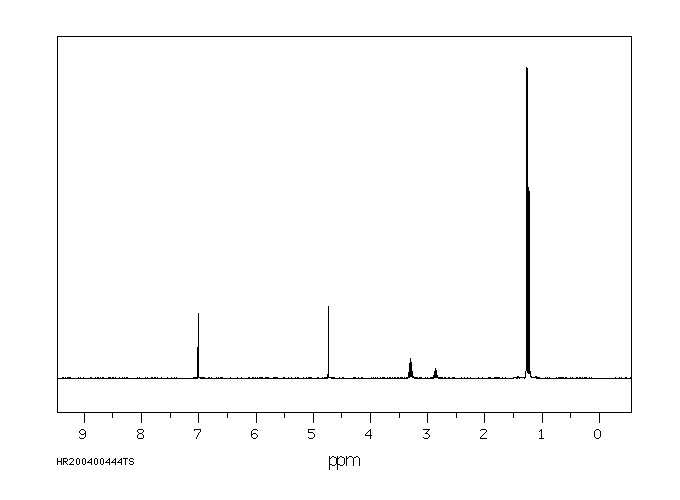
氢谱1HNMR
-
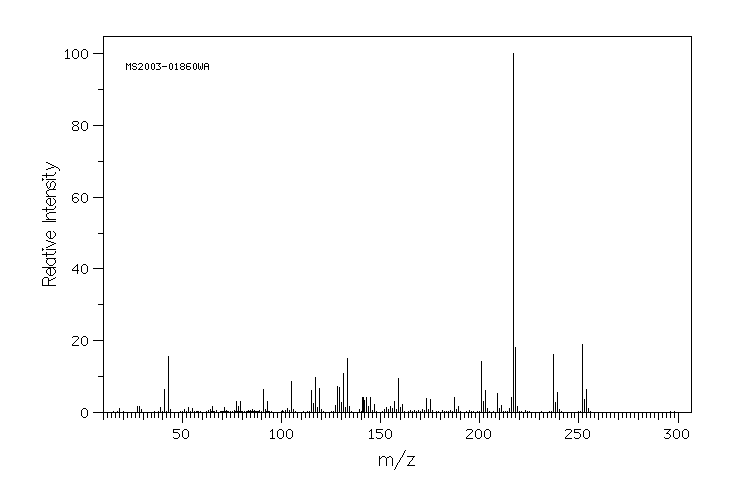
质谱MS
-
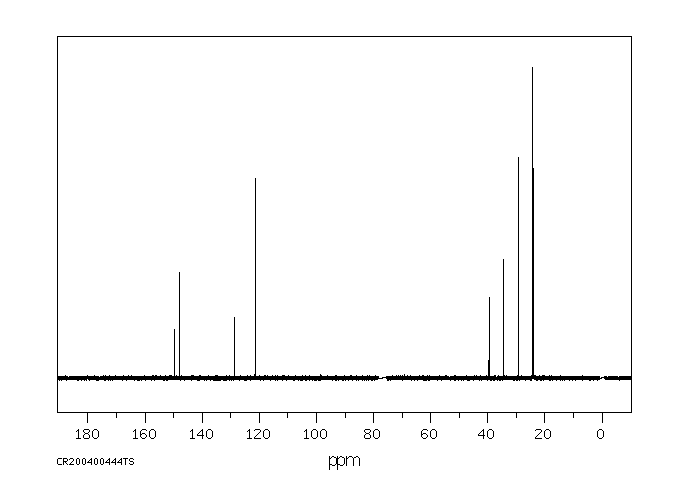
碳谱13CNMR
-
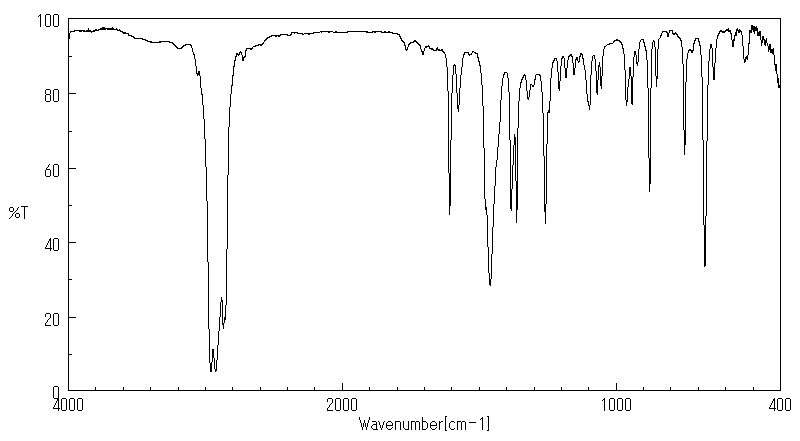
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(5β,6α,8α,10α,13α)-6-羟基-15-氧代黄-9(11),16-二烯-18-油酸
(3S,3aR,8aR)-3,8a-二羟基-5-异丙基-3,8-二甲基-2,3,3a,4,5,8a-六氢-1H-天青-6-酮
(2Z)-2-(羟甲基)丁-2-烯酸乙酯
(2S,4aR,6aR,7R,9S,10aS,10bR)-甲基9-(苯甲酰氧基)-2-(呋喃-3-基)-十二烷基-6a,10b-二甲基-4,10-dioxo-1H-苯并[f]异亚甲基-7-羧酸盐
(1aR,4E,7aS,8R,10aS,10bS)-8-[((二甲基氨基)甲基]-2,3,6,7,7a,8,10a,10b-八氢-1a,5-二甲基-氧杂壬酸[9,10]环癸[1,2-b]呋喃-9(1aH)-酮
(+)顺式,反式-脱落酸-d6
龙舌兰皂苷乙酯
龙脑香醇酮
龙脑烯醛
龙脑7-O-[Β-D-呋喃芹菜糖基-(1→6)]-Β-D-吡喃葡萄糖苷
龙牙楤木皂甙VII
龙吉甙元
齿孔醇
齐墩果醛
齐墩果酸苄酯
齐墩果酸甲酯
齐墩果酸溴乙酯
齐墩果酸二甲胺基乙酯
齐墩果酸乙酯
齐墩果酸3-O-alpha-L-吡喃鼠李糖基(1-3)-beta-D-吡喃木糖基(1-3)-alpha-L-吡喃鼠李糖基(1-2)-alpha-L-阿拉伯糖吡喃糖苷
齐墩果酸 beta-D-葡萄糖酯
齐墩果酸 beta-D-吡喃葡萄糖基酯
齐墩果酸 3-乙酸酯
齐墩果酸 3-O-beta-D-葡吡喃糖基 (1→2)-alpha-L-吡喃阿拉伯糖苷
齐墩果酸
齐墩果-12-烯-3b,6b-二醇
齐墩果-12-烯-3,24-二醇
齐墩果-12-烯-3,21,23-三醇,(3b,4b,21a)-(9CI)
齐墩果-12-烯-3,21,23-三醇,(3b,4b,21a)-(9CI)
齐墩果-12-烯-3,11-二酮
齐墩果-12-烯-2α,3β,28-三醇
齐墩果-12-烯-29-酸,3,22-二羟基-11-羰基-,g-内酯,(3b,20b,22b)-
齐墩果-12-烯-28-酸,3-[(6-脱氧-4-O-b-D-吡喃木糖基-a-L-吡喃鼠李糖基)氧代]-,(3b)-(9CI)
齐墩果-12-烯-28-酸,3,7-二羰基-(9CI)
齐墩果-12-烯-28-酸,3,21,29-三羟基-,g-内酯,(3b,20b,21b)-(9CI)
鼠特灵
鼠尾草酸醌
鼠尾草酸
鼠尾草酚酮
鼠尾草苦内脂
黑蚁素
黑蔓醇酯B
黑蔓醇酯A
黑蔓酮酯D
黑海常春藤皂苷A1
黑檀醇
黑果茜草萜 B
黑五味子酸
黏黴酮
黏帚霉酸