6-羟基-2-甲基喹啉 | 613-21-8
中文名称
6-羟基-2-甲基喹啉
中文别名
2-甲基-6-羟基喹啉
英文名称
6-hydroxyquinaldine
英文别名
2-methylquinolin-6-ol;6-hydroxy-2-methylquinoline;2-methyl-6-hydroxyquinoline;6-hydroxy-2-quinaldine;2-methylquinoline-6-ol
CAS
613-21-8
化学式
C10H9NO
mdl
MFCD00047600
分子量
159.188
InChiKey
USSQQASIZNTRAJ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:214.0 to 218.0 °C
-
沸点:284.68°C (rough estimate)
-
密度:1.1665
-
溶解度:溶于甲醇
-
稳定性/保质期:
性质与稳定性:在常温常压下,该物质不会分解。
计算性质
-
辛醇/水分配系数(LogP):2.2
-
重原子数:12
-
可旋转键数:0
-
环数:2.0
-
sp3杂化的碳原子比例:0.1
-
拓扑面积:33.1
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险品标志:Xi
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2933499090
-
储存条件:贮存: 将密封的容器储存在阴凉、干燥的地方。
SDS
| Name: | 2-Methylquinolin-6-ol 97% Material Safety Data Sheet |
| Synonym: | 6-Hydroxyquinaldin |
| CAS: | 613-21-8 |
Synonym:6-Hydroxyquinaldin
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 613-21-8 | 2-Methylquinolin-6-ol | 97% | unlisted |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 613-21-8: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: tan
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 198 - 200 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C10H9NO
Molecular Weight: 159
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 613-21-8 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-Methylquinolin-6-ol - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 37/39 Wear suitable gloves and eye/face
protection.
WGK (Water Danger/Protection)
CAS# 613-21-8: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 613-21-8 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 613-21-8 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 6-甲氧基-2-甲基喹啉 6-methoxyquinaldine 1078-28-0 C11H11NO 173.214 6-溴-2-甲基喹啉 6-Bromo-2-methyl-quinoline 877-42-9 C10H8BrN 222.084 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 6-甲氧基-2-甲基喹啉 6-methoxyquinaldine 1078-28-0 C11H11NO 173.214 2-甲基-6-乙氧基喹啉 6-ethoxy-2-methyl-quinoline 6628-28-0 C12H13NO 187.241 —— 6-isopropoxy-2-methylquinoline —— C13H15NO 201.268 6-甲氧基-喹啉-2-甲醛 6-methoxyquinoline-2-carbaldehyde 89060-22-0 C11H9NO2 187.198 —— 6-acetoxy-quinaldine 874949-64-1 C12H11NO2 201.225 —— 6-(benzyloxy)-2-methylquinoline —— C17H15NO 249.312 —— tert-butyl (2-methylquinolin-6-yl)carbonate 511231-54-2 C15H17NO3 259.305 —— 6-(benzyloxy)quinoline-2-carbaldehyde 1427753-53-4 C17H13NO2 263.296 —— 2-methyl-6-((triisopropylsilyl)oxy)quinolone 226915-63-5 C19H29NOSi 315.531 5-溴-2-甲基-6-喹啉醇 5-bromo-2-methyl-6-quinolinol 475682-41-8 C10H8BrNO 238.084 —— (R)-2-methyl-6-(pyrrolidin-3-yloxy)quinoline 927691-37-0 C14H16N2O 228.294 —— 6-((tert-butyldimethylsilyl)oxy)quinoline-2-carbaldehyde 1393449-76-7 C16H21NO2Si 287.434 —— 2-methyl-6-phenylquinoline 91875-31-9 C16H13N 219.286 —— methyl-phenyl-carbamic acid 2-methyl-quinolin-6-yl ester 548768-16-7 C18H16N2O2 292.337 - 1
- 2
反应信息
-
作为反应物:参考文献:名称:Synthesis of New Cholesterol- and Sugar-Anchored Squaraine Dyes: Further Evidence of How Electronic Factors Influence Dye Formation摘要:Synthesis of new quinaldine-based squaraine dyes linked to cellular recognition elements that exhibit near-infrared absorption (> 740 nm) are described. Both product analysis and theoretical calculations substantiate the interesting electronic effects of various substituents in the dye formation reaction. These results are useful in the synthesis of symmetrical and unsymmetrical squaraine dyes that can have potential biological and photodynamic therapeutical applications.DOI:10.1021/ol052639j
-
作为产物:描述:1,2,3,4-四氢喹哪啶 在 glucose dehydrogenase 、 葡萄糖 、 cytochrome P450 enzyme CYP102A1 A82M/F87A/H171L/E267F/Q307H/N319Y mutant 、 cytochrome P450 enzyme CYP102A1 R47L/Y51F/H171L/I263G/Q307H/N319Y/A328L mutant 、 C21H25N7O17P3(3-)*2H(1+)*Na(1+) 作用下, 以 aq. phosphate buffer 、 乙醇 为溶剂, 反应 64.0h, 生成 6-羟基-2-甲基喹啉参考文献:名称:CYP102A1(P450BM3)在喹啉和四氢喹啉的氧化中的多功能氧化酶活性。摘要:四氢喹啉,喹啉和二氢喹啉酮是药物分子中常见的核心基序。筛选细胞色素P450酶CYP102A1(P450BM3)的48个变体文库,然后基于初始命中的突变-选择性相关性进行定向诱变,使取代的四氢喹啉,喹啉和3,4-二氢-2-羟基化在两个环的大部分位置上,喹啉酮类以合成相关的比例(1.5 g L -1 天-1)。还观察到其他氧化酶活性,例如C-C键去饱和,芳构化和C-C键形成。这些酶变体在关键的活性位点残基S72,A82,F87,I263,E267,A328和A330处具有突变,为合成和药物发现这些构建基分子的氧官能化衍生物提供了直接且可持续的途径。DOI:10.1002/anie.201904157
文献信息
-
[EN] BICYCLIC ARYL SPHINGOSINE 1-PHOSPHATE ANALOGS<br/>[FR] ANALOGUES D’ARYLSPHINGOSINE-1-PHOSPHATE BICYCLIQUES申请人:BIOGEN IDEC INC公开号:WO2011017561A1公开(公告)日:2011-02-10Compounds that have agonist activity at one or more of the SlP receptors are provided. The compounds are sphingosine analogs that, after phosphorylation, can behave as agonists at SlP receptors.
-
Compounds and uses thereof for decreasing activity of hormone-sensitive lipase申请人:——公开号:US20030166644A1公开(公告)日:2003-09-04Use of compounds to inhibit hormone-sensitive lipase, pharmaceutical compositions comprising the compounds, methods of treatment employing these compounds and compositions, and novel compounds. The present compounds are inhibitors of hormone-sensitive lipase and may be useful in the treatment and/or prevention of medical disorders where a decreased activity of hormone-sensitive lipase is desirable.
-
Assembly of Diversely Substituted Quinolines via Aerobic Oxidative Aromatization from Simple Alcohols and Anilines作者:Jixing Li、Jinlong Zhang、Huameng Yang、Gaoxi JiangDOI:10.1021/acs.joc.6b03064日期:2017.3.17acid catalytic system has been established, providing a direct approach for the preparation of diverse substituted quinoline derivatives in high yields with wide functional group tolerance. Practically, the protocol can be easily scaled up to gram-scale and was utilized in the concise formal synthesis of a promising herbicide candidate.
-
Nickel-Catalyzed Alkoxy-Alkyl Interconversion with Alkylborane Reagents through C−O Bond Activation of Aryl and Enol Ethers作者:Lin Guo、Xiangqian Liu、Christoph Baumann、Magnus RuepingDOI:10.1002/anie.201607646日期:2016.12.5A nickel‐catalyzed alkylation of polycyclic aromatic methyl ethers as well as methyl enol ethers with B‐alkyl 9‐BBN and trialkylborane reagents that involves the cleavage of stable C(sp2)−OMe bonds is described. The transformation has a wide substrate scope and good chemoselectivity profile while proceeding under mild reaction conditions; it provides a versatile way to form C(sp2)−C(sp3) bonds that
-
AROMATIC SULFONE COMPOUND AS ALDOSTERONE RECEPTOR MODULATOR申请人:Dainippon Sumitomo Pharma Co., Ltd.公开号:EP1844768A1公开(公告)日:2007-10-17The present invention provides a compound represented by the following formula (I): [wherein, A represents a group of the following formula (A-1): etc., R1 and R2 each independently represent a hydrogen atom etc., Z represents CR3 etc., W represents CR4 etc., Q represents CR5 etc., R3, R4 and R5 each independently represent a hydrogen atom etc., Y represents an oxygen atom or sulfur atom, X represents an oxygen atom etc. and B represents an optionally substituted aryl group or optionally substituted heteroaryl group], the prodrug thereof or the pharmaceutically acceptable salt thereof for preventing or treating various diseases such as hypertesion, cerebral stroke, cardiac failure, etc.本发明提供了以下式(I)所表示的化合物: [其中,A表示以下式(A-1)的基团: 等等,R1和R2分别独立表示氢原子等,Z表示CR3等,W表示CR4等,Q表示CR5等,R3、R4和R5分别独立表示氢原子等,Y表示氧原子或硫原子,X表示氧原子等,B表示可选择地取代的芳基或可选择地取代的杂环基],其前药或其药学上可接受的盐,用于预防或治疗高血压、脑卒中、心力衰竭等各种疾病。
表征谱图
-
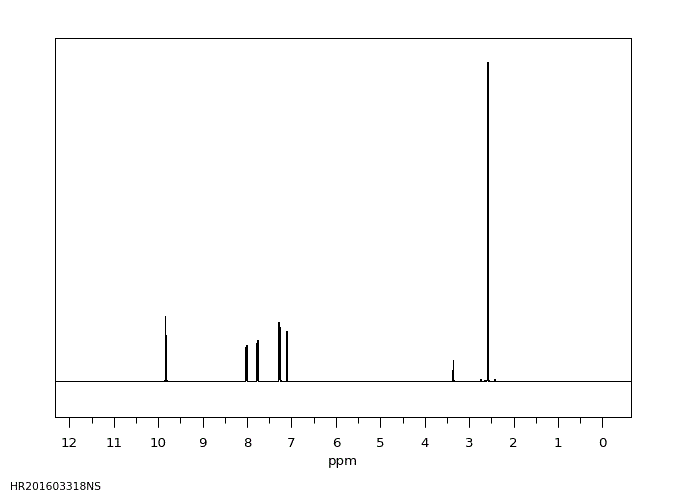
氢谱1HNMR
-
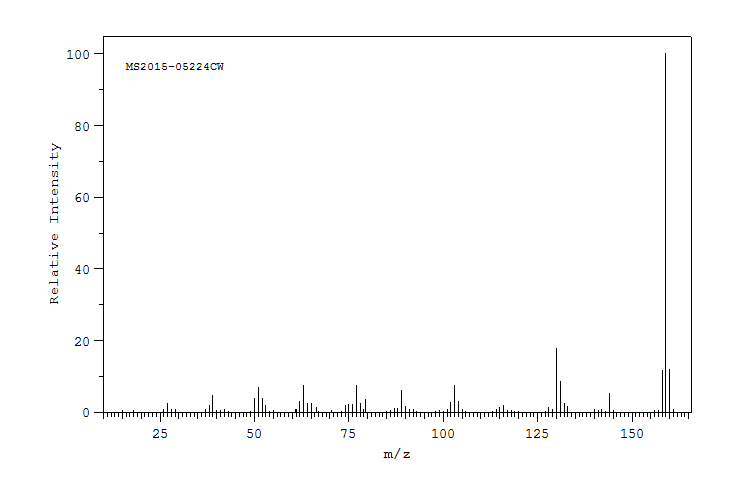
质谱MS
-
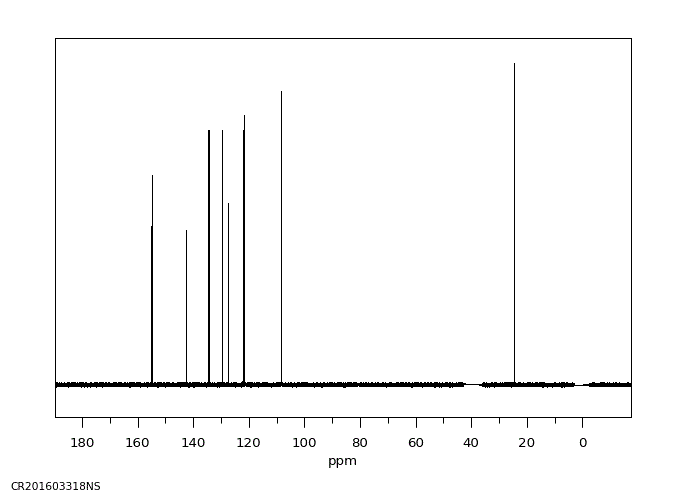
碳谱13CNMR
-
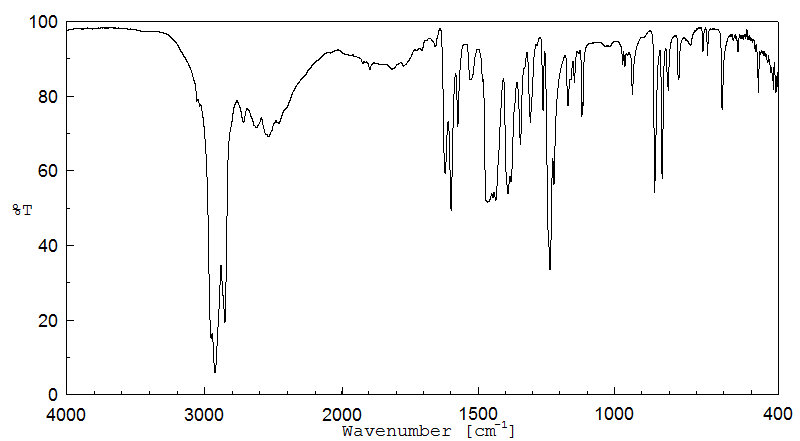
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-4-(叔丁基)-2-(喹啉-2-基)-4,5-二氢噁唑
(SP-4-1)-二氯双(喹啉)-钯
(E)-2-氰基-3-[5-(2,5-二氯苯基)呋喃-2-基]-N-喹啉-8-基丙-2-烯酰胺
(8α,9S)-(+)-9-氨基-七氢呋喃-6''-醇,值90%
(6,7-二甲氧基-4-(3,4,5-三甲氧基苯基)喹啉)
(1-羟基-5-硝基-8-氧代-8,8-dihydroquinolinium)
黄尿酸 8-甲基醚
麻保沙星EP杂质D
麻保沙星EP杂质B
麻保沙星EP杂质A
麦角腈甲磺酸盐
麦角腈
麦角灵
麦皮星酮
麦特氧特
高铁试剂
高氯酸3-苯基[1,3]噻唑并[3,2-f]5-氮杂菲-4-正离子
马波沙星EP杂质F
马波沙星
马来酸茚达特罗杂质
马来酸茚达特罗
马来酸维吖啶
马来酸来那替尼
马来酸四甲基铵
香草木宁碱
颜料红R-122
颜料红210
颜料红
顺式-苯并(f)喹啉-7,8-二醇-9,10-环氧化物
顺式-(alphaR)-N-(4-氯苯基)-4-(6-氟-4-喹啉基)-alpha-甲基环己烷乙酰胺
非那沙星
非那沙星
青花椒碱
青色素863
雷西莫特
隐花青
阿莫地喹-d10
阿莫地喹
阿莫吡喹N-氧化物
阿美帕利
阿米诺喹
阿立哌唑溴代杂质
阿立哌唑杂质B
阿立哌唑杂质38
阿立哌唑杂质1750
阿立哌唑杂质13
阿立哌唑杂质
阿立哌唑杂质
阿尔马尔
阿加曲班杂质43