4-苄氧基苯甲酸 | 29334-16-5
中文名称
4-苄氧基苯甲酸
中文别名
(4-溴苯基)(苯基)甲醇;4-溴二苯甲醇
英文名称
phenyl(4-bromophenyl)methanol
英文别名
(4-bromophenyl)(phenyl)methanol;4-bromobenzhydrol;(p-bromophenyl)phenylmethanol;4-bromo-α-phenylbenzenemethanol;p-bromobenzhydrol;(4-bromophenyl)-phenylmethanol
CAS
29334-16-5
化学式
C13H11BrO
mdl
——
分子量
263.134
InChiKey
WTIWDBNPPSHSCB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:61 °C
-
沸点:381.5±27.0 °C(Predicted)
-
密度:1.435±0.06 g/cm3(Predicted)
-
溶解度:溶于甲醇
计算性质
-
辛醇/水分配系数(LogP):3.4
-
重原子数:15
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.08
-
拓扑面积:20.2
-
氢给体数:1
-
氢受体数:1
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S26,S36
-
危险类别码:R37/38
-
危险品运输编号:UN 3077
-
海关编码:2906299090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:存储条件:室温、干燥且密封。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: (4-Bromophenyl)(phenyl)methanol
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: (4-Bromophenyl)(phenyl)methanol
CAS number: 29334-16-5
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C13H11BrO
Molecular weight: 263.1
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: (4-Bromophenyl)(phenyl)methanol
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: (4-Bromophenyl)(phenyl)methanol
CAS number: 29334-16-5
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C13H11BrO
Molecular weight: 263.1
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-溴苯甲酰苯 (4-bromophenyl)(phenyl)methanone 90-90-4 C13H9BrO 261.118 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— (R)-(4-bromophenyl)(phenyl)methanol 2955-37-5 C13H11BrO 263.134 —— (S)-4-bromophenyl(phenyl)methanol 73773-07-6 C13H11BrO 263.134 —— Bis-<4-brom-α-phenyl-benzyl>-aether 103051-25-8 C26H20Br2O 508.252 二苯甲醇 1,1-Diphenylmethanol 91-01-0 C13H12O 184.238 —— N,N-didesmethylbromazine 50583-08-9 C15H16BrNO 306.202 4-溴二苯基甲烷 1-benzyl-4-bromo-benzene 2116-36-1 C13H11Br 247.134 溴苯海明 2-[(4-bromophenyl)phenylmethoxy]-N,N-dimethylethaneamine 118-23-0 C17H20BrNO 334.256 —— p-benzylbenzohydrol 82639-40-5 C20H18O 274.362 4-溴苯甲酰苯 (4-bromophenyl)(phenyl)methanone 90-90-4 C13H9BrO 261.118 对溴苯甲酰溴 1-bromo-4-(bromo(phenyl)methyl)benzene 18066-89-2 C13H10Br2 326.03 1-(alpha-氯苄基)-4-溴苯 4-bromo-benzhydryl chloride 13391-38-3 C13H10BrCl 281.579 4-苯基-二苯基甲醇 4-phenylbenzhydrol 7598-80-3 C19H16O 260.335 —— 1-bromo-4-(1-phenylethyl)benzene 56913-50-9 C14H13Br 261.161 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:苯海拉明的两种新的甲基和吗啉衍生物对大鼠的抗炎作用摘要:苯海拉明是乙醇胺类的第一代组胺H 1受体拮抗剂之一,具有许多药理特性,包括抗炎作用。在这项研究中,溴(II)和I的两个新的甲苯基衍生物(Di [对甲苯基] [二甲基氨基乙氧基]甲烷(III)和(Di [对甲苯基] [2-吗啉代乙氧基]甲烷(IV))合成。用福尔马林和组胺诱导的大鼠爪水肿评估了它们的急性和慢性抗炎活性。还测量了福尔马林和组胺引起的足爪水肿,二甲苯引起的耳部水肿以及在腹膜腔涂乙酸后的腹膜炎中的血管通透性,并与II进行了比较。选择棉丸诱导的肉芽肿模型来诱导大鼠慢性炎症。新合成的苯海拉明类似物似乎可以有效减少急性炎症。结论是,新药的显着抗炎作用可能与其降低血管通透性机制或对H 1组胺受体的拮抗作用有关。DOI:10.1007/s00044-011-9891-y
-
作为产物:描述:参考文献:名称:Montagne, Recueil des Travaux Chimiques des Pays-Bas, 1908, vol. 27, p. 353摘要:DOI:
文献信息
-
Waste-Free Swift Synthesis of Symmetrical and Unsymmetrical Diarylmethyl Thioethers from Diaryl Carbinols作者:Rama Peddinti、Pallavi SinghDOI:10.1055/s-0036-1589022日期:2017.8to synthesize symmetrical and unsymmetrical diarylmethyl thioethers from diaryl carbinols and thiols in good to quantitative yields is reported. The thiol scope included alkyl and aryl thiols bearing electron-donating and electron-withdrawing groups. Short reaction time, high atom economy, inexpensive activator, free from workup and aryl halides, and gram-scale synthesis are the significant features
-
The Ritter Reaction under Truly Catalytic Brønsted Acid Conditions作者:Roberto Sanz、Alberto Martínez、Verónica Guilarte、Julia M. Álvarez-Gutiérrez、Félix RodríguezDOI:10.1002/ejoc.200700562日期:2007.10(DNBSA) catalyze the Ritter reaction of secondary benzylic alcohols giving rise to the corresponding N-benzylacetamides in usually high yields. Reactions can be conducted without exclusion of oxygen and without the need of dry solvents. With tertiary α,α-dimethylbenzylic alcohols a different pathway involving a formal dimerization reaction takes place under the acid-catalytic conditions used. (© Wiley-VCH
-
Chemoselective transfer hydrogenation of nitroarenes, aldehydes and ketones with propan-2-ol catalysed by Ni-stabilized zirconia作者:T. T. Upadhya、S. P. Katdare、D. P. Sabde、Veda Ramaswamy、A. SudalaiDOI:10.1039/a701518f日期:——Nickel-stabilized zirconia (Zr 0.8 Ni 0.2 O 2 ), a reusable, solid catalyst, efficiently catalyses the chemoselective reduction of nitroarenes, aldehydes and ketones using propan-2-ol and KOH, in the liquid phase, exhibiting the reactivity order NO 2 >> CO > C–X >>CC.
-
Base-free oxidation of alcohols enabled by nickel(<scp>ii</scp>)-catalyzed transfer dehydrogenation作者:Danfeng Ye、Zhiyuan Liu、Jonathan L. Sessler、Chuanhu LeiDOI:10.1039/d0cc03966g日期:——An efficient nickel(II)-catalyzed transfer dehydrogenation oxidation of alcohols is reported that relies on cyclohexanone as the formal oxidant and does not require the use of an external base. The synthetic utility of this protocol is demonstrated via the facile oxidation of structurally complicated natural products.
-
Effects of Phosphorus Substituents on Reactions of α-Alkoxyphosphonium Salts with Nucleophiles作者:Akihiro Goto、Kazuki Otake、Ozora Kubo、Yoshinari Sawama、Tomohiro Maegawa、Hiromichi FujiokaDOI:10.1002/chem.201200480日期:2012.9.3The effects of phosphorus substituents on the reactivity of α‐alkoxyphosphonium salts with nucleophiles has been explored. Reactions of α‐alkoxyphosphonium salts, prepared from various acetals and tris(o‐tolyl)phosphine, with a variety of nucleophiles proceeded efficiently. These processes represent the first examples of high‐yielding nucleophilic substitution reactions of α‐alkoxyphosphonium salts
表征谱图
-
氢谱1HNMR
-
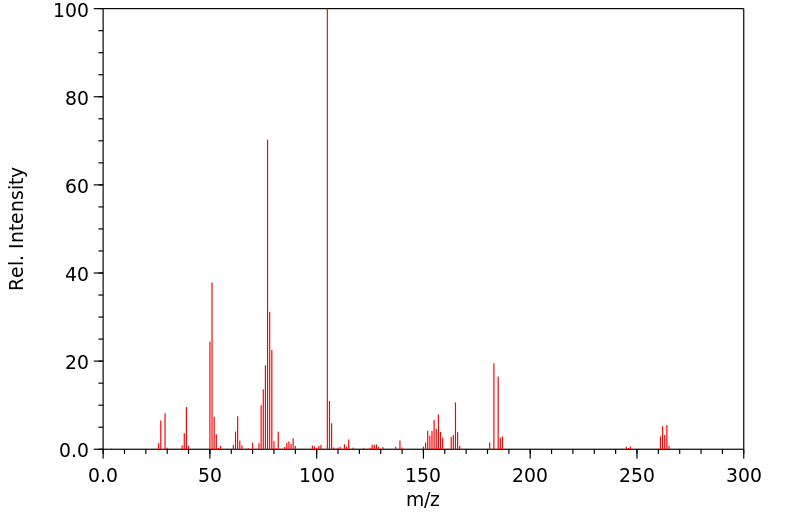
质谱MS
-
碳谱13CNMR
-
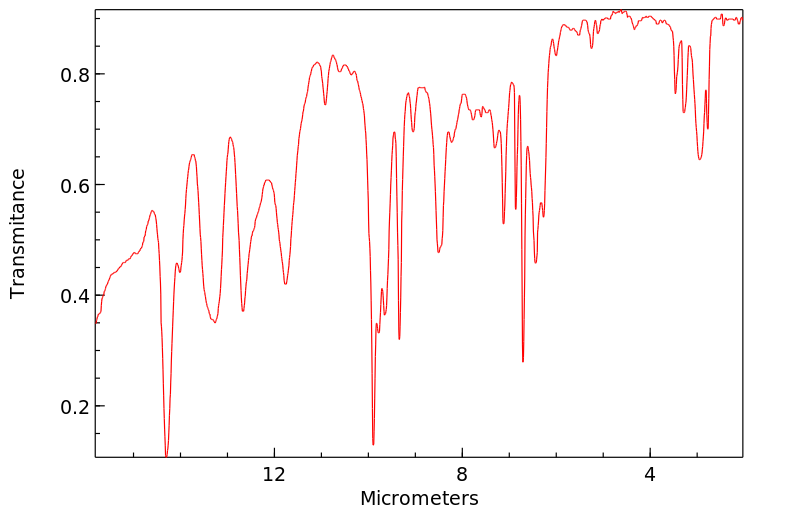
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫