Alpha-苄基苯基肼 | 614-31-3
中文名称
Alpha-苄基苯基肼
中文别名
Α-苄基苯基肼;1-苄基-1-苯基肼;α-苄基苯基肼
英文名称
N-benzyl-N-phenylhydrazine
英文别名
N-Benzyl-N-phenylhydrazin;1-benzyl-1-phenylhydrazine
CAS
614-31-3
化学式
C13H14N2
mdl
MFCD00059186
分子量
198.268
InChiKey
SQMOOVFBFVTTGF-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:164-166℃
-
沸点:218 °C / 38mmHg
-
密度:1,1 g/cm3
-
闪点:186℃
-
稳定性/保质期:
性质与稳定性:在常温常压下,该物质不会分解。
计算性质
-
辛醇/水分配系数(LogP):3.5
-
重原子数:15
-
可旋转键数:3
-
环数:2.0
-
sp3杂化的碳原子比例:0.076
-
拓扑面积:29.3
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险等级:IRRITANT
-
安全说明:S26,S36/37/39
-
危险类别码:R20/21/22
-
海关编码:2928000090
-
储存条件:贮存: 将密封的药瓶放入密封的主要容器中,并将其放置在阴凉、干燥的地方。
SDS
1-Benzyl-1-phenylhydrazine Revision number: 1
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 1-Benzyl-1-phenylhydrazine
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Category 2
Skin corrosion/irritation
Serious eye damage/eye irritation Category 2A
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
Pictograms or hazard symbols
Signal word Warning
Hazard statement Causes skin irritation
Causes serious eye irritation
Precautionary statements
[Prevention] Wash hands thoroughly after handling.
Wear protective gloves/eye protection/face protection.
[Response] IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 1-Benzyl-1-phenylhydrazine
Percent: >98.0%(T)
CAS Number: 614-31-3
Chemical Formula: C13H14N2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
1-Benzyl-1-phenylhydrazine
Section 4. FIRST AID MEASURES
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Dry chemical, foam, water spray, carbon dioxide.
Suitable extinguishing
media:
Specific hazards: Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapor or mist. Wash hands and face thoroughly after handling.
Use a ventilation, local exhaust if vapor or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Law is followed.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Liquid
Physical state (20°C):
Form: clear
Color: Pale yellow - Reddish yellow
Odor: No data available
pH: No data available
Melting point/freezing point:No data available
1-Benzyl-1-phenylhydrazine
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Boiling Point/Range: 218 °C/5.1kPa
Flash Point: No data available
Explosive limits
No data available
Lower:
Upper: No data available
1.10
Density:
Solubility: No data available
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
Reactivity: No special reactivity has been reported.
Conditions to avoid: Light-sensitive
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide, Nitrogen oxides (NOx)
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. Observe all federal, state and local
regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
1-Benzyl-1-phenylhydrazine
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 1-Benzyl-1-phenylhydrazine
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Category 2
Skin corrosion/irritation
Serious eye damage/eye irritation Category 2A
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
Pictograms or hazard symbols
Signal word Warning
Hazard statement Causes skin irritation
Causes serious eye irritation
Precautionary statements
[Prevention] Wash hands thoroughly after handling.
Wear protective gloves/eye protection/face protection.
[Response] IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 1-Benzyl-1-phenylhydrazine
Percent: >98.0%(T)
CAS Number: 614-31-3
Chemical Formula: C13H14N2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
1-Benzyl-1-phenylhydrazine
Section 4. FIRST AID MEASURES
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Dry chemical, foam, water spray, carbon dioxide.
Suitable extinguishing
media:
Specific hazards: Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapor or mist. Wash hands and face thoroughly after handling.
Use a ventilation, local exhaust if vapor or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Law is followed.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Liquid
Physical state (20°C):
Form: clear
Color: Pale yellow - Reddish yellow
Odor: No data available
pH: No data available
Melting point/freezing point:No data available
1-Benzyl-1-phenylhydrazine
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Boiling Point/Range: 218 °C/5.1kPa
Flash Point: No data available
Explosive limits
No data available
Lower:
Upper: No data available
1.10
Density:
Solubility: No data available
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
Reactivity: No special reactivity has been reported.
Conditions to avoid: Light-sensitive
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide, Nitrogen oxides (NOx)
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. Observe all federal, state and local
regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
1-Benzyl-1-phenylhydrazine
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 亚硝基苄基苯胺 N-benzyl-N-phenylnitrous amide 612-98-6 C13H12N2O 212.251 —— N-benzoyl-N-phenylhydrazine 579-45-3 C13H12N2O 212.251 苯甲基苯胺 N-Benzylaniline 103-32-2 C13H13N 183.253 —— N1-phenyl-N1-benzyl-N2-(tert-butoxycarbonyl)hydrazine 126953-22-8 C18H22N2O2 298.385 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 1-benzyl-1,2-diphenylhydrazine 40395-27-5 C19H18N2 274.365 —— 1,4-Diphenyl-1,4-dibenzyl-2-tetrazen 40756-81-8 C26H24N4 392.503 苯甲基苯胺 N-Benzylaniline 103-32-2 C13H13N 183.253 —— N'-benzyl-N'-phenylacetohydrazide —— C15H16N2O 240.305 —— 1-benzyl-2-benzylidene-1-phenylhydrazine 2725-46-4 C20H18N2 286.376 —— 4-methyl-benzaldehyde-(benzyl-phenyl-hydrazone) 80165-57-7 C21H20N2 300.403 —— cinnamaldehyde-(benzyl-phenyl-hydrazone) 84687-99-0 C22H20N2 312.414 —— cyanoacetic N-benzyl-N-phenylhydrazide 136136-45-3 C16H15N3O 265.315 —— N-benzoyl-N'-benzyl-N'-phenylhydrozine 38663-34-2 C20H18N2O 302.376 —— 4-methoxy-benzaldehyde-(benzyl-phenyl-hydrazone) 80165-58-8 C21H20N2O 316.403 —— D-threo-2-deoxy-pentose-(benzyl-phenyl-hydrazone) 16134-32-0 C18H22N2O3 314.384 —— 4-diethylamino-benzaldehyde-(benzyl-phenyl-hydrazone) 73276-71-8 C24H27N3 357.498 —— 2-(4-nitrobenzylidene)-1-benzyl-1-phenylhydrazine 62084-77-9 C20H17N3O2 331.374 —— erythrose-(benzyl-phenyl-hydrazone) —— C17H20N2O3 300.357 —— N-benzyl-N-[(E)-thiophen-2-ylmethylideneamino]aniline —— C18H16N2S 292.404 —— furfural-(benzyl-phenyl-hydrazone) 75179-99-6 C18H16N2O 276.338 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:三氯化 钛(iii)的水溶液将肼还原为胺†摘要:肼中N–N键的裂解被广泛用于胺的制备中,因此在有机合成中占有重要地位。在本文中,我们报告了一种新的方法,该方法可通过市售廉价的水溶液还原肼中的N–N键三氯化钛。反应可在从酸性到中性和碱性的宽pH范围内顺利进行,从而以良好的收率得到胺。此方法与包含官能团的底物兼容,例如,CC双键,苄基-氮键,苄氧基和酰基。DOI:10.1039/c1ob05328k
-
作为产物:描述:参考文献:名称:An efficient synthesis of 1,1-disubstituted hydrazines.摘要:通过Hofmann重排反应,从相应的1,1-二取代脲制备了1,1-二取代肼。除易被氧化脲外,产率相当好,因此本方法为1,1-二取代肼的合成提供了一种额外且高效的途径。DOI:10.1248/cpb.31.423
文献信息
-
Formation of Enehydrazine Intermediates through Coupling of Phenylhydrazines with Vinyl Halides: Entry into the Fischer Indole Synthesis作者:Fuxu Zhan、Guangxin LiangDOI:10.1002/anie.201207173日期:2013.1.21Cut to the chase: Direct formation of an enehydrazine, an intermediate in the classic Fischer indole synthesis, solves the regioselectivity problem associated with indolization. This approach not only achieves selective synthesis of indoles through proper selection of the vinyl halide, but also leads to quick construction of desoxyeseroline and esermethole, as well as the key structural motif in the
-
Reductive Hydrazination with Trichlorosilane: A Method for the Preparation of 1,1-Disubstituted Hydrazines作者:Tao Wang、Xiao Di、Chao Wang、Li Zhou、Jian SunDOI:10.1021/acs.orglett.6b00675日期:2016.4.15and facile method has been developed to prepare 1,1-disubstituted hydrazines via Lewis base promoted direct reductive hydrazination. Under the catalysis of hexamethylphosphoramide (HMPA) and N,N-dimethylacetamide (DMAc), respectively, various ketones and aldehydes could react with phenylhdrazines to prepare 1,1-disubstituted hydrazines with good to high yields.
-
Synthesis and characterization of new ferrocenyl bishydrazones作者:Nardjes Mouas Toma、Hocine Merazig、Jean-Claude Daran、Éric ManouryDOI:10.1016/j.crci.2015.06.002日期:2015.8Résumé New ferrocenyl bishydrazones (2a–2d) have been efficiently obtained from 1,1′-ferrocenedicarboxaldehyde by a straightforward synthesis. The four new compounds have been fully characterized by NMR (1H, 13C), high-resolution mass spectroscopy, and the molecular structure of compounds (2a–2d) has been elucidated by X-ray diffraction on single crystals.
-
Synthesis and Antimicrobial Studies of Coumarin-Substituted Pyrazole Derivatives as Potent Anti-Staphylococcus aureus Agents作者:Rawan Alnufaie、Hansa Raj KC、Nickolas Alsup、Jedidiah Whitt、Steven Andrew Chambers、David Gilmore、Mohammad A. AlamDOI:10.3390/molecules25122758日期:——In this paper, synthesis and antimicrobial studies of 31 novel coumarin-substituted pyrazole derivatives are reported. Some of these compounds have shown potent activity against methicillin-resistant Staphylococcus aureus (MRSA) with minimum inhibitory concentration (MIC) as low as 3.125 µg/mL. These molecules are equally potent at inhibiting the development of MRSA biofilm and the destruction of preformed
-
Mechanistic Dichotomy with Alkynes in the Formal Hydrohydrazination/Fischer Indolization Tandem Reaction Catalyzed by a Ph3PAuNTf2/pTSA Binary System作者:Nitin T. Patil、Ashok KonalaDOI:10.1002/ejoc.201001114日期:2010.12method involving a formal hydrohydrazination/ Fischer indolization tandem reaction to synthesize 2,3-disubstituted indoles from alkynes and arylhydrazines has been developed. The approach uses a Ph 3 PAuNTf 2 /pTSA·H 2O binary catalytic system in which a very low catalyst loading of Ph 3 PAuNTf 2 (2 mol-%) is required. The reaction time is very short and, most importantly, the reaction is not sensitive
表征谱图
-
氢谱1HNMR
-
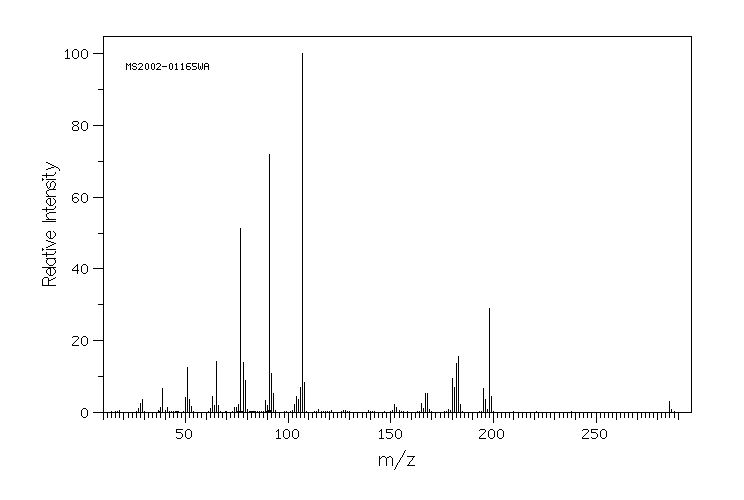
质谱MS
-
碳谱13CNMR
-
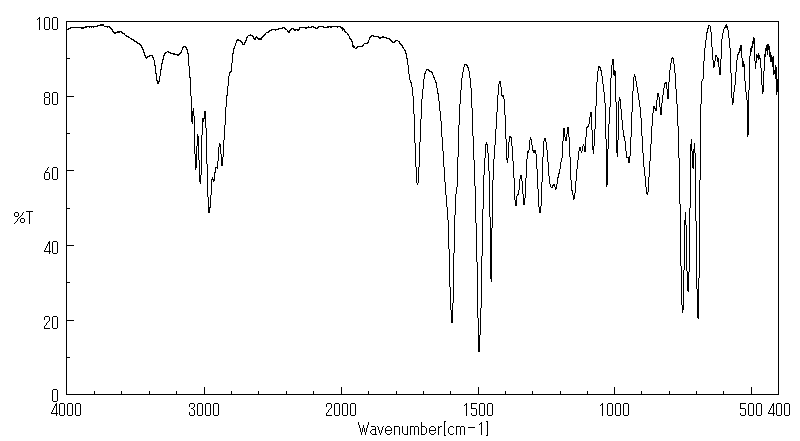
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫