2,4,6-三氟苯甲酰氯 | 79538-29-7
中文名称
2,4,6-三氟苯甲酰氯
中文别名
二氢-1,4,5,8-四羟基蒽醌
英文名称
2,4,6-trifluorobenzoyl chloride
英文别名
——
CAS
79538-29-7
化学式
C7H2ClF3O
mdl
——
分子量
194.54
InChiKey
SIFIJQFBERMWMU-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:167-168°C
-
密度:1.484
-
闪点:167-168°C
-
稳定性/保质期:
远离氧化物、酸和水分。
计算性质
-
辛醇/水分配系数(LogP):2.7
-
重原子数:12
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:4
安全信息
-
危险等级:8
-
危险品标志:C
-
安全说明:S26,S36/37/39,S45
-
危险类别码:R34
-
海关编码:2916399090
-
包装等级:II
-
危险类别:8
-
危险品运输编号:3265
-
危险性防范说明:P260,P264,P280,P301+P330+P331,P303+P361+P353,P304+P340,P305+P351+P338,P310,P321,P363,P405,P501
-
危险性描述:H314
-
储存条件:应将存放在充满干燥惰性气体的容器中,并储存在阴凉、干燥的地方。储存地点需加锁,钥匙须由技术人员及其助手保管。切勿与酸性物质一同存放,并要远离水源。
SDS
| Name: | 2 4 6-Trifluorobenzoyl chloride Material Safety Data Sheet |
| Synonym: | None |
| CAS: | 79538-29-7 |
Synonym:None
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 79538-29-7 | 2,4,6-Trifluorobenzoyl chloride | 98 | unlisted |
Risk Phrases: 34
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Causes burns.
Potential Health Effects
Eye:
Causes eye burns. Lachrymator (substance which increases the flow of tears).
Skin:
Causes skin burns.
Ingestion:
Causes gastrointestinal tract burns.
Inhalation:
Causes chemical burns to the respiratory tract.
Chronic:
Chronic exposure may cause effects similar to those of acute exposure.
Section 4 - FIRST AID MEASURES
Eyes: In case of contact, immediately flush eyes with plenty of water for at least 15 minutes. Get medical aid immediately.
Skin:
In case of contact, immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Get medical aid immediately. Wash clothing before reuse.
Ingestion:
If swallowed, do NOT induce vomiting. Get medical aid immediately.
If victim is fully conscious, give a cupful of water. Never give anything by mouth to an unconscious person.
Inhalation:
If inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Use of water will produce irritating and toxic vapors of hydrogen chloride.
Hydrochloric acid solutions react with most metals, forming flammable hydrogen gas.
Extinguishing Media:
Use carbon dioxide or dry chemical. Do NOT get water inside containers.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Avoid runoff into storm sewers and ditches which lead to waterways. Clean up spills immediately, observing precautions in the Protective Equipment section. Provide ventilation. Do not get water inside containers.
Section 7 - HANDLING and STORAGE
Handling:
Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Keep container tightly closed. Do not ingest or inhale. Use only in a chemical fume hood. Discard contaminated shoes.
Keep from contact with moist air and steam.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Corrosives area. Store protected from moisture.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 79538-29-7: Personal Protective Equipment Eyes: Wear chemical splash goggles.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: colorless
Odor: pungent odor
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water: Hydrolysis.
Specific Gravity/Density:
Molecular Formula: C7H2ClF3O
Molecular Weight: 194.54
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Exposure to moist air or water.
Incompatibilities with Other Materials:
Strong bases, strong oxidizing agents.
Hazardous Decomposition Products:
Hydrogen chloride, carbon monoxide, carbon dioxide, hydrogen fluoride gas.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 79538-29-7 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2,4,6-Trifluorobenzoyl chloride - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S.
Hazard Class: 8
UN Number: 3265
Packing Group: III
IMO
Shipping Name: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S.
Hazard Class: 8
UN Number: 3265
Packing Group: III
RID/ADR
Shipping Name: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S.
Hazard Class: 8
UN Number: 3265
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: C
Risk Phrases:
R 34 Causes burns.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 79538-29-7: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 79538-29-7 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 79538-29-7 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
2,4,6-三氟苯甲酰氯可作为有机合成中间体和医药中间体使用,主要应用于实验室研发及化工生产过程中。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,4,6-三氟苯甲酸 2,4,6-trifluorobenzoic acid 28314-80-9 C7H3F3O2 176.095 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2,4,6-三氟苯甲酰胺 2,4,6-trifluorobenzamide 82019-50-9 C7H4F3NO 175.11
反应信息
-
作为反应物:描述:2,4,6-三氟苯甲酰氯 在 4-二甲氨基吡啶 、 bis[dimethyl(μ-dimethylsulfide)platinum(II)] 、 三乙胺 作用下, 以 氘代四氢呋喃 、 二氯甲烷 为溶剂, 反应 72.25h, 生成 2-(2,4-Difluoro-6-methoxyphenyl)-4,5-dihydro-1,3-oxazole参考文献:名称:Pt催化CF活化范围的探索摘要:我们最近报道了使用 [Pt2Me4(SMe2)2] 作为预催化剂对多氟芳基亚胺进行催化甲基化和甲氧基化的方法。这些方法为部分功能化的芳基氟化物提供了一条新途径,其具有作为合成药物和材料的构建模块的潜在效用。我们在此报告为扩大这些流程的范围所做的努力,特别是对指导小组的选择。描述了与先前报道的 PtII 催化甲基化和甲氧基化范围的比较。DOI:10.1002/ejoc.201100478
-
作为产物:描述:参考文献:名称:几种卤代非硝基苯并噻嗪酮的合成、结构表征和抗分枝杆菌评价摘要:8-Nitro-1,3-benzothiazin-4-ones (BTZs),以 BTZ043 和 PBTZ169 为最先进的化合物,代表了一类新的强效抗结核药物,可不可逆地抑制癸二烯基磷酰-β- d-核糖-2'-差向异构酶 (DprE1),一种对病原体结核分枝杆菌细胞壁合成至关重要的酶。金分枝杆菌DSM 43999 和结核分枝杆菌H 37的合成、结构表征和体外测试报道了缺少硝基的卤代 2-(4-ethoxycarbonylpiperrazin-1-yl)-1,3-benzothiazin-4-ones 的 Rv。X 射线晶体学显示 BTZ 支架的结构可以显着偏离平面性。与最近的报道相反,本研究的结果表明,进一步研究卤代非硝基 BTZ 的抗结核活性并不是一种有前途的方法。DOI:10.1007/s00044-021-02735-4
-
作为试剂:描述:2-氯-6-羟基甲基吡啶-3-醇 在 四(三苯基膦)钯 咪唑 、 titanium(IV) isopropylate 、 盐酸 、 4-二甲氨基吡啶 、 lithium hydroxide 、 copper(l) iodide 、 迭氮酸 、 草酰氯 、 (S)-(-)-Carreira ligand 、 2,4,6-三氟苯甲酰氯 、 3,5-二叔丁基水杨酸 、 四丁基氟化铵 、 水 、 双氧水 、 双(三甲基硅烷基)氨基钾 、 二甲基亚砜 、 盐酸-N-乙基-Nˊ-(3-二甲氨基丙基)碳二亚胺 、 三乙胺 、 N,N-二异丙基乙胺 、 三苯基膦 、 硝苯酚 、 偶氮二甲酸二乙酯 作用下, 以 四氢呋喃 、 甲醇 、 二氯甲烷 、 N,N-二甲基甲酰胺 、 甲苯 、 乙腈 为溶剂, 反应 20.5h, 生成 [(Z)-(2R,7R,11R,16S)-14-Bromo-19-chloro-16-((S)-1-hydroxy-prop-2-ynyl)-9,9-dimethyl-4-oxo-11-prop-2-ynyl-5,8,10,17-tetraoxa-20-aza-tricyclo[16.2.2.07,11]docosa-1(21),14,18(22),19-tetraen-12-yn-2-yl]-carbamic acid tert-butyl ester参考文献:名称:Enantioselective Synthesis of Kedarcidin Chromophore Aglycon in Differentially Protected Form摘要:DOI:10.1002/1521-3773(20020315)41:6<1062::aid-anie1062>3.0.co;2-8
文献信息
-
<i>N</i>-Ammonium Ylide Mediators for Electrochemical C–H Oxidation作者:Masato Saito、Yu Kawamata、Michael Meanwell、Rafael Navratil、Debora Chiodi、Ethan Carlson、Pengfei Hu、Longrui Chen、Sagar Udyavara、Cian Kingston、Mayank Tanwar、Sameer Tyagi、Bruce P. McKillican、Moses G. Gichinga、Michael A. Schmidt、Martin D. Eastgate、Massimiliano Lamberto、Chi He、Tianhua Tang、Christian A. Malapit、Matthew S. Sigman、Shelley D. Minteer、Matthew Neurock、Phil S. BaranDOI:10.1021/jacs.1c03780日期:2021.5.26taking a first-principles approach guided by computation, these new mediators were identified and rapidly expanded into a library using ubiquitous building blocks and trivial synthesis techniques. The ylide-based approach to C–H oxidation exhibits tunable selectivity that is often exclusive to this class of oxidants and can be applied to real-world problems in the agricultural and pharmaceutical sectors强 C(sp 3 )-H 键的位点特异性氧化在有机合成中具有无可争议的效用。从简化对代谢物的获取和先导化合物的后期多样化到截断逆合成计划,学术界和工业界都越来越需要新的试剂和方法来实现这种转变。当前化学试剂的一个主要缺点是在结构和反应性方面缺乏多样性,这阻碍了用于快速筛选的组合方法的使用。在这方面,定向进化仍然最有希望在各种复杂环境中实现复杂的 C-H 氧化。在此,我们提出了一个设计合理的平台,该平台使用N-铵叶立德作为电化学驱动的氧化剂,用于位点特异性、化学选择性 C(sp 3 )-H 氧化。通过采用以计算为指导的第一性原理方法,这些新的介质被识别出来,并使用无处不在的构建块和简单的合成技术迅速扩展到一个库中。基于叶立德的 C-H 氧化方法表现出可调的选择性,这通常是此类氧化剂独有的,可应用于农业和制药领域的实际问题。
-
Synthesis and Antiproliferative Activities of <scp>OSW</scp> ‐1 Analogues Bearing 2”‐ <scp> <i>O</i> ‐ <i>p</i> ‐Acylaminobenzoyl </scp> Residues <sup>†</sup>作者:Lijun Sun、Di Zhu、Laura Olde Groote Beverborg、Ruina Wang、Yongjun Dang、Mingming Ma、Wei Li、Biao YuDOI:10.1002/cjoc.202000110日期:2020.10OSW‐1 is a well‐known natural saponin with potent antitumor activities. We have designed and prepared a small library of 22 OSW‐1 analogues with a variety of p‐acylamino‐benzoyl groups installed at C2” of the xylose residue, wherein a regioselective (1→3)‐glycosylation of arabinoside 3,4‐diol has been achieved by manipulation of the protecting groups on the imidate donors. Bioassays lead to new structure‐activity
-
Carbodiimide coupling reagent申请人:Eli Lilly and Company公开号:US05998630A1公开(公告)日:1999-12-07The present invention relates to compounds used for increasing activation of the 5-HT1F receptor.本发明涉及用于增加5-HT1F受体激活的化合物。
-
Development of novel 2,4-bispyridyl thiophene–based compounds as highly potent and selective Dyrk1A inhibitors. Part I: Benzamide and benzylamide derivatives作者:Sarah S. Darwish、Mohammad Abdel-Halim、Mohamed Salah、Ashraf H. Abadi、Walter Becker、Matthias EngelDOI:10.1016/j.ejmech.2018.07.050日期:2018.9effects especially over long treatment periods. Herein, we describe the design and synthesis of a series of amide functionalized 2,4-bispyridyl thiophene compounds, of which the 4-fluorobenzyl amide derivative (31b) displayed the highest potency against Dyrk1A and remarkable selectivity over closely related kinases (IC50: Dyrk1A = 14.3 nM; Dyrk1B = 383 nM, Clk1 > 2 μM). This degree of selectivity over蛋白激酶Dyrk1A调节与阿尔茨海默氏病(AD)的发生或发展相关的多个过程,例如通过tau蛋白,淀粉样前体蛋白(APP)的磷酸化以及与tau pre-mRNA的选择性剪接相关的蛋白。因此,Dyrk1A已被提议作为治疗AD的潜在靶标。但是,对同一个蛋白激酶家族的其他紧密相关的激酶(例如Dyrk1B和Dyrk2)或其他家族的激酶(例如Clk1)的共抑制作用限制了Dyrk1A抑制剂的使用,因为这可能会导致无法预料的副作用,尤其是在长期治疗中时期。在这里,我们描述了一系列酰胺官能化的2,4-双吡啶基噻吩化合物的设计和合成,其中4-氟苄基酰胺衍生物(31b)显示出对Dyrk1A的最高效力,并且比紧密相关的激酶具有显着的选择性(IC 50:Dyrk1A = 14.3 nM; Dyrk1B = 383 nM,Clk1> 2μM)。迄今为止,很少能达到对频繁击中目标的选择性。此外,31b在完整细胞中以高效率抑制了Dyrk1A(IC
-
Inhibitors of Bruton's Tyrosine Kinase申请人:Berthel Steven Joseph公开号:US20120040949A1公开(公告)日:2012-02-16This application discloses 6-(2-Hydroxymethyl-phenyl)-2-methyl-2H-pyridazin-3-one derivatives according to generic Formula I: wherein, variables X, R, and Y 4 , are defined as described herein, which inhibit Btk. The compounds disclosed herein are useful to modulate the activity of Btk and treat diseases associated with excessive Btk activity. The compounds are further useful to treat inflammatory and auto immune diseases associated with aberrant B-cell proliferation, such as rheumatoid arthritis. Also disclosed are compositions containing compounds of Formula I and at least one carrier, diluent or excipient.
表征谱图
-
氢谱1HNMR
-
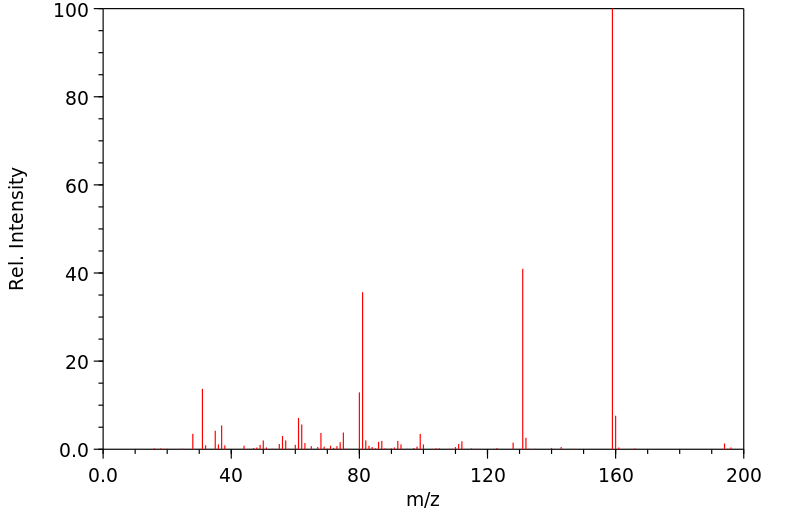
质谱MS
-
碳谱13CNMR
-
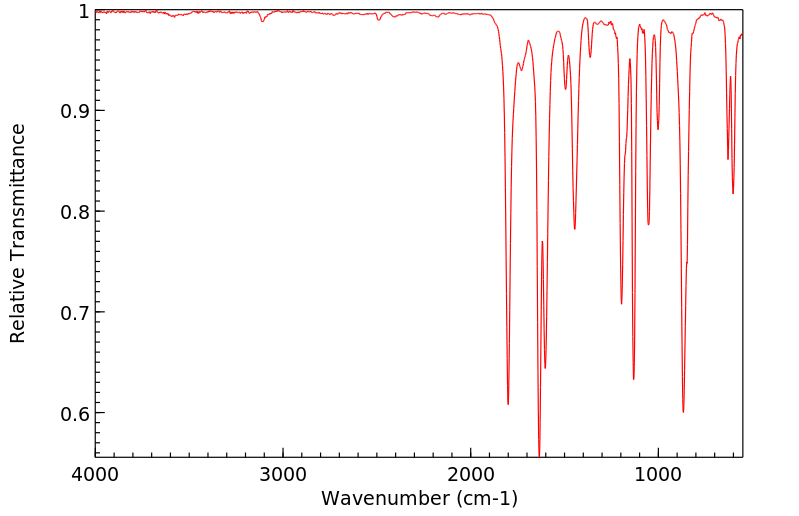
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫