苯戊酮 | 1009-14-9
中文名称
苯戊酮
中文别名
1-苯基-1-戊酮;丁基苯酮;戊苯酮
英文名称
phenyl butyl ketone
英文别名
valerophenone;Valerophenon;1-phenylpentan-1-one
CAS
1009-14-9
化学式
C11H14O
mdl
MFCD00009480
分子量
162.232
InChiKey
XKGLSKVNOSHTAD-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:-9 °C
-
沸点:244-245 °C (lit.)
-
密度:0.975 g/mL at 20 °C (lit.)
-
闪点:217 °F
-
溶解度:可溶于氯仿(少量)、乙酸乙酯(少量)
-
LogP:2.79
-
物理描述:Liquid
-
保留指数:1327
-
稳定性/保质期:
- 在常温常压下保持稳定,应避免与氧化物接触。
- 它存在于烟气中。
计算性质
-
辛醇/水分配系数(LogP):3
-
重原子数:12
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.363
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
TSCA:Yes
-
危险品标志:Xi
-
安全说明:S22,S24/25
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:2914399090
-
危险品运输编号:NONH for all modes of transport
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:请将容器密封保存,并存放在阴凉、干燥处。
SDS
| Name: | Valerophenone 98% Material Safety Data Sheet |
| Synonym: | 1-Phenyl-1-Pentanone |
| CAS: | 1009-14-9 |
Synonym:1-Phenyl-1-Pentanone
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 1009-14-9 | Valerophenone | 98% | 213-767-3 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Runoff from fire control or dilution water may cause pollution.
Extinguishing Media:
In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam. Use agent most appropriate to extinguish fire.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Avoid runoff into storm sewers and ditches which lead to waterways. Clean up spills immediately, observing precautions in the Protective Equipment section. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 1009-14-9: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to minimize contact with skin.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear light yellow
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 105 - 107 deg C @ 5.00mmHg
Freezing/Melting Point: -9 deg C
Autoignition Temperature: Not applicable.
Flash Point: 102 deg C ( 215.60 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: .9880g/cm3
Molecular Formula: C11H14O
Molecular Weight: 162.23
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Acids, bases, oxidizing agents, plastics.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 1009-14-9 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
Valerophenone - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 1009-14-9: No information available.
Canada
CAS# 1009-14-9 is listed on Canada's DSL List.
CAS# 1009-14-9 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 1009-14-9 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 5-bromo-1-phenyl-1-pentanone 4509-91-5 C11H13BrO 241.128 5-氯苯戊酮 5-Chloro-1-phenyl-1-pentanone 942-93-8 C11H13ClO 196.677 1-苯基-1,4-戊二酮 1-phenyl-1,4-pentadione 583-05-1 C11H12O2 176.215 1-苯基-4-戊烯-1-酮 1-Phenylpent-4-en-1-one 3240-29-7 C11H12O 160.216 苯丙酮 1-phenyl-propan-1-one 93-55-0 C9H10O 134.178 —— 3-bromo-1-phenylpentan-1-one —— C11H13BrO 241.128 3-羟基-1-苯基戊-1-酮 3-hydroxy-1-phenylpentan-1-one 88704-64-7 C11H14O2 178.231 4'-溴苯戊酮 4'-bromovalerophenone 7295-44-5 C11H13BrO 241.128 苯基环丙基甲酮 cyclopropyl phenyl ketone 3481-02-5 C10H10O 146.189 2-溴-1-苯基-1-戊酮 α-bromovalerophenone 49851-31-2 C11H13BrO 241.128 —— m-bromovalerophenone —— C11H13BrO 241.128 3-氯代苯丙酮 3-chloropropiophenone 936-59-4 C9H9ClO 168.623 苯乙酮 acetophenone 98-86-2 C8H8O 120.151 戊基苯 pentylbenzene 538-68-1 C11H16 148.248 —— 1-phenyl-heptan-3-one 19969-04-1 C13H18O 190.285 1-苯基-2-丙烯基-1-酮 PVK 768-03-6 C9H8O 132.162 苯甲酰腈 benzoyl cyanide 613-90-1 C8H5NO 131.134 - 1
- 2
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 苯丁酮 butyrophenone 495-40-9 C10H12O 148.205 1-苯基-1,4-戊二酮 1-phenyl-1,4-pentadione 583-05-1 C11H12O2 176.215 —— 4-fluoro-1-phenylpentan-1-one 1383116-00-4 C11H13FO 180.222 1-苯基-4-戊烯-1-酮 1-Phenylpent-4-en-1-one 3240-29-7 C11H12O 160.216 2-甲基-1-苯基戊-1-酮 2-methyl-1-phenylpentan-1-one 31366-05-9 C12H16O 176.258 1-苯基戊烷-1,3-二酮 1-Phenyl-1,3-pentanedione 5331-64-6 C11H12O2 176.215 —— 3-fluoro-1-phenylpentan-1-one —— C11H13FO 180.222 —— 3-bromo-1-phenylpentan-1-one —— C11H13BrO 241.128 3-羟基-1-苯基己烷-1-酮 3-hydroxy-1-phenylhexan-1-one 57548-41-1 C12H16O2 192.258 1-苯基戊烷-1,2-二酮 1-phenyl-1,2-pentanedione 20895-66-3 C11H12O2 176.215 —— (+/-)-(1'R,2'R)-(2'-methylcyclopropyl)phenyl methanone 126525-21-1 C11H12O 160.216 2-溴-1-苯基-1-戊酮 α-bromovalerophenone 49851-31-2 C11H13BrO 241.128 —— 2-fluoro-1-phenyl-1-pentanone 29114-66-7 C11H13FO 180.222 1-苯基-2-丙基-2-丙烯-1-酮 2-methylene-1-phenylpentan-1-one 354529-98-9 C12H14O 174.243 —— 2-chloro-1-phenylpentan-1-one 16130-53-3 C11H13ClO 196.677 2-羟基-1-苯基戊-1-酮 2-hydroxy-1-phenylpentan-1-one 20907-23-7 C11H14O2 178.231 —— (S)-(-)-2-hydroxy-1-phenyl-1-pentan-1-one 97551-18-3 C11H14O2 178.231 (R)-(+)-2-羟基-1-苯基-1-戊-1-酮 (R)-(+)-2-hydroxy-1-phenyl-1-pentan-1-one 97551-17-2 C11H14O2 178.231 —— 1-(3-amino-phenyl)-pentan-1-one 534-88-3 C11H15NO 177.246 1-(2-甲基苯基)-1-戊酮 1-(o-tolyl)pentan-1-one 20359-56-2 C12H16O 176.258 —— 3-Benzoyl-hexanenitrile 127081-21-4 C13H15NO 201.268 1-(2-羟基苯基)戊-1-酮 1-(2-hydroxyphenyl)pentan-1-one 18430-91-6 C11H14O2 178.231 —— 1-(2,6-dimethylphenyl)pentan-1-one —— C13H18O 190.285 1-(2-碘苯基)戊-1-酮 2-iodobutanophenone 32730-83-9 C11H13IO 288.128 苯乙酮 acetophenone 98-86-2 C8H8O 120.151 戊基苯 pentylbenzene 538-68-1 C11H16 148.248 —— 2-(Methylsulfanyl)-1-phenylpentan-1-one 61836-09-7 C12H16OS 208.324 1-(4-硝基苯基)-1-戊酮 1-(4-nitrophenyl)pentan-1-one 22857-30-3 C11H13NO3 207.229 2-(甲基氨基)-1-苯基戊-1-酮 pentedrone 879722-57-3 C12H17NO 191.273 —— 2,2-dichloro-1-phenylpentan-1-one 66255-86-5 C11H12Cl2O 231.122 —— (E)-1-phenyl-1,2-pentanedione 2-oxime 291542-11-5 C11H13NO2 191.23 —— 1-phenyl-pentane-1,2-dione-2-oxime 101708-94-5 C11H13NO2 191.23 —— 1-(2,6-Dideuteriophenyl)pentan-1-one 1529790-18-8 C11H14O 164.216 5-苯基戊酰胺 5-phenylpentanitrile 36603-28-8 C11H15NO 177.246 丁苯 1-butylbenzene 104-51-8 C10H14 134.221 —— α-N.N-Dimethylaminomethylvalerophenon 53207-32-2 C14H21NO 219.327 1-苯基-2-己酮 1-phenyl-2-hexanone 25870-62-6 C12H16O 176.258 - 1
- 2
- 3
- 4
反应信息
-
作为反应物:描述:苯戊酮 在 Jacobsen's catalyst sodium hypochlorite 、 4-苯基吡啶-N-氧化物 、 phosphate buffer 、 lithium diisopropyl amide 作用下, 以 四氢呋喃 、 二氯甲烷 为溶剂, 反应 19.0h, 生成 (S)-(-)-2-hydroxy-1-phenyl-1-pentan-1-one参考文献:名称:(salen)锰(III)络合物将烯醇磷酸酯不对称氧化为α-羟基酮。烯醇磷酸酯的取代方式对氧转移的立体化学的影响摘要:本文介绍了对各种不同取代的环状(E)和非环状(Z)-烯醇磷酸酯的对映选择性催化氧化的研究。无环(的不对称氧化Ž含有磷酸基团中的烷氧基取代基)烯醇磷酸盐2a中,Ç,ë -克,我和Ĵ和Ž -构型中含有磷酸基团的取代基的芳氧基烯醇磷酸盐2B,d,和^ h,得到光学活性α-羟基酮4a – j具有良好或高对映选择性的相反构型。研究了烯醇磷酸酯取代基的电子和空间效应对氧化的立体选择性的影响。DOI:10.1016/j.tet.2006.09.100
-
作为产物:参考文献:名称:Williams; Dickert; Krynitsky, Journal of the American Chemical Society, 1941, vol. 63, p. 2511摘要:DOI:
-
作为试剂:参考文献:名称:5,6-dihydropyrone derivatives as protease inhibitors and antiviral agents摘要:本发明涉及新型5,6-二氢吡喃衍生物及相关结构,这些衍生物具有强大的抑制HIV天冬氨酸蛋白酶,从而阻止HIV感染性的作用。5,6-二氢吡喃衍生物可用于开发治疗细菌和病毒感染和疾病,包括艾滋病的治疗方案。本发明还涉及多功能化5,6-二氢吡喃和相关结构的合成方法。公开号:US05789440A1
文献信息
-
Synthesis of Isoquinoline Derivatives via Palladium‐Catalyzed C−H/C−N Bond Activation of <i>N</i> ‐Acyl Hydrazones with <i>α</i> ‐Substituted Vinyl Azides作者:Biao Nie、Wanqing Wu、Wei Zeng、Qingyun Ren、Ji Zhang、Yingjun Zhang、Huanfeng JiangDOI:10.1002/adsc.201901394日期:2020.3.17A palladium‐catalyzed cyclization of N‐acetyl hydrazones with vinyl azides has been developed. Various substituted isoquinolines, including diverse fused isoquinolines can be prepared via this protocol in moderate to good yields. Mechanistic studies suggest that α‐substituted vinyl azide serves as an internal nitrogen source. Also, C−H bond activation and C−N bond cleavage have been realized using
-
A simple and efficient oxidation of alcohols with ruthenium on carbon作者:Shigeki Mori、Masato Takubo、Kazuya Makida、Takayoshi Yanase、Satoka Aoyagi、Tomohiro Maegawa、Yasunari Monguchi、Hironao SajikiDOI:10.1039/b908451g日期:——A simple, efficient, and environmentally-friendly heterogeneous Ru/C-catalyzed oxidation of secondary and primary alcohols without additives under an atmosphere of oxygen has been established.已经建立了在氧气气氛下没有添加剂的简单,有效和环境友好的Ru / C多相Ru / C催化仲醇和伯醇的氧化方法。
-
Highly Efficient Redox Isomerization of Allylic Alcohols and Transfer Hydrogenation of Ketones and Aldehydes Catalyzed by Ruthenium Complexes作者:Pei Nian Liu、Kun Dong Ju、Chak Po LauDOI:10.1002/adsc.201000667日期:2011.2.11A dicationic dichloro-bipyridine-ruthenium complex shows very high catalytic activity in redox isomerization of allylic alcohols but a relatively low one in transfer hydrogenation of ketones; surprisingly, the analogous dimethyl-bipyridine-ruthenium complex shows reverse catalytic activities in the two reactions.
-
Alcohols for the α-Alkylation of Methyl Ketones and Indirect Aza-Wittig Reaction Promoted by Nickel Nanoparticles作者:Francisco Alonso、Paola Riente、Miguel YusDOI:10.1002/ejoc.200800729日期:2008.10Nickel nanoparticles have been found to activate primary alcohols used for the α-alkylation of ketones or in indirect aza-Wittig reactions. These processes involve hydrogen transfer from the alcohol to the intermediate α,β-unsaturated ketone or imine, respectively. All these reactions are carried out in the absence of any ligand, hydrogen acceptor or base under mild reaction conditions. For the first
-
New method for generation of β-oxido carbenoid via ligand exchange reaction of sulfoxides: A versatile procedure for one-carbon homologation of carbonyl compounds作者:Tsuyoshi Satoh、Norifumi Itoh、Kaoru Gengyo、Sae Takada、Naoyuki Asakawa、Yumi Yamani、Koji YamakawaDOI:10.1016/s0040-4020(01)89299-1日期:1994.1one-carbon homologation of carbonyl compounds is described. The method is based on the rearrangement of β-oxido carbenoid which is generated via the ligand exchange reaction of the sulfinyl group of α-chloro β-hydroxy sulfoxide with tert-butyllithium. Addition of the carbanion of aryl 1-chloroalkyl sulfoxides to carbonyl compounds gave the adducts in good yields. The β-oxido carbenoid rearrangement of the
表征谱图
-
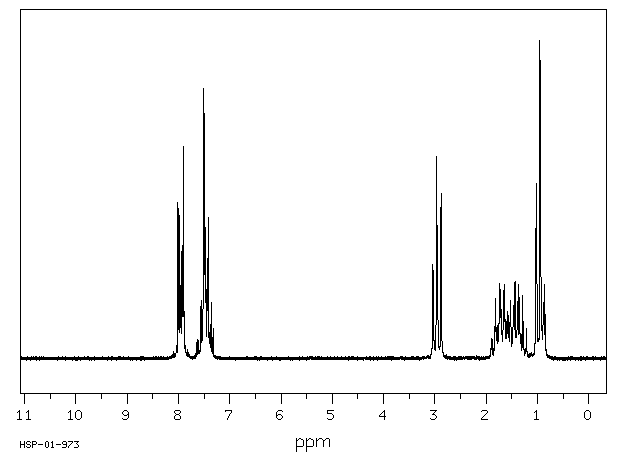
氢谱1HNMR
-
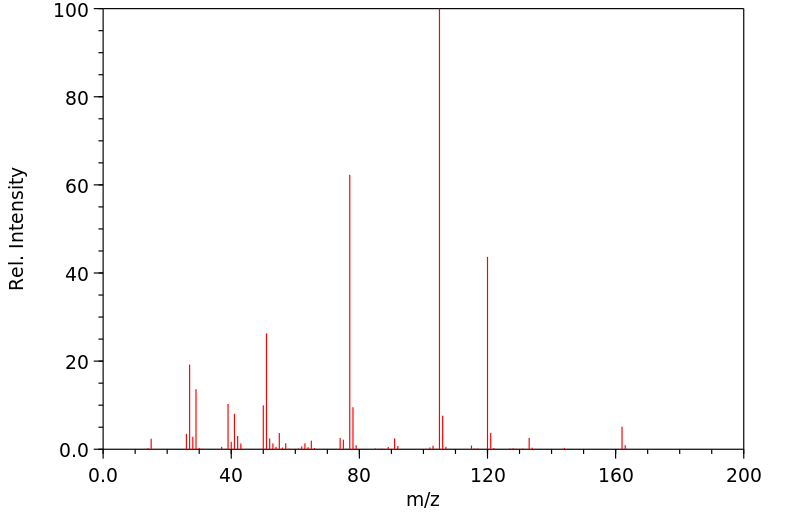
质谱MS
-
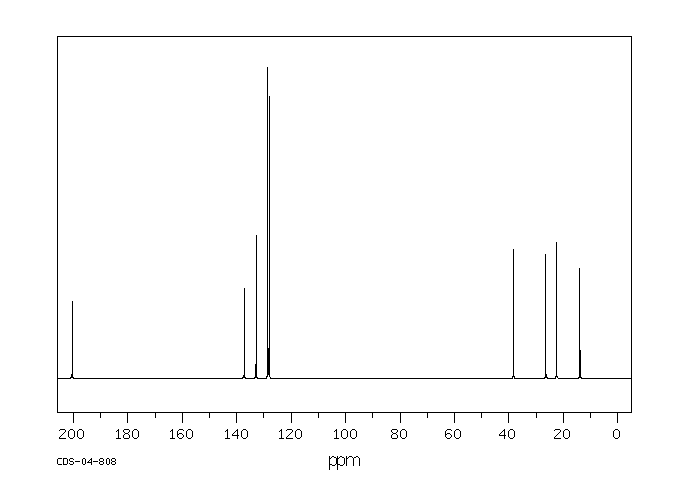
碳谱13CNMR
-
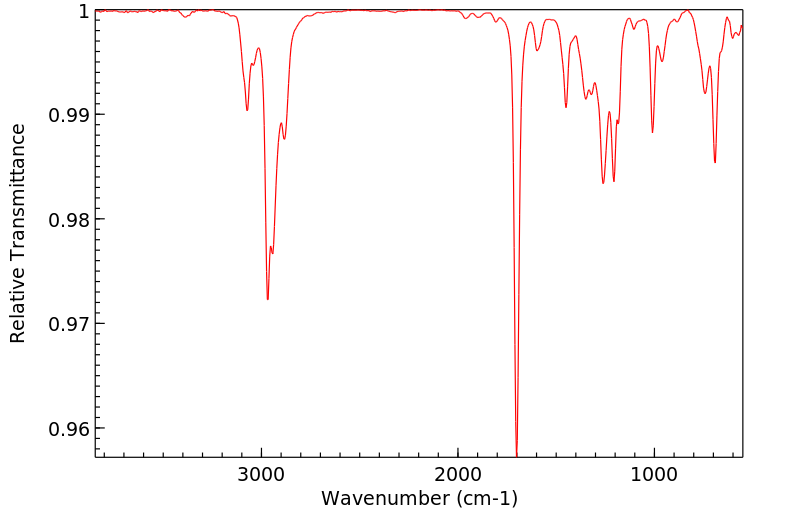
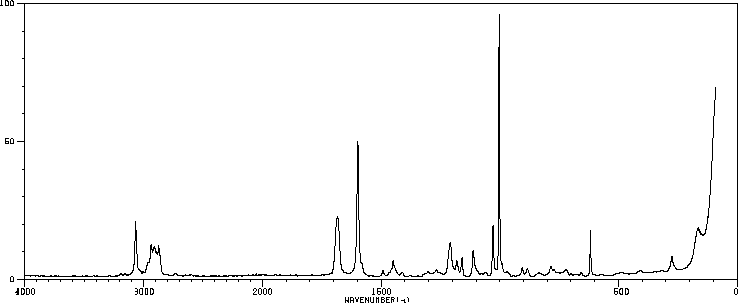
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷