1,3-二氯-5-氯甲基苯 | 3290-06-0
中文名称
1,3-二氯-5-氯甲基苯
中文别名
3,5-二氯苄氯;3,5-二氯氯苄
英文名称
1,3-dichloro-5-chloromethyl-benzene
英文别名
3,5-dichlorobenzyl chloride;1,3-dichloro-5-(chloromethyl)benzene
CAS
3290-06-0
化学式
C7H5Cl3
mdl
——
分子量
195.476
InChiKey
ZFLRKAMKGYNFPH-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:35°C
-
沸点:230.4°C (estimate)
-
密度:1.3950 (estimate)
计算性质
-
辛醇/水分配系数(LogP):3.6
-
重原子数:10
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.14
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
危险等级:LACHRYMATOR, CORROSIVE
-
危险品标志:C
-
危险类别码:R34
-
危险品运输编号:UN 3261
-
海关编码:2903999090
-
安全说明:S26,S36/37/39,S45
-
包装等级:III
-
危险类别:8
-
危险性防范说明:P280,P305+P351+P338,P310
-
危险性描述:H314
-
储存条件:2-8°C
SDS
| Name: | 1 3-Dichloro-5-(chloromethyl)benzene 95+% Material Safety Data Sheet |
| Synonym: | 3,5-Dichlorobenzyl chlorid |
| CAS: | 3290-06-0 |
Synonym:3,5-Dichlorobenzyl chlorid
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 3290-06-0 | 1,3-Dichloro-5-(chloromethyl)benzene | 95+% | unlisted |
Risk Phrases: 34
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Causes burns.
Potential Health Effects
Eye:
Causes eye burns.
Skin:
Causes skin burns.
Ingestion:
Causes gastrointestinal tract burns.
Inhalation:
Causes chemical burns to the respiratory tract.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin:
Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Do not induce vomiting. Get medical aid immediately.
Inhalation:
Get medical aid immediately. Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use foam, dry chemical, or carbon dioxide.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Corrosives area.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 3290-06-0: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: off-white
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 35 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature: Not available.
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C7H5Cl3
Molecular Weight: 195
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Oxidizing agents, bases, amines.
Hazardous Decomposition Products:
Hydrogen chloride, chlorine, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 3290-06-0 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
1,3-Dichloro-5-(chloromethyl)benzene - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S.*
Hazard Class: 8
UN Number: 3261
Packing Group: III
IMO
Shipping Name: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S.
Hazard Class: 8
UN Number: 3261
Packing Group: III
RID/ADR
Shipping Name: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S.
Hazard Class: 8
UN Number: 3261
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: C
Risk Phrases:
R 34 Causes burns.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 3290-06-0: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 3290-06-0 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 3290-06-0 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3,5-二氯苯甲醇 3,5-dichlorobenzyl alcohol 60211-57-6 C7H6Cl2O 177.03 3,5-二氯苯甲醛 3,5-dichlorobenzaldehyde 10203-08-4 C7H4Cl2O 175.014 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— (3,5-dichlorophenyl)methanethiol 65963-17-9 C7H6Cl2S 193.097 (3,5-二氯苄基)-肼 3,5-dichlorobenzylhydrazine 51421-25-1 C7H8Cl2N2 191.06
反应信息
-
作为反应物:描述:1,3-二氯-5-氯甲基苯 在 potassium permanganate 、 sodium hydride 作用下, 以 水 、 N,N-二甲基甲酰胺 、 丙酮 为溶剂, 反应 16.17h, 生成 1-[(3,5-dichlorophenyl)methyl]indole-3-carboxylic acid参考文献:名称:与氯硝胺相关的吲哚衍生物摘要:报道了 1-benzylindole-3- 羧酸的合成和抗肿瘤活性。化合物 16b 在 1 位带有与 Lonidamine (1) 相同的替代物,被证明是最具活性的衍生物。DOI:10.1002/ardp.19843171007
-
作为产物:参考文献:名称:除草剂噁嗪草酮中间体3,5-二氯苄氯的合成方法摘要:本发明公开了一种除草剂噁嗪草酮中间体3,5‑二氯苄氯的合成方法,属于农药制药领域,其特征在于,将3,5‑二氯苯甲酸、复氢化合物、金属氯化物在聚乙二醇作用下反应得到3,5‑二氯苄氯。反应过程为:1)在保护气条件下,在反应釜中加入3,5‑二氯苯甲酸、溶剂A和复氢化合物,升温至40~60℃反应5~12min,加入金属氯化物、聚乙二醇和碱水,开启超声辅助,设定好功率和频率,控制温度为90~110℃,反应压力维持1.5~2个大气压,保持2~4h反应结束;2)将体系冷却后,加入约1~2倍反应液体积的溶剂B,过滤除去不溶物,分层,有机相用干燥剂干燥后,经浓缩、重结晶得到3,5‑二氯苄氯。本发明步骤少,副反应少,产率高,排污少,操作安全。公开号:CN109232164A
文献信息
-
吡咯并吡唑类衍生物、其制备方法及其在医药上的应用
-
[EN] MLKL INHIBITORS<br/>[FR] INHIBITEURS MLKL申请人:NAT INSTITUTE OF BIOLOGICAL SCIENCES BEIJING公开号:WO2018157800A1公开(公告)日:2018-09-07Purine derivatives that inhibit cellular necroptosis and/or human MLKL, pharmaceutical compositions thereof, and methods of treating an MLKL-mediated disorder with an effective amount of the compound or composition. Said MLKL-mediated disorder is pathology associated necroptosis, including ischemia-reperfusion damage, neurodegeneration, and inflammatory diseases such as acute pancreatitis, multiple sclerosis, inflammatory bowel disease, and allergic colitis.
-
Palladium-catalyzed carbonylative cyclization of benzyl chlorides with anthranils for the synthesis of 3-arylquinolin-2(1<i>H</i>)-ones作者:Jian-Li Liu、Ren-Rui Xu、Wei Wang、Xinxin Qi、Xiao-Feng WuDOI:10.1039/d1ob00298h日期:——An efficient carbonylative procedure for the synthesis of 3-arylquinoin-2(1H)-ones has been established. Through a palladium-catalyzed aminocarbonylation of benzyl chlorides with anthranils, a variety of 3-arylquinoin-2(1H)-one products were obtained in moderate to excellent yields with good functional group tolerance.
-
Carbon-halogen bond activation by a structurally constrained phosphorus(III) platform作者:Penglong Wang、Qin Zhu、Yi Wang、Guixiang Zeng、Jun Zhu、Congqing ZhuDOI:10.1016/j.cclet.2020.11.005日期:2021.4The σ-bond activation by main group element has received enormous attention from theoretical and experimental chemists. Here, the reaction of C–X (X = Cl, Br, I) bonds in benzyl and allyl halides with a pincer-type phosphorus(III) species was reported. A series of structurally robust phosphorus(V) compounds were formed via the formal oxidative addition reactions of C–X bonds to the phosphorus(III)
-
Glucagon antagonists/inverse agonists申请人:Noro Nordisk A/S公开号:US06503949B1公开(公告)日:2003-01-07Disclosed is a novel class of compounds of formula (I) wherein V, A, Y, Z, R1, E, X and D are as defined in the specification. These compounds act to antagonize the action of the glucagon hormone on the glucagon receptor. Owing to their antagonizing effect of the glucagon receptor, the compounds are suitable for treating or preventing glucagon-mediated conditions and diseases such as hyperglycemia, Type 1 diabetes, Type 2 diabetes and obesity.揭示了一类新的化合物,其化学式为(I),其中V、A、Y、Z、R1、E、X和D的定义如规范中所述。这些化合物作用于拮抗胰高血糖素激素对胰高血糖素受体的作用。由于这些化合物对胰高血糖素受体的拮抗作用,它们适用于治疗或预防由胰高血糖素介导的疾病和病症,如高血糖、1型糖尿病、2型糖尿病和肥胖症。
表征谱图
-
氢谱1HNMR
-
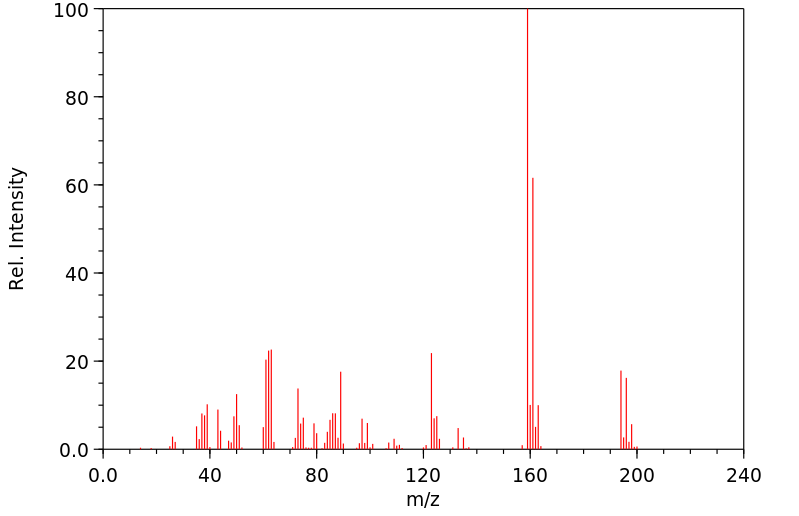
质谱MS
-
碳谱13CNMR
-
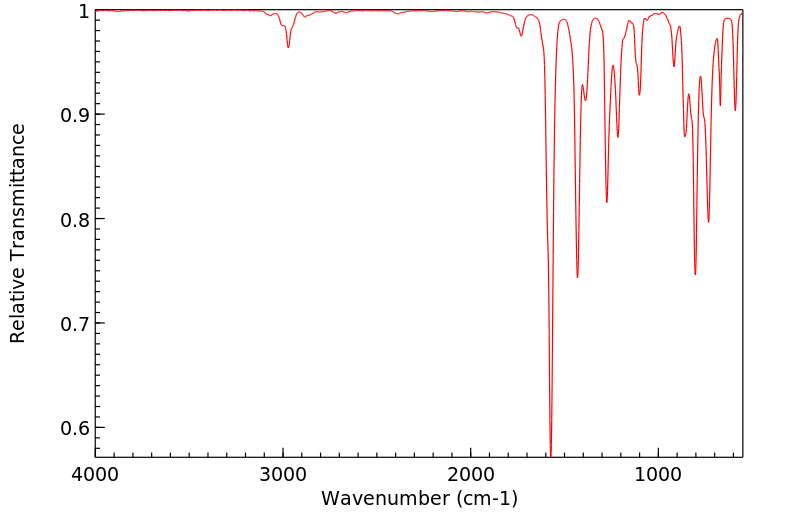
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫