3-(苄氨基)丙腈 | 706-03-6
中文名称
3-(苄氨基)丙腈
中文别名
3-(苯甲基氨基)丙正离子基腈;3-(苯甲基氨基)丙腈;3-(苄氨基)丙腈;3-(苯甲基氨基)丙腈, 97+%
英文名称
3-(benzylamino)propionitrile
英文别名
3-(benzylamino)propanenitrile;3-(N-benzylamino)propionitrile
CAS
706-03-6
化学式
C10H12N2
mdl
MFCD00001955
分子量
160.219
InChiKey
MWTGBAURSCEGSL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:70 °C
-
沸点:276.13°C (rough estimate)
-
密度:1.024 g/mL at 25 °C (lit.)
-
闪点:>230 °F
-
溶解度:溶于二甲基亚砜。
-
稳定性/保质期:
远离氧化物、酸、酸性氯化物及酸酐。
计算性质
-
辛醇/水分配系数(LogP):1
-
重原子数:12
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:35.8
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险等级:6.1
-
危险品标志:Xn
-
安全说明:S23,S26,S37/39
-
危险类别码:R20/21/22
-
WGK Germany:3
-
海关编码:2926909090
-
包装等级:III
-
危险类别:6.1
-
危险品运输编号:UN3276
-
危险性防范说明:P261,P280,P305+P351+P338
-
危险性描述:H302+H312+H332,H315,H319,H335
-
储存条件:存放在密封容器内,并放置在阴凉、干燥处。储存地点须远离氧化剂。避免与酸或酸性物质一同存放,远离含氯酸和酸酐的区域。
SDS
| Name: | 3-(Benzylamino)propionitrile 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 706-03-6 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 706-03-6 | 3-(Benzylamino)propionitrile | 97 | 211-890-7 |
Risk Phrases: 20/21/22
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled. Can produce delayed pulmonary edema.
Chronic:
Prolonged or repeated skin contact may cause dermatitis. Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Do not induce vomiting. If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Clean up spills immediately, observing precautions in the Protective Equipment section. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Use with adequate ventilation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 706-03-6: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear, colorless
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: Not available.
Autoignition Temperature: Not applicable.
Flash Point: > 110 deg C (> 230.00 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.0240 g/cm3
Molecular Formula: C10H12N2
Molecular Weight: 160.22
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, excess heat.
Incompatibilities with Other Materials:
Acid anhydrides, acid chlorides, acids, strong oxidizing agents, carbon dioxide.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 706-03-6 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
3-(Benzylamino)propionitrile - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
IMO
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
RID/ADR
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
Safety Phrases:
S 23 Do not inhale gas/fumes/vapour/spray.
WGK (Water Danger/Protection)
CAS# 706-03-6: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 706-03-6 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 706-03-6 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 1-(苯基甲基)氮丙啶 1-benzylaziridine 1074-42-6 C9H11N 133.193 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 3-苄胺基丙胺 N-benzyl-1,3-diaminopropane 13910-48-0 C10H16N2 164.25 3-(N-苄基-N-甲基氨基)丙腈 3-(N-Benzyl-N-methylamino)propionitril 23873-65-6 C11H14N2 174.246 N-乙基苄胺 N-ethylbenzylamine 14321-27-8 C9H13N 135.209 —— N-ethyl-N-benzyl-β-alanine nitrile —— C12H16N2 188.272 3,3'-(苄基亚氨基)二丙腈 N-benzylbis(2-cyanoethyl)amine 782-87-6 C13H15N3 213.282 3-苄氨基丙酸 N-benzyl-β-alanine 5426-62-0 C10H13NO2 179.219 —— N-(2-cyanoethyl)-N-cyanomethyl-benzylamine 161693-84-1 C12H13N3 199.255 N-苄基乙酰胺 N-(phenylmethyl)acetamide 588-46-5 C9H11NO 149.192 —— N-benzyl-N-(2-cyanoethyl)cyanamide 651718-08-0 C11H11N3 185.228 —— 3-[4-Aminobutyl(benzyl)amino]propanenitrile 125718-87-8 C14H21N3 231.341 —— N-(2-cyanoethyl)-N-(3-cyanopropyl)benzylamine 1216-04-2 C14H17N3 227.309 —— N1-benzyl-N1-ethyl-propane-1,3-diamine 140134-63-0 C12H20N2 192.304 N,N-二甲基苄胺 N,N'-dimethylbenzylamine 103-83-3 C9H13N 135.209 3-[苄基(甲基)氨基]丙酸 3-[benzyl(methyl)amino]propanoic acid 149692-49-9 C11H15NO2 193.246 —— N-(3-aminobutyl)-N-(3-aminopropyl)(phenylmethyl)amine 73038-05-8 C14H25N3 235.373 —— N-benzyl-N-(2-cyanoethyl)acetamide 59629-66-2 C12H14N2O 202.256 - 1
- 2
反应信息
-
作为反应物:参考文献:名称:Efficient, asymmetric synthesis of (−)-isooncinotine摘要:Asymmetric synthesis of (-)-isooncinotine, a 22-membered lactam of spermidine alkaloid, starting from resolution of 2-piperidineethanol with (S)-10-camphorsulfonic acid is reported. Michael addition, amidations, and aluminum hydride reduction were applied to form the moiety of spermidine. Retro-Michael addition was observed when beta-amido- and beta-amino-propionitriles were reduced by LAH. The effects of LAH versus AlH3 were discussed. The synthesis of the skeleton of this macrolide is achieved with ring-closing metathesis of the diene prepared from acylation of the spermidine. (c) 2007 Elsevier Ltd. All rights reserved.DOI:10.1016/j.tet.2007.01.068
-
作为产物:描述:1-苄基-3-吡咯烷酮 在 ammonium hydroxide 、 dipotassium peroxodisulfate 、 copper(II) nitrate 作用下, 以 乙腈 为溶剂, 反应 15.0h, 以10%的产率得到3-(苄氨基)丙腈参考文献:名称:铜氨介导的羰基化合物的氧化摘要:NH 4 OH水溶液中的铜(II),在大多数情况下,与O 2或K 2 S 2 O 8作为辅助氧化剂一起使用时,如果有良好的碳电絮凝剂,可将酮氧化裂解为腈。对于未活化的酮,观察到的主要反应是酮亚胺的氧化偶联反应,生成嗪。DOI:10.1016/s0040-4039(00)74473-x
文献信息
-
Pyrrolidine derivatives申请人:——公开号:US20020049243A1公开(公告)日:2002-04-25The present invention relates to pyrrolidine derivatives and dimeric forms and/or pharmaceutically acceptable esters, and/or salts thereof. The compounds are useful as inhibitors of metalloproteases, e.g. zinc proteases, particularly zinc hydrolases, and which are effective in treating disease states are associated with vasoconstriction of increasing occurrences.
-
Graphite-Supported Gold Nanoparticles as Efficient Catalyst for Aerobic Oxidation of Benzylic Amines to Imines and<i>N</i>-Substituted 1,2,3,4-Tetrahydroisoquinolines to Amides: Synthetic Applications and Mechanistic Study作者:Man-Ho So、Yungen Liu、Chi-Ming Ho、Chi-Ming CheDOI:10.1002/asia.200900261日期:2009.10.5of N‐substituted 1,2,3,4‐tetrahydroisoquinolines in the presence of aqueous NaHCO3 solution gave the corresponding amides in good yields (83–93 %) with high selectivity (up to amide/enamide=93:4) (6 examples). The same protocol can be applied to the synthesis of benzimidazoles from the reaction of o‐phenylenediamines with benzaldehydes under aerobic conditions (8 examples). By simple centrifugation使用氧气作为末端氧化剂对胺进行选择性氧化是绿色化学中的重要领域。在这项工作中,我们描述了使用石墨负载的金纳米颗粒(AuNPs / C)催化环状和非环状苄胺的好氧氧化为相应的亚胺,具有中等至优异的底物转化率(43-100%)和产品收率(66–99%)(19个例子)。在NaHCO 3水溶液存在下,对N-取代的1,2,3,4-四氢异喹啉进行氧化,得到的酰胺产率高(83-93%),选择性高(酰胺/酰胺= 93:4)( 6个示例)。相同的协议可以从反应被施加到苯并咪唑的合成Ò在好氧条件下将苯二胺与苯甲醛一起使用(8个例子)。通过简单的离心,可以回收AuNPs / C并重复使用十次,以将二苄基胺氧化为N-亚苄基(苯基)甲胺,而不会显着降低催化活性和选择性。可以将协议“ AuNPs / C + O 2 ”调整为克级,通过氧化10 g 1,2,3,4-可以得到8.9 g(84%分离出的产率)3,4-二氢异
-
Aza-Michael reactions in water using functionalized ionic liquids as the recyclable catalysts作者:X. Liu、M. Lu、G. Gu、T. LuDOI:10.1007/bf03245908日期:2011.9Some recyclable SO3H-functionalized ionic liquids have been used as catalysts in water for the aza-Michael reactions of amines with α,β-unsaturated compounds to produce β-amino compounds. High yields of the products, short reaction times, mild reaction conditions, simple experimental procedure, reusable catalysts and no obvious loss of the catalytic activity make this protocol a contribution to organic
-
<i>o</i>-Iodoxybenzoic Acid (IBX) as a Viable Reagent in the Manipulation of Nitrogen- and Sulfur-Containing Substrates: Scope, Generality, and Mechanism of IBX-Mediated Amine Oxidations and Dithiane Deprotections作者:K. C. Nicolaou、Casey J. N. Mathison、Tamsyn MontagnonDOI:10.1021/ja0400382日期:2004.4.28o-Iodoxybenzoic acid (IBX), a highly versatile hypervalent iodine(V) reagent, was found to efficiently mediate the dehydrogenation of amines in addition to facilitating the oxidative cleavage of dithioacetals and dithioketals. Through the development of relevant IBX-based protocols, a plethora of useful synthetic intermediates, including imines, oximes, ketones, and aromatic N-heterocycles, were found
-
[EN] 3-PHOSPHOGLYCERATE DEHYDROGENASE INHIBITORS AND USES THEREOF<br/>[FR] INHIBITEURS DE LA 3-PHOSPHOGLYCÉRATE DÉSHYDROGÉNASE ET LEURS UTILISATIONS申请人:RAZE THERAPEUTICS INC公开号:WO2017156177A1公开(公告)日:2017-09-14The present invention provides compounds, compositions thereof, and methods of using the same.本发明提供了化合物、其组合物以及使用这些化合物的方法。
表征谱图
-
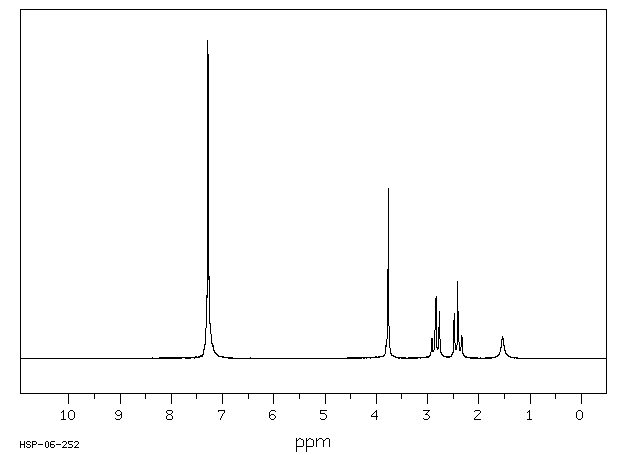
氢谱1HNMR
-
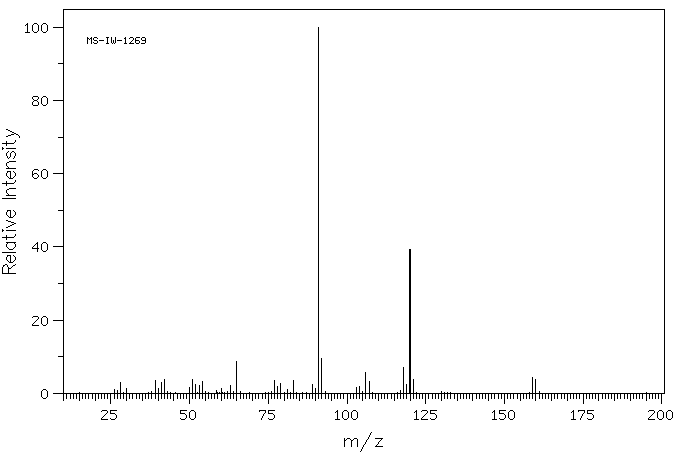
质谱MS
-
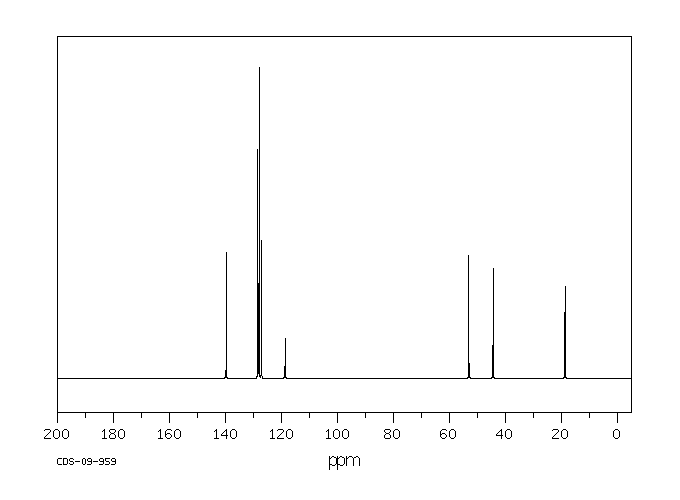
碳谱13CNMR
-
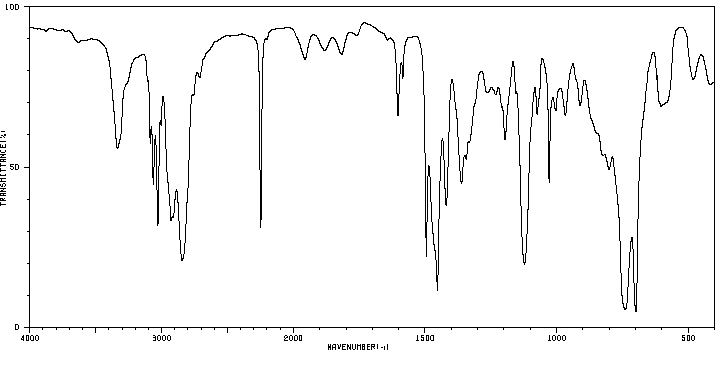
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫