甘氨酰-DL-丝氨酸 | 687-38-7
中文名称
甘氨酰-DL-丝氨酸
中文别名
甘氨酰-DL-丝氨酸水合物
英文名称
N-glycylserine
英文别名
glycyl-serine;2-[(2-azaniumylacetyl)amino]-3-hydroxypropanoate
CAS
687-38-7
化学式
C5H10N2O4
mdl
——
分子量
162.145
InChiKey
BCCRXDTUTZHDEU-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:207 °C
-
沸点:288.82°C (rough estimate)
-
密度:1.4394 (rough estimate)
-
溶解度:可溶于酸性水溶液(轻微)、水(轻微)
-
物理描述:Solid
-
稳定性/保质期:
在常温常压下稳定,应远离氧化物。
计算性质
-
辛醇/水分配系数(LogP):-4.7
-
重原子数:11
-
可旋转键数:4
-
环数:0.0
-
sp3杂化的碳原子比例:0.6
-
拓扑面积:113
-
氢给体数:4
-
氢受体数:5
安全信息
-
WGK Germany:3
-
海关编码:2924199090
-
储存条件:存放在密封容器内,并置于阴凉、干燥处。请将储存地点远离氧化剂和火源。
SDS
Glycyl-DL-serine Hydrate Revision number: 5
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Glycyl-DL-serine Hydrate
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Glycyl-DL-serine Hydrate
Percent: ....
CAS Number: 687-38-7
Synonyms: H-Gly-DL-Ser-OH
Chemical Formula: C5H10N2O4·xH2O
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Glycyl-DL-serine Hydrate
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Handling is performed in a well ventilated place. Wear suitable protective equipment.
Technical measures:
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Form: Crystal- Powder
Colour: White - Almost white
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
No data available
Boiling point/range:
Flash point: No data available
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
No data available
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Glycyl-DL-serine Hydrate
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Nitrogen oxides (NOx)
products:
Section 11. TOXICOLOGICAL INFORMATION
No data available
Acute Toxicity:
Skin corrosion/irritation: No data available
No data available
Serious eye
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
No data available
Reproductive toxicity:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
No data available
Fish:
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
No data available
Soil adsorption (Koc):
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
Not listed
UN-No:
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Glycyl-DL-serine Hydrate
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Glycyl-DL-serine Hydrate
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Glycyl-DL-serine Hydrate
Percent: ....
CAS Number: 687-38-7
Synonyms: H-Gly-DL-Ser-OH
Chemical Formula: C5H10N2O4·xH2O
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Glycyl-DL-serine Hydrate
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Handling is performed in a well ventilated place. Wear suitable protective equipment.
Technical measures:
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Form: Crystal- Powder
Colour: White - Almost white
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
No data available
Boiling point/range:
Flash point: No data available
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
No data available
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Glycyl-DL-serine Hydrate
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Nitrogen oxides (NOx)
products:
Section 11. TOXICOLOGICAL INFORMATION
No data available
Acute Toxicity:
Skin corrosion/irritation: No data available
No data available
Serious eye
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
No data available
Reproductive toxicity:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
No data available
Fish:
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
No data available
Soil adsorption (Koc):
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
Not listed
UN-No:
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Glycyl-DL-serine Hydrate
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 氯乙酰基-DL-丝氨酸 N-chloroacetyl-DL-serine 67206-28-4 C5H8ClNO4 181.576 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— N-glycyl-serine methyl ester 66328-96-9 C6H12N2O4 176.172 —— leucyl=>glycyl=>serine 6511-06-4 C11H21N3O5 275.305
反应信息
-
作为反应物:描述:参考文献:名称:Bergmann; Miekeley, Hoppe-Seyler's Zeitschrift fur Physiologische Chemie, 1924, vol. 140, p. 143摘要:DOI:
-
作为产物:参考文献:名称:Epitaxial growth of ZnO films on Si(111)摘要:在本文中,我们报告了利用脉冲激光沉积技术在硅基底上生长氧化锌薄膜的情况。这些薄膜直接沉积在硅(111)上,也使用了氮化铝和氮化镓薄缓冲层。所有薄膜都具有与基底法线对齐的 c 轴优先取向。使用氮化铝和氮化镓缓冲层的薄膜具有优先面内取向的外延,而直接生长在硅(111)上的薄膜则具有随机面内取向。由于薄膜中存在拉伸应变,拉曼模式的频率降低,带边光致发光峰发生红移。讨论了观察到的双轴应变的各种可能来源。DOI:10.1557/jmr.2002.0361
文献信息
-
Thermodynamic properties of peptide solutions. Part 6.—The amino acid side-chain contributions to the partial molar volumes and heat capacities of some tripeptides in aqueous solution作者:Julia F. Reading、Gavin R. HedwigDOI:10.1039/ft9908603117日期:——molar volumes, V02, and partial molar heat capacities, C0p, 2, have been determined for the tripeptides glycyl-L-valylglycine, glycyl-DL-serylglycine and the D and L isomers of glycylleucylglycine in aqueous solution at 298.15 K. These V02 and C0p, 2 results, in conjunction with those for glycylglycylglycine, were used to estimate the contribution of the side chains to the thermodynamic properties. The
-
Amide bond hydrolysis in peptides and cyclic peptides catalyzed by a dimeric Zr(iv)-substituted Keggin type polyoxometalate作者:Hong Giang T. Ly、Gregory Absillis、Tatjana N. Parac-VogtDOI:10.1039/c3dt50705j日期:——reactions exhibited a bell-shaped profile with the fastest hydrolysis observed in the pD range of 5.5–6.0. Interaction of 1 with Gly-Ser and Gly-Gly via their amine nitrogen and amide oxygen was proven by 13C NMR spectroscopy. The effective hydrolysis of Gly-Ser in the presence of 1 is most likely a combination of the polarization of the amide oxygen due to its binding to the Zr(IV) ion in 1 and the关于水解的详细动力学研究 甘氨酰丝氨酸 (甘氨酸) 和 甘氨酰甘氨酸 (甘氨酸)在二聚锆的存在(IV) -取代的Keggin型多金属氧酸盐(ET 2 NH 2)8 [α-PW 11 ø 39的Zr(μ-OH)(H 2 O)} 2 ]·7H 2 O(1)通过1 H,13 C和31 P NMR光谱学的组合进行。观察到的水解速率常数甘氨酸 和 甘氨酸在pD 5.4和60°C时,分别为63.3×10 -7 s -1和4.44×10 -7 s -1,与未催化反应相比,具有显着的加速作用。两种反应速率常数的pD依赖性显示出钟形曲线,在5.5-6.0的pD范围内观察到最快的水解作用。相互作用1与甘氨酸 和 甘氨酸 13 C NMR光谱证实了它们通过胺氮和酰胺氧的作用。有效水解甘氨酸在1的存在下,最有可能是由于酰胺氧的极化与1中的Zr(IV)离子的结合以及酰胺羰基碳上的Ser羟基的分子内侵蚀所致。还研究
-
<b>An Approach to the Specific Cleavage of Peptide Bonds. I. The Acyl Migration in Dipeptides Containing Hydroxyamino Acids in Anhydrous Hydrogen Fluoride</b>作者:S. Sakakibara、K. H. Shin、G. P. HessDOI:10.1021/ja00883a059日期:1962.12dilute ammonia solution. The combined filtrates were evaporated to small bulk (15 ml.) and acidified with acetic acid yielding quininic acid (207 mg., 83%). Decarboxylation of the Quininic Acid and Phenylation of the 6-Methoxyquinoline.—Dry quininic acid (205 mg.) was mixed with copper chromite catalyst" (205 mg.) and heated (15) D. J. Lussman, E. R. Kirch and O. L. Webster, J. Am. Phorm. Assoc., Set.硫酸奎宁-C14'的氧化。-硫酸奎宁(488mg)溶解在10%硫酸(10ml)中,加入二氧化锰(105mg)。将混合物升至沸点并在半小时内加入溶解在水(2毫升)中的三氧化铬(1.0克)。回流持续3小时。然后加入热水(90毫升)和15N氨(20毫升),静置18小时后。在蒸汽浴上借助硅藻土过滤混合物。残留物用热的稀氨溶液萃取数次。将合并的滤液蒸发至小体积(15毫升)并用乙酸酸化,得到奎宁酸(207毫克,83%)。奎宁酸的脱羧和 6-甲氧基喹啉的苯基化。-将干燥的奎宁酸 (205 毫克) 与亚铬酸铜催化剂混合"(205 毫克。) 和加热 (15) DJ Lussman, ER Kirch 和 OL Webster, J. Am。佛姆。副,设置。Ed., 40, 368 (1951)。
-
Design, synthesis and antimicrobial activity of usnic acid derivatives作者:Kartsev Victor、Lichitsky Boris、Geronikaki Athina、Petrou Anthi、Smiljkovic Marija、Kostic Marina、Radanovic Oliver、Soković MarinaDOI:10.1039/c8md00076j日期:——fungal species was Trichoderma viride, while Penicillium versicolor var. cyclopium appeared to be the most resistant. It should be mentioned that in general most of the compounds showed weaker antibacterial activity, but better antifungal properties than usnic acid itself. The results allow us to conclude that the title compounds are good lead compounds for novel more active antibacterial drugs. On the松萝酸是一种二苯并呋喃,最初是从产生次生代谢产物的地衣中分离出来的,作为一种抗生素而闻名,但还具有其他一些有趣的特性。因此,本文的目的是设计新的松萝酸衍生物并评估其抗菌活性。所有新合成的化合物均具有良好的抗菌活性,MIC范围为1.02–50.93 × 10 -2 mmol mL -1,MBC范围为2.05–70.57 × 10 -2 mmol mL -1。最敏感的细菌种类是金黄色葡萄球菌,而铜绿假单胞菌和大肠杆菌是 ATCC 菌株中耐药性最强的,MRSA 是所有测试细菌(ATCC 和临床分离株)中耐药性最强的。它们的抗真菌活性非常强(MIC = 0.35–7.53 × 10 -2 mmol mL -1和 MFC = 0.70–15.05 × 10 -2 mmol mL -1)——优于参考化合物和松萝酸本身。最敏感的真菌物种是绿色木霉,而杂色青霉变种。cyclopium似乎是最具抵抗力的。应
-
Synthesis of New Purine Derivatives Containing α- and ω-Amino Acid Fragments作者:V. V. Musiyak、I. A. Nizova、T. V. Matveeva、G. L. Levit、V. P. Krasnov、V. N. CharushinDOI:10.1134/s1070428019060046日期:2019.6New conjugates of purine and 2-aminopurine with several α- and ω-amino acids have been synthesized following two approaches based on the condensation and nucleophilic substitution reactions. The enantiomeric purity of the isolated compounds has been confirmed by reversed-phase HPLC using a chiral stationary phase to demonstrate the absence of racemization during the synthesis. The conjugates are inactive
表征谱图
-
氢谱1HNMR
-
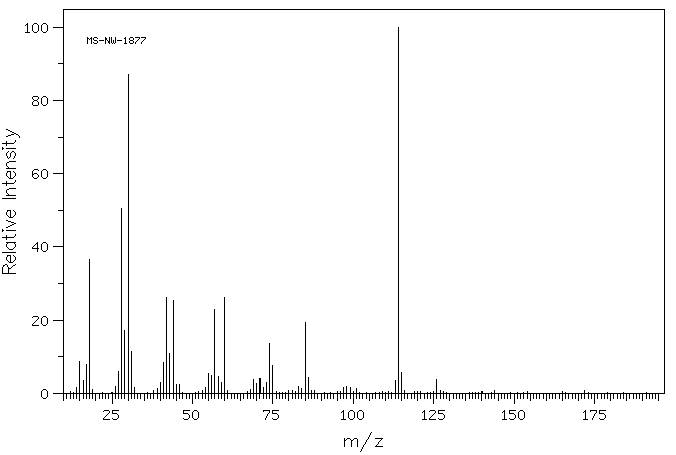
质谱MS
-
碳谱13CNMR
-
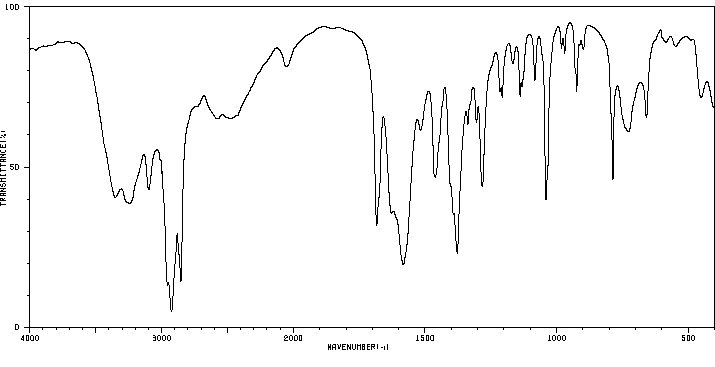
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(甲基3-(二甲基氨基)-2-苯基-2H-azirene-2-羧酸乙酯)
(±)-盐酸氯吡格雷
(±)-丙酰肉碱氯化物
(d(CH2)51,Tyr(Me)2,Arg8)-血管加压素
(S)-(+)-α-氨基-4-羧基-2-甲基苯乙酸
(S)-阿拉考特盐酸盐
(S)-赖诺普利-d5钠
(S)-2-氨基-5-氧代己酸,氢溴酸盐
(S)-2-[[[(1R,2R)-2-[[[3,5-双(叔丁基)-2-羟基苯基]亚甲基]氨基]环己基]硫脲基]-N-苄基-N,3,3-三甲基丁酰胺
(S)-2-[3-[(1R,2R)-2-(二丙基氨基)环己基]硫脲基]-N-异丙基-3,3-二甲基丁酰胺
(S)-1-(4-氨基氧基乙酰胺基苄基)乙二胺四乙酸
(S)-1-[N-[3-苯基-1-[(苯基甲氧基)羰基]丙基]-L-丙氨酰基]-L-脯氨酸
(R)-乙基N-甲酰基-N-(1-苯乙基)甘氨酸
(R)-丙酰肉碱-d3氯化物
(R)-4-N-Cbz-哌嗪-2-甲酸甲酯
(R)-3-氨基-2-苄基丙酸盐酸盐
(R)-1-(3-溴-2-甲基-1-氧丙基)-L-脯氨酸
(N-[(苄氧基)羰基]丙氨酰-N〜5〜-(diaminomethylidene)鸟氨酸)
(6-氯-2-吲哚基甲基)乙酰氨基丙二酸二乙酯
(4R)-N-亚硝基噻唑烷-4-羧酸
(3R)-1-噻-4-氮杂螺[4.4]壬烷-3-羧酸
(3-硝基-1H-1,2,4-三唑-1-基)乙酸乙酯
(2S,4R)-Boc-4-环己基-吡咯烷-2-羧酸
(2S,3S,5S)-2-氨基-3-羟基-1,6-二苯己烷-5-N-氨基甲酰基-L-缬氨酸
(2S,3S)-3-((S)-1-((1-(4-氟苯基)-1H-1,2,3-三唑-4-基)-甲基氨基)-1-氧-3-(噻唑-4-基)丙-2-基氨基甲酰基)-环氧乙烷-2-羧酸
(2S)-2,6-二氨基-N-[4-(5-氟-1,3-苯并噻唑-2-基)-2-甲基苯基]己酰胺二盐酸盐
(2S)-2-氨基-N,3,3-三甲基-N-(苯甲基)丁酰胺
(2S)-2-氨基-3-甲基-N-2-吡啶基丁酰胺
(2S)-2-氨基-3,3-二甲基-N-(苯基甲基)丁酰胺,
(2S)-2-氨基-3,3-二甲基-N-2-吡啶基丁酰胺
(2S,4R)-1-((S)-2-氨基-3,3-二甲基丁酰基)-4-羟基-N-(4-(4-甲基噻唑-5-基)苄基)吡咯烷-2-甲酰胺盐酸盐
(2R,3'S)苯那普利叔丁基酯d5
(2R)-2-氨基-3,3-二甲基-N-(苯甲基)丁酰胺
(2-氯丙烯基)草酰氯
(1S,3S,5S)-2-Boc-2-氮杂双环[3.1.0]己烷-3-羧酸
(1R,5R,6R)-5-(1-乙基丙氧基)-7-氧杂双环[4.1.0]庚-3-烯-3-羧酸乙基酯
(1R,4R,5S,6R)-4-氨基-2-氧杂双环[3.1.0]己烷-4,6-二羧酸
齐特巴坦
齐德巴坦钠盐
齐墩果-12-烯-28-酸,2,3-二羟基-,苯基甲基酯,(2a,3a)-
齐墩果-12-烯-28-酸,2,3-二羟基-,羧基甲基酯,(2a,3b)-(9CI)
黄酮-8-乙酸二甲氨基乙基酯
黄荧菌素
黄体生成激素释放激素(1-6)
黄体生成激素释放激素 (1-5) 酰肼
黄体瑞林
麦醇溶蛋白
麦角硫因
麦芽聚糖六乙酸酯
麦根酸