异硫氰酸仲丁酯 | 4426-79-3
中文名称
异硫氰酸仲丁酯
中文别名
异硫氰酸丁酯
英文名称
sec-butyl isothiocyanate
英文别名
2-butyl isothiocyanate;2-isothiocyanatobutane;sec-Butylisothiocyanat;s-butyl isothiocyanate;1-methylpropyl isothiocyanate
CAS
4426-79-3
化学式
C5H9NS
mdl
MFCD00041108
分子量
115.199
InChiKey
TUFJIDJGIQOYFY-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:58 °C
-
密度:0.93
-
LogP:2.81
-
物理描述:Colourless to yellow liquid; Sharp green slightly irritating aroma
-
溶解度:Very slightly soluble in water; freely soluble in ether
-
折光率:1.490-1.497
-
保留指数:937;904;925
-
稳定性/保质期:
在常温常压下,该物质稳定存在,表现为无色透明至黄色的液体。
计算性质
-
辛醇/水分配系数(LogP):2.8
-
重原子数:7
-
可旋转键数:2
-
环数:0.0
-
sp3杂化的碳原子比例:0.8
-
拓扑面积:44.4
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:IRRITANT-HARMFUL
-
危险等级:8
-
安全说明:S26,S27,S38
-
危险类别码:R23/24/25
-
危险品运输编号:2927
-
海关编码:2930909090
-
储存条件:请将药品存放在避光、通风干燥的地方,并密封保存。
SDS
异硫氰酸仲丁酯 修改号码:5
模块 1. 化学品
产品名称: sec-Butyl Isothiocyanate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害
急性毒性(经口) 第3级
急性毒性(吸入) 第3级
皮肤腐蚀/刺激 第2级
严重损伤/刺激眼睛 2A类
特异性靶器官毒性 呼吸道刺激
- 单一接触 [第3级]
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 危险
危险描述 易燃液体和蒸气
吸入或吞咽会中毒。
造成皮肤刺激
造成严重眼刺激
可能造成呼吸刺激
防范说明
异硫氰酸仲丁酯 修改号码:5
模块 2. 危险性概述
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
避免吸入。
只能在室外或通风良好的环境下使用。
使用本产品时切勿吃东西,喝水或吸烟。
处理后要彻底清洗双手。
穿戴防护手套/护目镜/防护面具。
[急救措施] 吸入:将受害者移到新鲜空气处,在呼吸舒适的地方保持休息。呼叫解毒中心/医生。
食入:立即呼叫解毒中心/医生。
眼睛接触:用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续冲洗。
眼睛接触:求医/就诊
皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激:求医/就诊。
脱掉被污染的衣物,清洗后方可重新使用。
若感不适:呼叫解毒中心/医生。
[储存] 存放于通风良好处。保持容器密闭。
存放处须加锁。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 异硫氰酸仲丁酯
百分比: >98.0%(GC)
CAS编码: 4426-79-3
俗名: Isothiocyanic Acid sec-Butyl Ester
分子式: C5H9NS
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。立即呼叫解毒中心/医生。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用大量肥皂和水轻轻洗。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 立即呼叫解毒中心/医生。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,二氧化碳
不适用的灭火剂: 水(有可能扩大灾情。)
特殊危险性: 小心,燃烧或高温下可能分解产生毒烟。
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
异硫氰酸仲丁酯 修改号码:5
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用特殊的个人防护用品(自携式呼吸器)。远离溢出物/泄露处并处在上风处。确保
紧急措施: 足够通风。
泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
存放于惰性气体环境中。
存放处须加锁。
远离不相容的材料比如氧化剂存放。
气敏
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 半面罩或全面罩呼吸器,自携式呼吸器(SCBA),供气呼吸器等。依据当地和政府法
规,使用通过政府标准的呼吸器。
手部防护: 防渗手套。
眼睛防护: 护目镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防渗防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
外形(20°C): 液体
外观: 透明-微浊
颜色: 极淡的黄色-黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
沸点/沸程 159 °C
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 0.94
溶解度:
[水] 无资料
[其他溶剂] 无资料
异硫氰酸仲丁酯 修改号码:5
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
避免接触的条件: 火花, 明火, 静电
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳, 氮氧化物 (NOx), 硫氧化物
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
副危险性: 第6.1类:毒害品
UN编号: 1992
正式运输名称: 易燃液体, 有毒的, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
异硫氰酸仲丁酯 修改号码:5
模块16 - 其他信息
N/A
模块 1. 化学品
产品名称: sec-Butyl Isothiocyanate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害
急性毒性(经口) 第3级
急性毒性(吸入) 第3级
皮肤腐蚀/刺激 第2级
严重损伤/刺激眼睛 2A类
特异性靶器官毒性 呼吸道刺激
- 单一接触 [第3级]
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 危险
危险描述 易燃液体和蒸气
吸入或吞咽会中毒。
造成皮肤刺激
造成严重眼刺激
可能造成呼吸刺激
防范说明
异硫氰酸仲丁酯 修改号码:5
模块 2. 危险性概述
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
避免吸入。
只能在室外或通风良好的环境下使用。
使用本产品时切勿吃东西,喝水或吸烟。
处理后要彻底清洗双手。
穿戴防护手套/护目镜/防护面具。
[急救措施] 吸入:将受害者移到新鲜空气处,在呼吸舒适的地方保持休息。呼叫解毒中心/医生。
食入:立即呼叫解毒中心/医生。
眼睛接触:用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续冲洗。
眼睛接触:求医/就诊
皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激:求医/就诊。
脱掉被污染的衣物,清洗后方可重新使用。
若感不适:呼叫解毒中心/医生。
[储存] 存放于通风良好处。保持容器密闭。
存放处须加锁。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 异硫氰酸仲丁酯
百分比: >98.0%(GC)
CAS编码: 4426-79-3
俗名: Isothiocyanic Acid sec-Butyl Ester
分子式: C5H9NS
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。立即呼叫解毒中心/医生。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用大量肥皂和水轻轻洗。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 立即呼叫解毒中心/医生。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,二氧化碳
不适用的灭火剂: 水(有可能扩大灾情。)
特殊危险性: 小心,燃烧或高温下可能分解产生毒烟。
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
异硫氰酸仲丁酯 修改号码:5
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用特殊的个人防护用品(自携式呼吸器)。远离溢出物/泄露处并处在上风处。确保
紧急措施: 足够通风。
泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
存放于惰性气体环境中。
存放处须加锁。
远离不相容的材料比如氧化剂存放。
气敏
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 半面罩或全面罩呼吸器,自携式呼吸器(SCBA),供气呼吸器等。依据当地和政府法
规,使用通过政府标准的呼吸器。
手部防护: 防渗手套。
眼睛防护: 护目镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防渗防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
外形(20°C): 液体
外观: 透明-微浊
颜色: 极淡的黄色-黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
沸点/沸程 159 °C
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 0.94
溶解度:
[水] 无资料
[其他溶剂] 无资料
异硫氰酸仲丁酯 修改号码:5
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
避免接触的条件: 火花, 明火, 静电
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳, 氮氧化物 (NOx), 硫氧化物
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
副危险性: 第6.1类:毒害品
UN编号: 1992
正式运输名称: 易燃液体, 有毒的, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
异硫氰酸仲丁酯 修改号码:5
模块16 - 其他信息
N/A
反应信息
-
作为反应物:参考文献:名称:Reymann, Chemische Berichte, 1874, vol. 7, p. 1288摘要:DOI:
-
作为产物:描述:N-butan-2-ylcarbamodithioate;triethylazanium 以 二氯甲烷 为溶剂, 反应 0.33h, 以0.115 g的产率得到异硫氰酸仲丁酯参考文献:名称:微波直接合成异硫氰酸酯摘要:微波技术的应用允许完成从胺类合成异硫氰酸酯的新颖,通用,“绿色”的一锅操作规程。反应易于扩展并且在不消旋手性胺的情况下进行。在这些条件下,中间体二硫代氨基甲酸酯可分解为异硫氰酸酯,而无需任何其他脱硫剂。DOI:10.1002/ejoc.201900105
文献信息
-
Synthesis and antitumor activity of new thiosemicarbazones of 2-acetylimidazo[4,5-<i>b</i>]pyridine作者:Stavros Mylonas、Athanasios MamalisDOI:10.1002/jhet.5570420705日期:2005.11A number of thiosemicarbazones of 2-acetyl-imidazo[4,5-b]pyridine were prepared in order to investigate their in vitro antineoplastic activities. Three compounds: (i) 2-acetylimidazo[4,5-b]pyridin-4-sec-butyl-3-thiosemicarbazone [(A7), NSC674098], (ii) 2-acetylimidazo[4,5-b]pyridin-4-tert-butyl-3-thiosemi-carbazone [(A9), NSC674099], (iii) 2-acetylimidazo[4,5-b]pyridin-4-cyclohexyl-3-thiosemicarbozone为了研究它们的体外抗肿瘤活性,制备了许多2-乙酰基-咪唑并[4,5- b ]吡啶的硫半脲。三种化合物:(i)2-乙酰基咪唑并[4,5- b ]吡啶-4-仲丁基-3-硫代半缩氨基甲酰胺[(A 7),NSC674098],(ii)2-乙酰基咪唑并[4,5- b ]吡啶-4-叔丁基-3-thiosemicarbozone [(A 9),NSC674099],(iii)2-乙酰基咪唑并[4,5- b ]吡啶-4-环己基-3-thiosemicarbozone [[A 11),NSC674101]对某些测试的细胞系显示出显着的活性。NCI的生物学评估委员会决定应进行进一步的二级检测(这些化合物已针对前列腺癌进行了检测)。
-
(Propargylsulfanyl)-2-aza-1,3,5-trienes as a direct source for novel family of highly functionalized 4,5-dihydro-1,3-thiazoles作者:Nina A. Nedolya、Ol'ga A. Tarasova、Alexander I. Albanov、Lyudmila V. Klyba、Boris A. TrofimovDOI:10.1016/j.tet.2016.12.064日期:2017.22-(1-alkoxyprop-1-enyl)-5-ethenylidene-4,5-dihydro-1,3-thiazoles has been disclosed through the reaction of (propargylsulfanyl)-2-aza-1,3,5-trienes (readily available from alkoxyallenes, isothiocyanates, and propargyl bromide) with potassium or sodium tert-butoxide (1.1–1.4 equiv) under mild conditions [DMSO/THF (∼1:4–5), ca. −30 °C, 15–30 min]. The process proceeds through deprotonation of an activated SCH2 group
-
Mass spectra of new heterocycles: X. Fragmentation of the molecular ions of 1-alkyl(cycloalkyl, aryl)-3-alkoxy(aryl)-2-methylsulfanyl-1H-pyrroles作者:L. V. Klyba、N. A. Nedolya、O. A. Tarasova、E. R. Zhanchipova、O. G. VolostnykhDOI:10.1134/s1070428010070134日期:2010.7The mass spectra of previously unknown 1-alkyl(cycloalkyl, aryl)-3-alkoxy(aryl)-2-methylsulfanyl-1H-pyrroles were studied. Fragmentation of all 3-alkoxy-substituted pyrroles under electron impact (70 eV) follow both ether and sulfide decomposition paths; In particular, 1-R-substituted 3-methoxy-2-methylsulfanyl-1H-pyrroles (R = Me, Et, i-Pr, s-Bu, cyclo-C5H9, cyclo-C6H11, Ph) lose methyl radical group研究了以前未知的1-烷基(环烷基,芳基)-3-烷氧基(芳基)-2-甲基硫基-1 H-吡咯的质谱。所有3-烷氧基取代的吡咯在电子轰击(70 eV)下的裂解均遵循醚和硫化物的分解路径。特别是1-R-取代的3-甲氧基-2-甲基硫烷基-1 H-吡咯(R = Me,Et,i- Pr,s- Bu,环-C 5 H 9,环-C 6 H 11,Ph )从甲氧基和甲基硫烷基基团失去甲基基团。1秒的质谱丁基和1环烷基吡咯也有一个强峰(10-49%),该峰来自通过同步氢转移裂解NR键形成的奇电子[ M C n H 2 n ] +·离子。3-烷氧基-1-异丙基-2-甲基硫烷基-1 H-吡咯(Alk = Et,i -Pr,t -Bu)断裂中的O-Alk键断裂伴随重排过程,导致相应的烯烃和1-电子的1-异丙基-2-甲基硫烷基-1 H-吡咯-3-醇离子。1-烷基-2-甲基硫烷基-3-苯基-1 H-吡咯的主要裂解途径(Alk
-
Antimicrobial Activity of Newly Synthesized Isothiocyanate Derivatives Against Pathogenic Plant Microorganisms作者:M. Kado、Misao KojimaDOI:10.1002/jps.2600651039日期:1976.10Fifteen reaction products of isothiocyanates with cysteine, seven reaction products of isothiocyanates with 2,3-dimercapto-1-propanol, and four reaction products of isothiocyanates with sulfanilamide were synthesized. Their antimicrobial activity against pathogenic plant microorganisms was investigated.
-
Effect on <i>K</i><sub>ATP</sub> Channel Activation Properties and Tissue Selectivity of the Nature of the Substituent in the 7- and the 3-Position of 4<i>H</i>-1,2,4-Benzothiadiazine 1,1-Dioxides作者:Stéphane Boverie、Marie-Hélène Antoine、Fabian Somers、Bénédicte Becker、Sophie Sebille、Raogo Ouedraogo、Stéphane Counerotte、Bernard Pirotte、Philippe Lebrun、Pascal de TullioDOI:10.1021/jm0311339日期:2005.5.1structurally related to previously described potassium channel openers such as the benzothiadiazine dioxide BPDZ 73, were tested as putative K(ATP) channel activators on the pancreatic endocrine tissue and on the vascular smooth muscle tissue. The nature of the substituent introduced in the 7-position as well as the nature of the alkylamino side chain in the 3-position strongly affected both potency and tissue本工作探讨了在7位上不同取代的3-烷基氨基-4H-1,2,4-苯并噻二嗪1,1-二氧化物。那些与先前描述的钾通道开放剂(例如二氧化苯并噻二嗪BPDZ 73)在结构上相关的化合物已在胰腺内分泌组织和血管平滑肌组织上作为推定的K(ATP)通道活化剂进行了测试。在7位引入的取代基的性质以及在3位引入的烷基氨基侧链的性质强烈影响4H-1,2,4-苯并噻二嗪1,1-二氧化物的效能和组织选择性。因此,在7位上带有甲基或甲氧基或在该位置上没有取代基并带有乙基,异丙基的化合物,发现3-位的环丁基或环丁基氨基是有效的和选择性的胰岛素从大鼠胰腺B细胞(即10a,10b,12b,12d,22c)释放的抑制剂。相反,3-烷基氨基-7-三氟甲基-(20a-c)和3-烷基氨基-7-戊基-4H-1,2,4-苯并噻二嗪1,1-二氧化物(11a,b)在表面上表现出明显的肌松活性。大鼠主动脉环。在后一种化合物中,3-烷基氨基
表征谱图
-
氢谱1HNMR
-
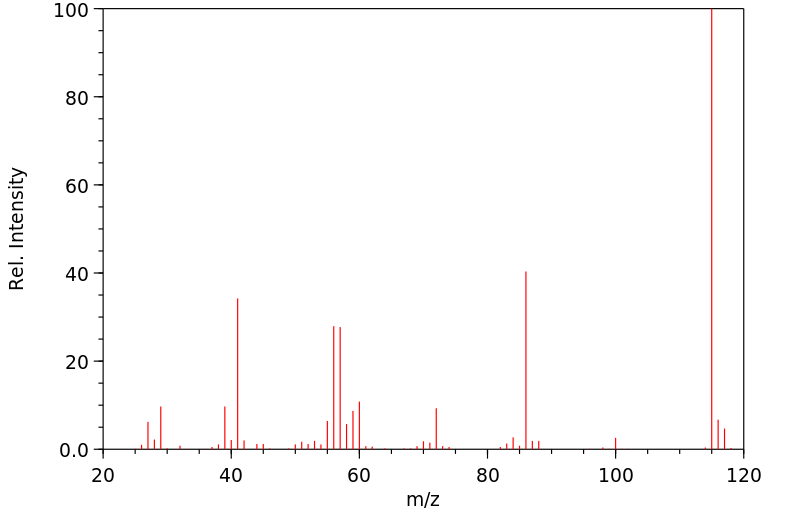
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-2-异硫氰基-1-环戊羧酸乙酯
顺-2-异硫氰基-1-环己烷羧酸乙酯
羰基异氰酸酯异硫氰酸酯
羰基二异硫氰酸酯
硫代异氰酸环戊酯
甲代烯丙基异硫氰酸酯
环己烷羰基异硫氰酸酯
环己基异硫氰酸脂
环丙基甲基异硫氰酸酯
环丁烷羰基异硫氰酸酯
异硫氰酸甲酯
异硫氰酸甲氧基甲酯
异硫氰酸甲基环己酯
异硫氰酸环辛酯
异硫氰酸环庚脂
异硫氰酸环十二酯
异硫氰酸环丙酯
异硫氰酸烯丙酯
异硫氰酸溴代乙酯
异硫氰酸氯代乙酯
异硫氰酸氨基甲酰
异硫氰酸异戊酯
异硫氰酸异丙酯
异硫氰酸异丁酯
异硫氰酸己酯
异硫氰酸叔辛酯
异硫氰酸十六酯
异硫氰酸十一烷酯
异硫氰酸仲丁酯
异硫氰酸乙酰酯
异硫氰酸乙酯
异硫氰酸乙烯
异硫氰酸3-丁烯酯
异硫氰酸2-甲氧基乙酯
异硫氰酸1-金刚烷酯
异硫氰酰甲酸甲酯
异硫氰酰甲酸乙酯
异硫氰基环丁烷
异硫代氰酰基乙醛二甲基乙缩醛
异氰酸丙酯
己烷,1-[(2-异硫氰基乙基)硫代]-
己烯,1-异硫氰基-
天然芥菜籽油
叔戊基异硫氰酸酯
叔丁基异硫氰酸酯
双(1-异硫氰基乙基)醚
十八烷基异氰酸酯
二(异硫氰酰甲基)醚
丙脒,N-丁基-2-氯-
三芥子酸甘油酯