5-氟苯乙睛 | 653-30-5
中文名称
5-氟苯乙睛
中文别名
5-氟苯乙腈;;五氟苯乙腈;2,3,4,5,6-五氟苯基乙腈;5-氟苯乙腈
英文名称
(pentafluorophenyl)acetonitrile
英文别名
2,3,4,5,6-pentafluorophenylacetonitrile;Pentafluorphenyl-acetonitril;pentafluorobenzyl cyanide;2-(perfluorophenyl)acetonitrile;2-(pentafluorophenyl)acetonitrile;2-(2,3,4,5,6-pentafluorophenyl)acetonitrile
CAS
653-30-5
化学式
C8H2F5N
mdl
——
分子量
207.103
InChiKey
YDNOJUAQBFXZCR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:107-111 °C17 mm Hg(lit.)
-
密度:1.61 g/mL at 25 °C(lit.)
-
闪点:>230 °F
-
最大波长(λmax):255nm(CH3CN)(lit.)
-
暴露限值:NIOSH: IDLH 25 mg/m3
-
稳定性/保质期:
远离氧化物。
计算性质
-
辛醇/水分配系数(LogP):2.1
-
重原子数:14
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.12
-
拓扑面积:23.8
-
氢给体数:0
-
氢受体数:6
安全信息
-
危险等级:6.1
-
危险品标志:Xn
-
危险类别码:R20/21/22,R36/37/38
-
危险品运输编号:3276
-
WGK Germany:3
-
海关编码:2926909090
-
包装等级:III
-
危险类别:6.1
-
安全说明:S26,S37/39
-
危险性防范说明:P260,P264,P270,P271,P280,P301+P310,P302+P352,P304+P340,P305+P351+P338,P311,P312,P321,P322,P330,P332+P313,P337+P313,P361,P362,P363,P403+P233,P405,P501
-
危险性描述:H301,H311,H315,H319,H331
-
储存条件:应将存放在密封容器内,并放置在阴凉、干燥处。存储地点需远离氧化剂。
SDS
| Name: | 2 3 4 5 6-Pentafluorophenylacetonitrile Material Safety Data Sheet |
| Synonym: | |
| CAS: | 653-30-5 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 653-30-5 | 2,3,4,5,6-Pentafluorophenylacetonitril | 100 | 211-498-6 |
Risk Phrases: 20/21/22 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.
Irritating to eyes, respiratory system and skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Wash clothing before reuse.
Ingestion:
If victim is conscious and alert, give 2-4 cupfuls of milk or water.
Never give anything by mouth to an unconscious person. Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas.
Extinguishing Media:
Use extinguishing media most appropriate for the surrounding fire.
In case of fire, use carbon dioxide, dry chemical powder or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Clean up spills immediately, observing precautions in the Protective Equipment section. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Use only in a well-ventilated area.
Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Wash clothing before reuse.
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 653-30-5: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear, colorless
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 107.0 - 111.0 deg C @ 17.00mm
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: 109 deg C ( 228.20 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.6100g/cm3
Molecular Formula: C8H2F5N
Molecular Weight: 207.10
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, excess heat.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, oxides of nitrogen, irritating and toxic fumes and gases, carbon dioxide, hydrogen fluoride gas.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 653-30-5 unlisted.
LD50/LC50:
Not available.
CAS# Oral, rat: LD50 = 270 mg/kg.; Inhalation, rat: LC50: = 430 mg/m3/2H.; Dermal, rabbit: LD50 = 270 mg/kg. The toxicity of this product is partially based on the hazards associated with phenylacetonitrile. Carcinogenicity:
2,3,4,5,6-Pentafluorophenylacetonitrile - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
WGK (Water Danger/Protection)
CAS# 653-30-5: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 653-30-5 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 653-30-5 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
化学性质:这是一种无色至浅黄色的液体,沸点在107℃至111℃(17mmHg),闪点超过110℃,折光率为1.4390,比重为1.610。
用途:广泛应用于医药、农药及液晶材料中间体领域。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,3,4,5,6-五氟甲苯 2,3,4,5,6-pentafluorotoluene 771-56-2 C7H3F5 182.093 五氟苯甲醛 perfluorobenzaldehyde 653-37-2 C7HF5O 196.076 α-溴-2,3,4,5,6-五氟甲基苯酸酯 (bromomethyl)pentafluorobenzene 1765-40-8 C7H2BrF5 260.989 五氟苯甲醇 (2,3,4,5,6-pentafluorophenyl)methanol 440-60-8 C7H3F5O 198.092 2,3,4,5,6-五氟苄基氯 2,3,4,5,6-pentafluorobenzyl chloride 653-35-0 C7H2ClF5 216.538 —— ethyl 2-cyano-2-(perfluorophenyl)acetate 2340-87-6 C11H6F5NO2 279.166 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— (2,3,5,6-tetrafluorophenyl)acetonitrile 53001-67-5 C8H3F4N 189.112 2-(五氟苯基)乙醇 2-(pentafluorophenyl)ethanol 653-31-6 C8H5F5O 212.119 2,3,4,5,6-五氟苯乙胺 2-(2,3,4,5,6-pentafluorophenyl)ethan-1-amine 1583-76-2 C8H6F5N 211.134 —— pentafluorophenylfluoroacetonitrile 91407-85-1 C8HF6N 225.093 2-(2,3,4,5,6-五氟苯基)乙酰胺 2.3.4.5.6-Pentafluor-phenylacetamid 653-20-3 C8H4F5NO 225.118 5-氟苯乙酸 2,3,4,5,6-pentafluorophenylacetic acid 653-21-4 C8H3F5O2 226.103 —— Bis(pentafluorophenyl)acetonitrile 42238-34-6 C14HF10N 373.153 —— α-(2,3,5,6-Tetrafluor-phenyl)-2,3,4,5,6-pentafluor-phenylacetonitril 42254-09-1 C14H2F9N 355.162 —— ethyl pentafluorophenylacetate 784-35-0 C10H7F5O2 254.156 —— α-(4-Cyanmethyl-2,3,5,6-tetrafluor-phenyl)-2,3,4,5,6-pentafluor-phenylacetonitril 28744-84-5 C16H3F9N2 394.199 —— α-(4-Chlor-2,3,5,6-tetrafluor-phenyl)-2,3,4,5,6-pentafluor-phenylacetonitril 42238-36-8 C14HClF9N 389.607 —— α-(4-Brom-2,3,5,6-tetrafluor-phenyl)-2,3,4,5,6-pentafluor-phenylacetonitril 42238-37-9 C14HBrF9N 434.058 - 1
- 2
反应信息
-
作为反应物:描述:5-氟苯乙睛 在 manganese(IV) oxide 、 potassium fluoride 、 sodium tetrahydroborate 、 三氟乙酸 作用下, 以 四氢呋喃 、 N,N-二甲基甲酰胺 、 苯 为溶剂, 反应 67.0h, 生成 4,5,6,7-四氟吲哚参考文献:名称:7,8,9,10-四氟玫瑰树碱的合成摘要:我们用六氟苯和氰基乙酸乙酯通过 1-(苯磺酰基)-4,5,6,7-四氟吲哚分九步合成了一种新型玫瑰树碱类似物 7,8,9,10-四氟玫瑰树碱。关键步骤是吲哚锂化,随后与 3,4-吡啶二羧酸酐偶联得到酮内酰胺。内酰胺与甲基锂反应,然后用硼氢化钠还原,得到 7,8,9,10-四氟玫瑰树碱。DOI:10.24820/ark.5550190.p010.414
-
作为产物:描述:参考文献:名称:173.芳族多氟化合物。第八部分 五氟苯甲醛及相关的五氟苯基酮和羧酸摘要:DOI:10.1039/jr9610000808
文献信息
-
Synthesis and Structure−Activity Relationships of 2-Pyridones: A Novel Series of Potent DNA Gyrase Inhibitors as Antibacterial Agents作者:Qun Li、Daniel T. W. Chu、Akiyo Claiborne、Curt S. Cooper、Cheuk M. Lee、Kathleen Raye、Kristine B. Berst、Pamela Donner、Weibo Wang、Lisa Hasvold、Anthony Fung、Zhenkun Ma、Michael Tufano、Robert Flamm、Linus L. Shen、John Baranowski、Angela Nilius、Jeff Alder、Jonathan Meulbroek、Kennan Marsh、DeAnne Crowell、Yuhua Hui、Louis Seif、Laura M. Melcher、Rodger Henry、Steven Spanton、Ramin Faghih、Larry L. Klein、S. Ken Tanaka、Jacob J. PlattnerDOI:10.1021/jm960207w日期:1996.1.1Two novel series of 2-pyridones were synthesized by transposition of the nitrogen of 4-quinolones to the bridgehead position. This subtle interchange of the nitrogen atom with a carbon atom yielded two novel heterocyclic nuclei, pyrido[1,2-alpha]pyrimidine and quinolizine, which had not previously been evaluated as antibacterial agents and were found to be potent inhibitors of DNA gyrase. Quinolizines通过将4-喹诺酮类的氮转位至桥头位置,合成了两个新颖的2-吡啶酮系列。氮原子与碳原子的这种微妙的交换产生了两个新的杂环核,吡啶并[1,2-α]嘧啶和喹啉嗪,它们先前未被评估为抗菌剂,并且被发现是DNA促旋酶的有效抑制剂。(S)-45a(ABT-719)等在9位甲基的喹唑啉酮具有出色的广谱抗菌活性。最值得注意的是,它们对耐药菌具有活性,例如耐甲氧西林的金黄色葡萄球菌,耐万古霉素的肠球菌菌株和耐环丙沙星的生物。此外,2-吡啶酮还具有良好的理化和药代动力学特性。这些2-吡啶酮是通过10-17个线性转化从商购可得的起始原料合成的。通过X射线晶体学分析确定由该序列产生的加合物(S)-45a(ABT-719)的结构。
-
Discovery of Novel Non-Peptide CCR1 Receptor Antagonists作者:Howard P. Ng、Karen May、John G. Bauman、Ameen Ghannam、Imadul Islam、Meina Liang、Richard Horuk、Joseph Hesselgesser、R. Michael Snider、H. Daniel Perez、Michael M. MorrisseyDOI:10.1021/jm990316l日期:1999.11.1number of chronic inflammatory diseases, most notably multiple sclerosis and rheumatoid arthritis. Because these ligands share a common receptor, CCR1, we sought to discover antagonists for this receptor as an approach to treating these disorders. A novel series of 4-hydroxypiperidines has been discovered by high throughput screening (HTS) which potently inhibits the binding of MIP-1alpha and RANTES toCCR1受体的配体(MIP-1alpha和RANTES)与多种慢性炎症性疾病有关,最明显的是多发性硬化症和类风湿关节炎。由于这些配体共享一个共同的受体CCR1,因此我们寻求发现该受体的拮抗剂作为治疗这些疾病的一种方法。通过高通量筛选(HTS)已经发现了一系列新型的4-羟基哌啶,其有效抑制了MIP-1α和RANTES与重组人CCR1趋化因子受体的结合。该模板各部分的结构活性关系被描述为:最初的HTS导联1已通过合成优化为高效受体拮抗剂6s。与其他人类7-TM受体相比,该化合物对CCR1的抑制作用具有至少200倍的选择性,包括其他趋化因子受体。此外,从体外功能测定中获得的数据证明了化合物6s和与结构相关的类似物对CCR1受体的功能拮抗作用具有浓度依赖性。以化合物6s为代表的有效和选择性CCR1受体拮抗剂的发现和优化可能代表了一种治疗慢性炎性疾病的新方法。
-
정공 주입 특성을 구비한 유기 화합물 및 그를 이용한 유기 발광 소자와 디스플레이 장치申请人:LG Display Co.,Ltd. 엘지디스플레이 주식회사(119981018655) Corp. No ▼ 110111-0393134公开号:KR20160083249A公开(公告)日:2016-07-12본 발명은, 하기 화학식 1: 화학식 1 로 표시되는 정공 주입 특성을 구비한 유기 화합물 및 그를 이용한 유기 발광 소자와 디스플레이 장치에 관한 것으로서, 본 발명에 따르면 유기 발광 소자의 구동 전압을 낮출 수 있다.本发明涉及如下化学式1所示的具有正空注入特性的有机化合物及其用于的有机发光器件和显示装置,根据本发明,可以降低有机发光器件的驱动电压。
-
Diazaphospholene-Catalyzed Hydrodefluorination of Polyfluoroarenes with Phenylsilane via Concerted Nucleophilic Aromatic Substitution作者:Jingjing Zhang、Xiao Zhao、Jin-Dong Yang、Jin-Pei ChengDOI:10.1021/acs.joc.1c02360日期:2022.1.7The metal-free catalytic C–F bond activation of polyfluoroarenes was achieved with diazaphospholene as the catalyst and phenylsilane as the terminal reductant. Density functional theory calculations suggested a concerted nucleophilic aromatic substitution mechanism.
-
Extended benzodifuran–furan derivatives as example of π-conjugated materials obtained from sustainable approach作者:Chady Moussallem、Frédéric Gohier、Charlotte Mallet、Magali Allain、Pierre FrèreDOI:10.1016/j.tet.2012.07.079日期:2012.10The synthesis of extended benzodifuran–furan systems are described as an example of π-conjugated materials prepared by following a green approach with only water being produced as waste and using furan derivatives from renewable sources. Investigation of their optical and electrochemical properties shows that the new compounds present electronic properties compatible for application in organic electronics
表征谱图
-
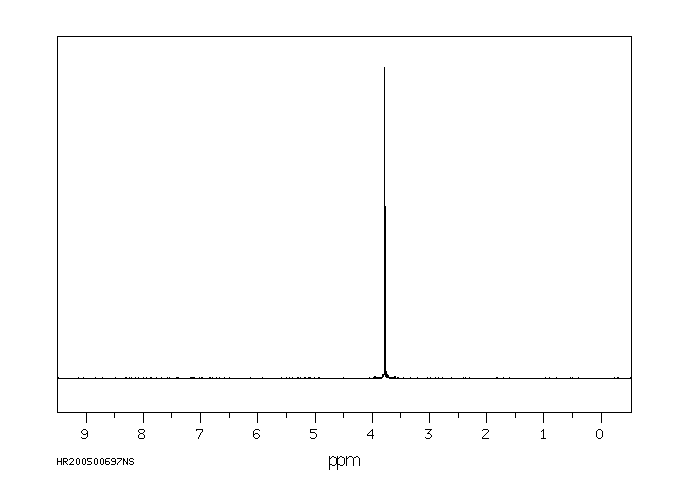
氢谱1HNMR
-
质谱MS
-
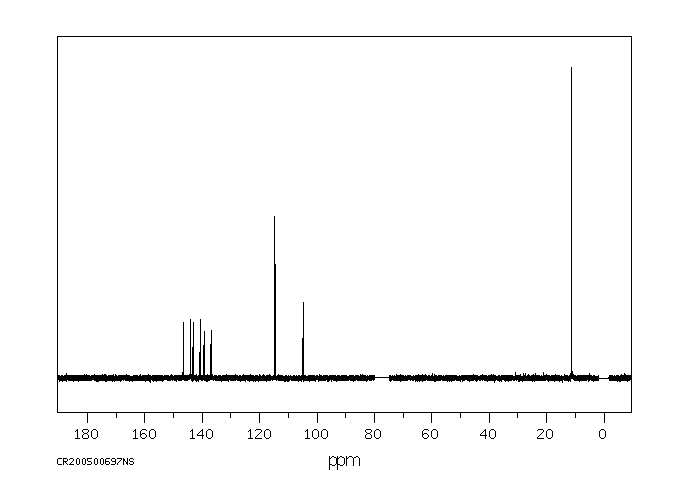
碳谱13CNMR
-
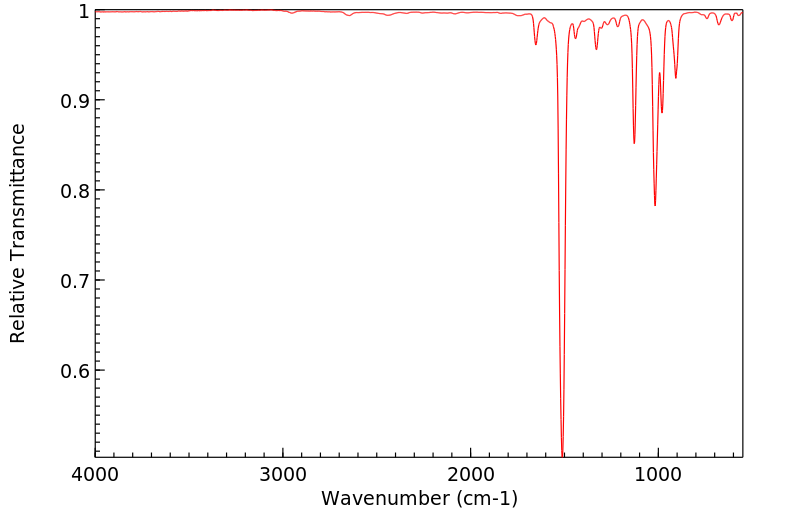
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫