4-甲基-1-萘醛 | 33738-48-6
中文名称
4-甲基-1-萘醛
中文别名
——
英文名称
4-methyl-1-naphthaldehyde
英文别名
1-methyl-4-naphthaldehyde;4-methylnaphthaldehyde;4-methylnaphthalene-1-carboxaldehyde;4-methylnaphthalene-1-carbaldehyde
CAS
33738-48-6
化学式
C12H10O
mdl
MFCD00060691
分子量
170.211
InChiKey
LANRGTXVAPBSIA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:32-36 °C(lit.)
-
沸点:259.85°C (rough estimate)
-
密度:1.1252
-
闪点:>230 °F
-
溶解度:可溶于氯仿(少许)、甲醇(少许)
-
稳定性/保质期:
如果按照规格使用和储存,则不会分解,未有已知危险发生。
避免与强氧化剂接触。
计算性质
-
辛醇/水分配系数(LogP):3
-
重原子数:13
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.083
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
WGK Germany:3
-
危险品运输编号:NONH for all modes of transport
-
海关编码:2912299000
-
危险性防范说明:P233,P260,P261,P264,P271,P280,P302+P352,P304,P304+P340,P305+P351+P338,P312,P321,P332+P313,P337+P313,P340,P362,P403,P403+P233,P405,P501
-
危险性描述:H315,H319,H335
-
储存条件:请将药品存放在密闭、阴凉干燥处,并保持良好通风。
SDS
Section 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE
Product identifiers
Product name : 4-Methyl-1-naphthaldehyde
CAS-No. : 33738-48-6
Relevant identified uses of the substance or mixture and uses advised against
Identified uses : Laboratory chemicals, Manufacture of substances
Section 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
This substance is not classified as dangerous according to Directive 67/548/EEC.
Label elements
The product does not need to be labelled in accordance with EC directives or respective national laws.
Other hazards - none
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
Formula : C12H10O
Molecular Weight : 170,21 g/mol
Section 4. FIRST AID MEASURES
Description of first aid measures
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration.
In case of skin contact
Wash off with soap and plenty of water.
In case of eye contact
Flush eyes with water as a precaution.
If swallowed
Never give anything by mouth to an unconscious person. Rinse mouth with water.
Most important symptoms and effects, both acute and delayed
To the best of our knowledge, the chemical, physical, and toxicological properties have not been
thoroughly investigated.
Indication of any immediate medical attention and special treatment needed
no data available
Section 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
Special hazards arising from the substance or mixture
Carbon oxides
Advice for firefighters
Wear self contained breathing apparatus for fire fighting if necessary.
Further information
no data available
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Avoid dust formation. Avoid breathing vapors, mist or gas.
Environmental precautions
Do not let product enter drains.
Methods and materials for containment and cleaning up
Sweep up and shovel. Keep in suitable, closed containers for disposal.
Reference to other sections
For disposal see section 13.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Provide appropriate exhaust ventilation at places where dust is formed.Normal measures for preventive fire
protection.
Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place.
Specific end uses
no data available
Section 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Control parameters
Components with workplace control parameters
Exposure controls
Appropriate engineering controls
General industrial hygiene practice.
Personal protective equipment
Eye/face protection
Use equipment for eye protection tested and approved under appropriate government standards
such as NIOSH (US) or EN 166(EU).
Skin protection
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique
(without touching glove's outer surface) to avoid skin contact with this product. Dispose of
contaminated gloves after use in accordance with applicable laws and good laboratory practices.
Wash and dry hands.
The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and
the standard EN 374 derived from it.
Body Protection
Choose body protection in relation to its type, to the concentration and amount of dangerous
substances, and to the specific work-place., The type of protective equipment must be selected
according to the concentration and amount of the dangerous substance at the specific workplace.
Respiratory protection
Respiratory protection is not required. Where protection from nuisance levels of dusts are desired,
use type N95 (US) or type P1 (EN 143) dust masks. Use respirators and components tested and
approved under appropriate government standards such as NIOSH (US) or CEN (EU).
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
a) Appearance Form: solid
b) Odour no data available
c) Odour Threshold no data available
d) pH no data available
e) Melting point/freezing Melting point/range: 32 - 36 °C - lit.
point
f) Initial boiling point and no data available
boiling range
g) Flash point 110 °C - closed cup
h) Evaporation rate no data available
i) Flammability (solid, gas) no data available
j) Upper/lower no data available
flammability or
explosive limits
k) Vapour pressure no data available
l) Vapour density no data available
m) Relative density no data available
n) Water solubility no data available
o) Partition coefficient: n- no data available
octanol/water
p) Autoignition no data available
temperature
q) Decomposition no data available
temperature
r) Viscosity no data available
s) Explosive properties no data available
t) Oxidizing properties no data available
Other safety information
no data available
Section 10. STABILITY AND REACTIVITY
Reactivity
no data available
Chemical stability
no data available
Possibility of hazardous reactions
no data available
Conditions to avoid
no data available
Incompatible materials
Strong oxidizing agents
Hazardous decomposition products
Other decomposition products - no data available
Section 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity
no data available
Skin corrosion/irritation
no data available
Serious eye damage/eye irritation
no data available
Respiratory or skin sensitization
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
IARC: No component of this product present at levels greater than or equal to 0.1% is identified as
probable, possible or confirmed human carcinogen by IARC.
Reproductive toxicity
no data available
Specific target organ toxicity - single exposure
no data available
Specific target organ toxicity - repeated exposure
no data available
Aspiration hazard
no data available
Potential health effects
Inhalation
May be harmful if inhaled. May cause respiratory tract irritation.
Ingestion May be harmful if swallowed.
Skin May be harmful if absorbed through skin. May cause skin irritation.
Eyes May cause eye irritation.
Signs and Symptoms of Exposure
To the best of our knowledge, the chemical, physical, and toxicological properties have not been
thoroughly investigated.
Additional Information
RTECS: Not available
Section 12. ECOLOGICAL INFORMATION
Toxicity
no data available
Persistence and degradability
no data available
Bioaccumulative potential
no data available
Mobility in soil
no data available
Results of PBT and vPvB assessment
no data available
Other adverse effects
no data available
Section 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company.
Contaminated packaging
Dispose of as unused product.
Section 14. TRANSPORT INFORMATION
UN number
ADR/RID: - IMDG: - IATA: -
UN proper shipping name
ADR/RID: Not dangerous goods
IMDG: Not dangerous goods
IATA: Not dangerous goods
Transport hazard class(es)
ADR/RID: - IMDG: - IATA: -
Packaging group
ADR/RID: - IMDG: - IATA: -
Environmental hazards
ADR/RID: no IMDG Marine pollutant: no IATA: no
Special precautions for user
no data available
SECTION 15 - REGULATORY INFORMATION
N/A
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 1,4-二甲基萘 1,4-dimethylnaphthalene 571-58-4 C12H12 156.227 甲基萘 1-Methylnaphthalene 90-12-0 C11H10 142.2 (1-甲基萘-4-基)甲醇 4-methyl-1-naphthylmethanol 57322-44-8 C12H12O 172.227 9,10-二氰基蒽 9,10-Dicyanoanthracene 1217-45-4 C16H8N2 228.253 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2,4-dimethyl-1-formylnaphthalene 36225-26-0 C13H12O 184.238 4-甲基-1-萘甲酸 4-Methyl-1-naphthoic acid 4488-40-8 C12H10O2 186.21 甲酮,(4-甲基-1-萘基)苯基- 1-benzoyl-4-methylnaphthalene 116672-26-5 C18H14O 246.309
反应信息
-
作为反应物:描述:4-甲基-1-萘醛 在 2-氨基苯磺酸 、 [双(三氟乙酰氧基)碘]苯 、 bis(dibenzylideneacetone)-palladium(0) 作用下, 以 1,2-二氯乙烷 为溶剂, 以60%的产率得到4-methyl-8-hydroxy-1-naphthaldehyde参考文献:名称:Palladium-Catalyzed Regiospecific peri- and ortho-C–H Oxygenations of Polyaromatic Rings Mediated by Tunable Directing Groups摘要:DOI:10.1021/acs.orglett.0c03701
-
作为产物:参考文献:名称:Dearomative Allylation of Naphthyl Cyanohydrins by Palladium Catalysis: Catalyst-Enhanced Site Selectivity摘要:A dearomative allylation of naphthyl cyanohydrins with allyl borates and allyl stannanes under palladium catalysis was developed. At the initial stage of this study, the dearomative reaction (C4 substitution of the aromatics) was competing with benzyl substitution. To circumvent this issue, the use of palladium and meta-disubstituted triarylphosphine as the catalyst in a 1:1 ratio was found to enhance the site selectivity, furnishing the desired dearomatized products. Further derivatizations of products were also successful.DOI:10.1021/acs.orglett.0c00897
文献信息
-
In Silico Driven Design and Synthesis of Rhodanine Derivatives as Novel Antibacterials Targeting the Enoyl Reductase InhA作者:Liudas Slepikas、Gianpaolo Chiriano、Remo Perozzo、Sébastien Tardy、Agata Kranjc、Ophélie Patthey-Vuadens、Hajer Ouertatani-Sakouhi、Sébastien Kicka、Christopher F. Harrison、Tiziana Scrignari、Karl Perron、Hubert Hilbi、Thierry Soldati、Pierre Cosson、Eduardas Tarasevicius、Leonardo ScapozzaDOI:10.1021/acs.jmedchem.5b01620日期:2016.12.22computational studies. Their antimicrobial activity was assessed against Mycobacterium marinum (Mm) (a model for Mtb), Pseudomonas aeruginosa (Pa), Legionella pneumophila (Lp), and Enterococcus faecalis (Ef) by using anti-infective, antivirulence, and antibiotic assays. Nineteen out of 34 compounds reduced Mm virulence at 10 μM. 33 exhibited promising antibiotic activity against Mm with a MIC of 0.21 μM and showed在这里,我们报告针对结核分枝杆菌结核(Mtb)反-2-烯酰基酰基载体蛋白还原酶(InhA)的4-噻唑烷酮(若丹宁)衍生物的设计,合成和生物学评估。在罗丹宁环第5位具有庞大芳族取代基且在N -3位具有色氨酸残基的化合物对InhA的活性最高,IC 50值为2.7至30μM。实验数据显示与计算研究一致的相关性。他们的抗菌活性评估了对结核分枝杆菌(Mm)(铜绿假单胞菌(Mtb)的模型)。Pa),嗜肺军团菌(Lp)和粪肠球菌(Ef),方法是使用抗感染,抗毒力和抗生素检测方法。34种化合物中有19种降低了10μM的Mm毒力。33的MIC为0.21μM,对Mm表现出有希望的抗生素活性,在30μM的抗感染试验中,Lp的生长降低了89%。32显示对Ef的高抗生素活性,MIC为0.57μM。
-
INHIBITORS OF HEMOPOIETIC CELL KINASE (P59-HCK) AND THEIR USE IN THE TREATMENT OF INFLUENZA INFECTION申请人:Charron Catherine Elisabeth公开号:US20120244120A1公开(公告)日:2012-09-27The present invention relates inter alia to the treatment or prevention of influenza virus infection (including subtypes influenza A virus, influenza B virus, avian strain H5N1, A/H1N1, H3N2 and/or pandemic influenza) using compounds which inhibit the activity of p59-HCK and to a method of screening for a candidate drug substance intended to prevent or treat influenza virus infection in a subject, said method comprising identifying a test substance capable of inhibiting p59-HCK activity.本发明涉及治疗或预防流感病毒感染(包括亚型流感A病毒、流感B病毒、禽流感H5N1、A/H1N1、H3N2和/或大流行性流感)的化合物,这些化合物抑制p59-HCK的活性,并涉及一种筛选候选药物物质的方法,该方法旨在预防或治疗受试者的流感病毒感染,所述方法包括识别能够抑制p59-HCK活性的试验物质。
-
Highly Regioselective Aromatic C–H Allylation of <i>N</i>-(Arylmethyl)sulfonimides with Allyl Grignard Reagents Involving Benzylic C–N Cleavage作者:Meng-Zeng Zhu、Dong Xie、Shi-Kai TianDOI:10.1021/acs.orglett.1c02437日期:2021.9.3C–H functionalization of benzyl electrophiles with nucleophiles via palladium-catalyzed benzylic C–N cleavage. A range of N-(1-naphthylmethyl)sulfonimides, N-(2-thienylmethyl)sulfonimides, and N-(2-furanylmethyl)sulfonimides smoothly underwent palladium-catalyzed aromatic C–H allylation with allyl Grignard reagents at room temperature, delivering structurally diverse substituted 1-allylnaphthalenes and
-
Controlled Photochemical Release of Nitric Oxide from <i>O</i><sup>2</sup>-Naphthylmethyl- and <i>O</i><sup>2</sup>-Naphthylallyl-Substituted Diazeniumdiolates作者:K. Mani Bushan、Hua Xu、Patrick H. Ruane、Raechelle A. D'Sa、Christopher M. Pavlos、Joseph A. Smith、Tevye C. Celius、John P. ToscanoDOI:10.1021/ja027957h日期:2002.10.1effect on the observed photochemistry, with the appropriate substitution pattern resulting in efficient diazeniumdiolate photorelease. Observed nitric oxide release rates from these photoprecursors are consistent with those expected for normal thermal dissociation of the diazeniumdiolate in aqueous solutions and show the same pH dependence.
-
Manganese(III)-Mediated Formylation of Aromatic Compounds in the Presence of Malonic Acid作者:Hiroshi Nishino、Katsunori Tsunoda、Kazu KurosawaDOI:10.1246/bcsj.62.545日期:1989.2the presence of manganese(III) acetate gives naphthalenecarbaldehydes and naphthalenecarboxylic acids. Similar reactions of anthracene, pyrene, and methoxybenzenes also yield formylated and carboxylated products. It was found that the formyl group introduced to the aromatic ring was not derived from carboxymethyl radical generated directly by the thermolysis of manganese(III) acetate, but from a dicarboxymethyl
表征谱图
-
氢谱1HNMR
-
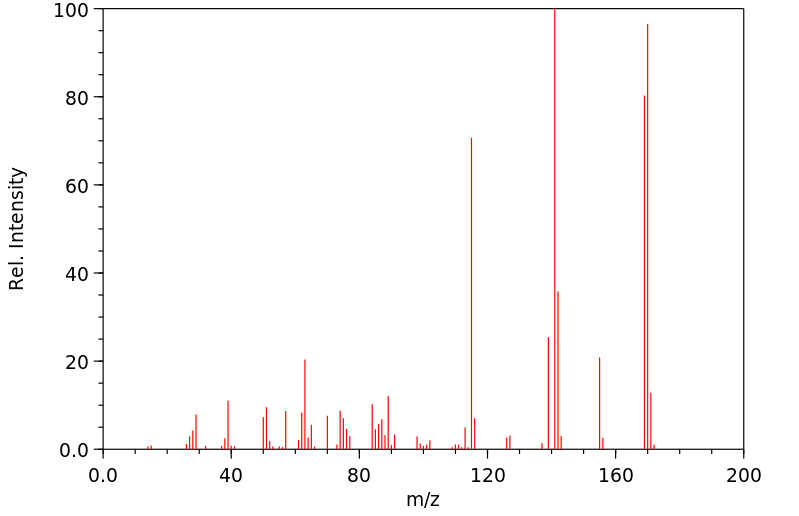
质谱MS
-
碳谱13CNMR
-
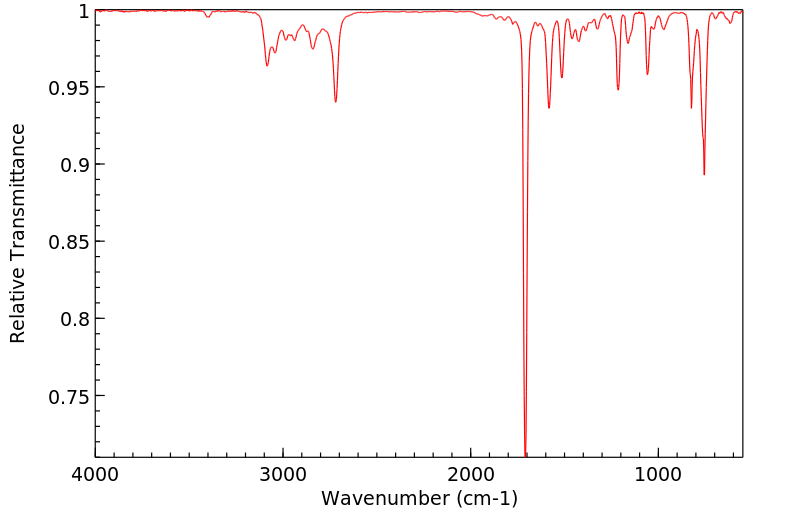
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-溴烯醇内酯
(R)-3,3''-双([[1,1''-联苯]-4-基)-[1,1''-联萘]-2,2''-二醇
(3S,3aR)-2-(3-氯-4-氰基苯基)-3-环戊基-3,3a,4,5-四氢-2H-苯并[g]吲唑-7-羧酸
(3R,3’’R,4S,4’’S,11bS,11’’bS)-(+)-4,4’’-二叔丁基-4,4’’,5,5’’-四氢-3,3’’-联-3H-二萘酚[2,1-c:1’’,2’’-e]膦(S)-BINAPINE
(11bS)-2,6-双(3,5-二甲基苯基)-4-羟基-4-氧化物-萘并[2,1-d:1'',2''-f][1,3,2]二氧磷
(11bS)-2,6-双(3,5-二氯苯基)-4羟基-4-氧-二萘并[2,1-d:1'',2''-f][1,3,2]二氧磷杂七环
(11bR)-2,6-双[3,5-双(1,1-二甲基乙基)苯基]-4-羟基-4-氧化物-二萘并[2,1-d:1'',2''-f][1,3,2]二氧杂磷平
黄胺酸
马兜铃对酮
马休黄钠盐一水合物
马休黄
食品黄6号
食品红40铝盐色淀
飞龙掌血香豆醌
颜料黄101
颜料红70
颜料红63
颜料红53:3
颜料红5
颜料红48单钠盐
颜料红48:2
颜料红4
颜料红261
颜料红258
颜料红220
颜料红22
颜料红214
颜料红2
颜料红19
颜料红185
颜料红184
颜料红170
颜料红148
颜料红147
颜料红146
颜料红119
颜料红114
颜料红 9
颜料红 21
颜料橙7
颜料橙46
颜料橙38
颜料橙3
颜料橙22
颜料橙2
颜料橙17
颜料橙 5
颜料棕1
顺式-阿托伐醌-d5
雄甾烷-3,17-二酮