2-甲氧基-6-甲基苯甲醛 | 54884-55-8
中文名称
2-甲氧基-6-甲基苯甲醛
中文别名
2-甲基-6-甲氧基苯甲醛
英文名称
2-methoxy-6-methyl-benzaldehyde
英文别名
2-Methoxy-6-methylbenzaldehyde
CAS
54884-55-8
化学式
C9H10O2
mdl
MFCD08235006
分子量
150.177
InChiKey
QLUHWZADTAIBKK-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:40-41℃
-
沸点:256℃
-
密度:1.062
-
闪点:113℃
计算性质
-
辛醇/水分配系数(LogP):1.7
-
重原子数:11
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.222
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
安全信息
-
海关编码:2912499000
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:温度:2-8°C,保持在惰性气体氛围中。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 2-Methoxy-6-methylbenzaldehyde
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2-Methoxy-6-methylbenzaldehyde
CAS number: 54884-55-8
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C9H10O2
Molecular weight: 150.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 2-Methoxy-6-methylbenzaldehyde
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2-Methoxy-6-methylbenzaldehyde
CAS number: 54884-55-8
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C9H10O2
Molecular weight: 150.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-羟基-6-甲基苯甲醛 2-hydroxy-6-methylbenzaldehyde 18362-36-2 C8H8O2 136.15 2,3-二甲基苯甲醚 2,3-dimethylanisole 2944-49-2 C9H12O 136.194 2-甲氧基-6-甲基苯甲酸 2-methoxy-6-methyl-benzoic acid 6161-65-5 C9H10O3 166.177 2-甲氧基-6-甲基苯甲酸甲酯 methyl 2-methyl-6-methoxybenzoate 79383-44-1 C10H12O3 180.203 —— (2-methoxy-6-methylphenyl)methanol 89244-39-3 C9H12O2 152.193 —— 3-(Bromomethyl)-2-methylanisole 70264-73-2 C9H11BrO 215.09 2-(溴甲基)-1-甲氧基-3-甲基苯 2-(Bromomethyl)-3-methylanisole 89244-40-6 C9H11BrO 215.09 2-甲氧基-6-甲基苯甲酸乙酯 ethyl 2-methoxy-6-methylbenzoate 6520-83-8 C11H14O3 194.23 二甲酚 2,3-Dimethylphenol 526-75-0 C8H10O 122.167 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-羟基-6-甲基苯甲醛 2-hydroxy-6-methylbenzaldehyde 18362-36-2 C8H8O2 136.15 2-甲氧基-6-甲基苯甲酸 2-methoxy-6-methyl-benzoic acid 6161-65-5 C9H10O3 166.177 3-溴-6-甲氧基-2-甲基苯甲醛 3-bromo-6-methoxy-2-methylbenzaldehyde 137644-93-0 C9H9BrO2 229.073 1-(2-甲氧基-6-甲基苯基)乙酮 2'-methoxy-6'-methylacetophenone 6161-64-4 C10H12O2 164.204 —— 4-((2-formyl-3-methylphenoxy)methyl)benzoic acid 1360618-29-6 C16H14O4 270.285 —— methyl 4-((2-formyl-3-methylphenoxy)methyl)benzoate 1360618-17-2 C17H16O4 284.312 2-甲氧基-6-甲基苯甲酸甲酯 methyl 2-methyl-6-methoxybenzoate 79383-44-1 C10H12O3 180.203 —— 2-bromo-1-(2-methoxy-6-methylphenyl)ethanone 126524-09-2 C10H11BrO2 243.1 —— 2-methoxy-6-methylbenzoyl cyanide 71687-95-1 C10H9NO2 175.187 —— coahuilensol, methyl ether 72473-09-7 C10H12O 148.205 —— (2-methoxy-6-methylphenyl)methanol 89244-39-3 C9H12O2 152.193 2-甲氧基-6-甲基苯甲腈 2-methoxy-6-methyl-benzonitrile 53005-44-0 C9H9NO 147.177 1-(2-羟基-6-甲基苯基)乙酮 2'-hydroxy-6'-methylacetophenone 41085-27-2 C9H10O2 150.177 3-溴-6-羟基-2-甲基苯甲醛 3-bromo-6-hydroxy-2-methylbenzaldehyde 137644-94-1 C8H7BrO2 215.046 —— 1-(2-methoxy-6-methylphenyl)-2-phenylethanone 1408089-50-8 C16H16O2 240.302 2-甲氧基-6-甲基苯甲醛肟 2-methoxy-6-methylbenzaldehyde oxime 1030446-72-0 C9H11NO2 165.192 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:NOVEL COMPOUNDS AS MODULATORS OF PROTEIN KINASES摘要:本发明提供了PI3K蛋白激酶调节剂,其制备方法,含有它们的药物组合物,以及使用它们进行治疗、预防和/或改善激酶介导的疾病或紊乱的方法。公开号:US20120289496A1
-
作为产物:描述:5-methoxybicyclo[4.2.0]octa-1,3,5-trien-7-ol 在 sodium methylate 作用下, 以 甲醇 为溶剂, 以99%的产率得到2-甲氧基-6-甲基苯甲醛参考文献:名称:芳炔[2+2]环加成和苯并环丁烯醇开环合成氧化邻甲基苯甲醛摘要:本文描述了从市售溴代芳烃开始制备氧化邻甲基苯甲醛衍生物的两步程序。该合成的特点是通过与乙醛烯醇锂的苯炔[2+2]环加成反应,将甲基和甲酰基同时且高度区域选择性地安装到苯核上,以高产率得到相应的苯并环丁烯醇。苯并环丁烯醇在碱性条件下在甲醇中进行键选择性开环,得到标题化合物。DOI:10.2533/chimia.2018.870
文献信息
-
Chemoselective Reduction of Sterically Demanding <i>N</i>,<i>N</i>-Diisopropylamides to Aldehydes作者:Peihong Xiao、Zhixing Tang、Kai Wang、Hua Chen、Qianyou Guo、Yang Chu、Lu Gao、Zhenlei SongDOI:10.1021/acs.joc.7b02868日期:2018.2.16EtOTf and LiAlH(OEt)3 in the absence of base were found to be optimal for reducing extremely sterically demanding 2,6-disubstituted N,N-diisopropylbenzamides. The reaction tolerates various reducible functional groups, including aldehyde and ketone. 1H NMR studies confirmed the formation of imidates stable in water. The synthetic usefulness of this methodology was demonstrated with N,N-diisopropylamide-directed已经开发出一种用于化学选择性地将空间上需要的N,N-二异丙基酰胺还原成醛的顺序一锅法。在该反应中,酰胺用EtOTf活化形成酰亚胺,然后用LiAlH(OR)3 [R = t- Bu,Et]还原,通过水解所得到的半缩醛产生醛。非亲核碱基2,6-DTBMP显着提高了反应效率。发现EtOTf / 2,6-DTBMP和LiAlH(O- t- Bu)3的组合对于还原烷基,烯基,炔基和2-单取代的芳基N,N-二异丙基酰胺是最佳的。相反,EtOTf和LiAlH(OEt)3在没有碱的情况下,发现其对于还原空间上要求极高的2,6-二取代的N,N-二异丙基苯甲酰胺是最佳的。该反应可耐受各种可还原的官能团,包括醛和酮。1 H NMR研究证实了在水中稳定形成的酰亚胺。这种方法的综合实用性通过N,N-二异丙基酰胺定向的邻位金属化和CH键的活化得到了证明。
-
Selective Electrochemical Oxygenation of Alkylarenes to Carbonyls作者:Xue Li、Fang Bai、Chaogan Liu、Xiaowei Ma、Chengzhi Gu、Bin DaiDOI:10.1021/acs.orglett.1c02651日期:2021.10.1An efficient electrochemical method for benzylic C(sp3)–H bond oxidation has been developed. A variety of methylarenes, methylheteroarenes, and benzylic (hetero)methylenes could be converted into the desired aryl aldehydes and aryl ketones in moderate to excellent yields in an undivided cell, using O2 as the oxygen source and lutidinium perchlorate as an electrolyte. On the basis of cyclic voltammetry
-
Copper-catalyzed (4+1) and (3+2) cyclizations of iodonium ylides with alkynes作者:Hao Liang、Xiaobo He、Yaqi Zhang、Bin Chen、Jia-sheng Ouyang、Yongsu Li、Bendu Pan、Chitreddy V. Subba Reddy、Wesley Ting Kwok Chan、Liqin QiuDOI:10.1039/d0cc04373g日期:——The copper(II)-catalyzed (4+1) cyclizations and copper(I)-catalyzed (3+2) cycloadditions of iodonium ylides and alkynes were successfully developed by employing efficient and safe iodonium ylides instead of traditional diazo compounds. Highly functionalized dimethyl (E)-3-benzylideneindoline-2,2-dicarboxylates and methyl 5-(2-hydroxyphenyl)-2-methoxy-4-phenylfuran-3-carboxylates were conveniently prepared
-
Synthesis of (+)-Varitriol Analogues via Novel and Versatile Building Blocks Based on Julia Olefination作者:Annamalai Senthilmurugan、Indrapal Singh AidhenDOI:10.1002/ejoc.200901012日期:2010.1The synthesis of (+)-varitriol (1) analogues was achieved through the use of Julia olefination. The potential anticancer properties of 1 coupled with our interest in developing building blocks that enable olefin formation under the Julia protocol constitute the basis of our research project. Efforts are aimed at the synthesis of building blocks 2 and 3 and to explore their use towards the synthesis
-
Benzofurane, benzothiophene, benzothiazol derivatives as FXR modulators申请人:Merck Sante公开号:EP2110374A1公开(公告)日:2009-10-21The present invention relates to compounds of formula (I) wherein the substituents are as defined in the claims, including pharmaceutical compositions thereof and for their use in the treatment and/or prevention and/or amelioration of one or more symptoms of disease or disorders related to the activity of FXR. The invention is also directed to intermediates and to a method of preparation of compounds of formula (I).本发明涉及式(I)的化合物,其中取代基如权利要求中定义的那样,包括其药物组合物及其用于治疗和/或预防及/或改善与FXR活性相关的疾病或障碍的一个或多个症状。本发明还涉及中间体以及化合物式(I)的制备方法。
表征谱图
-
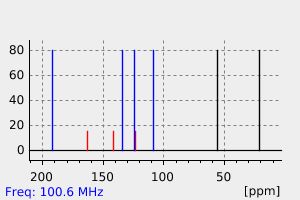
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫