4-二甲氨基苯甲酸甲酯 | 1202-25-1
中文名称
4-二甲氨基苯甲酸甲酯
中文别名
4-二甲基氨基苯甲酸甲酯
英文名称
methyl 4-(N,N-dimethylamino)benzoate
英文别名
methyl 4-(dimethylamino)benzoate;methyl p-(N,N-dimethylamino)benzoate;methyl p-(dimethylamino)benzoate;4-dimethylaminobenzoic acid methyl ester
CAS
1202-25-1
化学式
C10H13NO2
mdl
MFCD00014905
分子量
179.219
InChiKey
DBQGARDMYOMOOS-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:100-102°C
-
沸点:311.75°C (rough estimate)
-
密度:1.1248 (rough estimate)
-
稳定性/保质期:
如果按照规格使用和储存,则不会分解,未有已知危险反应。请避免接触氧化物。
计算性质
-
辛醇/水分配系数(LogP):2.4
-
重原子数:13
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:29.5
-
氢给体数:0
-
氢受体数:3
安全信息
-
安全说明:S22,S24/25
-
海关编码:2922499990
-
危险性防范说明:P261,P301+P312,P302+P352,P304+P340,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:请将贮藏器密封保存,并存放在阴凉干燥处。同时,确保工作环境具有良好的通风或排气设施。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: Methyl 4-(dimethylamino)benzoate
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: Methyl 4-(dimethylamino)benzoate
CAS number: 1202-25-1
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C10H13NO2
Molecular weight: 179.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: Methyl 4-(dimethylamino)benzoate
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: Methyl 4-(dimethylamino)benzoate
CAS number: 1202-25-1
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C10H13NO2
Molecular weight: 179.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-(甲氨基)苯甲酸甲酯 methyl 4-(N-methyl)aminobenzoate 18358-63-9 C9H11NO2 165.192 4-二甲氨基苯甲酸 p-N,N-dimethylaminobenzoic acid 619-84-1 C9H11NO2 165.192 4-氨基苯甲酸甲酯 4-methoxycarbonyl aniline 619-45-4 C8H9NO2 151.165 4-(二甲氨基)苄醇 N,N-dimethyl-4-hydroxymethylaniline 1703-46-4 C9H13NO 151.208 对二甲氨基苯甲醛 4-dimethylamino-benzaldehyde 100-10-7 C9H11NO 149.192 对硝基苯甲酸甲酯 4-nitrobenzoic acid methyl ester 619-50-1 C8H7NO4 181.148 4-二甲氨基苯甲酰氯 4-(dimethylamino)benzoyl chloride 4755-50-4 C9H10ClNO 183.637 —— 4-(dimethylamino)benzaldehyde-formyl-d1 42007-05-6 C9H11NO 150.184 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 4-(甲氨基)苯甲酸甲酯 methyl 4-(N-methyl)aminobenzoate 18358-63-9 C9H11NO2 165.192 4-二甲氨基苯甲酸乙酯 ethyl p-dimethyaminolbenzoate 10287-53-3 C11H15NO2 193.246 4-二甲氨基苯甲酸 p-N,N-dimethylaminobenzoic acid 619-84-1 C9H11NO2 165.192 —— methyl 4-((cyanomethyl)methylamino)benzoate 51934-23-7 C11H12N2O2 204.228 —— n-decyl 4-dimethylaminobenzoate 125628-88-8 C19H31NO2 305.461 —— N-(2-carbomethoxyphenyl)-N-methylacetamide 37619-13-9 C11H13NO3 207.229 N-甲基-N-亚硝基-4-氨基苯甲酸甲酯 methyl 4-(methyl(nitroso)amino)benzoate 18600-49-2 C9H10N2O3 194.19 对二甲氨基苯甲酸异辛酯 2-ethylhexyl 4-(dimethylamino)benzoate 21245-02-3 C17H27NO2 277.407 —— 4-dimethylamino-benzoic acid-(1-methyl-heptyl ester) —— C17H27NO2 277.407 4-(二甲氨基)苄醇 N,N-dimethyl-4-hydroxymethylaniline 1703-46-4 C9H13NO 151.208 对二甲氨基苯甲醛 4-dimethylamino-benzaldehyde 100-10-7 C9H11NO 149.192 —— 4-dimethylamino-benzoic acid-(2-piperidino-ethyl ester) —— C16H24N2O2 276.379 3-溴-4-(二甲基氨基)苯甲酸甲酯 methyl 3-bromo-4-(N,N-dimethylamino)benzoate 71695-21-1 C10H12BrNO2 258.115 3-氨基-4-二甲氨基苯甲酸 3-amino-4-dimethylaminobenzoic acid 78196-46-0 C9H12N2O2 180.206 —— methyl 4-(N,N-dimethylamino)benzenecarbothioate 182114-86-9 C10H13NOS 195.285 对硝基苯甲酸甲酯 4-nitrobenzoic acid methyl ester 619-50-1 C8H7NO4 181.148 —— methyl 4-(methyl(3-oxo-3-phenylpropyl)amino)benzoate 89787-34-8 C18H19NO3 297.354 4-(二甲基氨基)苯甲酰胺 4-(dimethylamino)benzamide 6083-47-2 C9H12N2O 164.207 N,N-二甲基对甲苯胺 Dimethyl-p-toluidine 99-97-8 C9H13N 135.209 —— Methyl 4-[bis(3-oxo-3-phenylpropyl)amino]benzoate 89787-37-1 C26H25NO4 415.489 —— Methyl 4-{methyl[(2-oxocyclohexyl)methyl]amino}benzoate 89787-26-8 C16H21NO3 275.348 对二甲氨基苯甲酰肼 4-(dimethylamino)benzohydrazide 19353-92-5 C9H13N3O 179.222 - 1
- 2
- 3
反应信息
-
作为反应物:描述:4-二甲氨基苯甲酸甲酯 在 二乙基硅烷 、 C15H27Br2CoN3 、 potassium tert-butylate 作用下, 以 甲苯 为溶剂, 反应 12.0h, 以39%的产率得到对二甲氨基苯甲醛参考文献:名称:钴催化从酯选择性合成醛和醇。摘要:据报道酯被有效和选择性地还原为醛和醇,其中简单的钴夹钳催化剂使用二乙基硅烷作为还原剂催化两种转化。显着地,反应选择性由二乙基硅烷的化学计量控制。DOI:10.1039/d0cc03076g
-
作为产物:描述:参考文献:名称:Bischoff,E., Chemische Berichte, 1889, vol. 22, p. 347摘要:DOI:
文献信息
-
SILANE COUPLING COMPOUNDS AND MEDICAL AND/OR DENTAL CURABLE COMPOSITIONS COMPRISING THE SAME申请人:KABUSHIKI KAISHA SHOFU公开号:US20190300552A1公开(公告)日:2019-10-03The present invention relate to a novel silane coupling agent and a medical and/or dental curable composition comprising the same. It is an object of the present invention to provide a novel silane coupling agent that imparts high affinity to a radical polymerizable monomer, thereby imparting high mechanical strength, flexibility and durability when used for a medical and/or dental curable composition, and an inorganic filler surface-treated with the novel silane coupling agent and a novel medical and/or dental curable composition. A silane coupling agent including repeating units such as a urethane bond and polyethylene glycol (ether bond) at a specific position is used.
-
Cleavage of Carboxylic Esters by Aluminum and Iodine作者:Dayong Sang、Huaxin Yue、Yang Fu、Juan TianDOI:10.1021/acs.joc.1c00034日期:2021.3.5A one-pot procedure for deprotecting carboxylic esters under nonhydrolytic conditions is described. Typical alkyl carboxylates are readily deblocked to the carboxylic acids by the action of aluminum powder and iodine in anhydrous acetonitrile. Cleavage of lactones affords the corresponding ω-iodoalkylcarboxylic acids. Aryl acetylates undergo deacetylation with the participation of the neighboring group
-
Silver-catalyzed intermolecular amination of fluoroarenes作者:Yu Wang、Chenlong Wei、Ruyun Tang、Haosheng Zhan、Jing Lin、Zhenhua Liu、Weihua Tao、Zhongxue FangDOI:10.1039/c8ob01749b日期:——A novel highly selective Ag-catalyzed intermolecular amination of fluoroarenes has been developed. This transformation starts from readily available 4-carbonyl fluorobenzene and NaN3 or other nitrogen-source, via amination followed by C–F bond cleavage, thus affording the desired 4-carbonyl arylamine products under mild conditions. The reaction is accelerated using a small amount of water. This pathway
-
A metal-free direct C (sp<sup>3</sup>)–H cyanation reaction with cyanobenziodoxolones作者:Ming-Xue Sun、Yao-Feng Wang、Bao-Hua Xu、Xin-Qi Ma、Suo-Jiang ZhangDOI:10.1039/c8ob00173a日期:——A metal-free protocol of direct C(sp3)–H cyanation with cyanobenziodoxolones functioning as both cyanating reagents and oxidants was developed. Unactivated substrates, such as alkanes, ethers and tertiary amines, were thereby transformed to the corresponding nitriles in moderate to high yields. Mechanistic studies indicated that the cyanation proceeded with two potential pathways, which is highly dependent
-
Electrochemical Activation of Diverse Conventional Photoredox Catalysts Induces Potent Photoreductant Activity**作者:Colleen P. Chernowsky、Alyah F. Chmiel、Zachary K. WickensDOI:10.1002/anie.202107169日期:2021.9.20Herein, we disclose that electrochemical stimulation induces new photocatalytic activity from a range of structurally diverse conventional photocatalysts. These studies uncover a new electron-primed photoredox catalyst capable of promoting the reductive cleavage of strong C(sp2)−N and C(sp2)−O bonds. We illustrate several examples of the synthetic utility of these deeply reducing but otherwise safe
表征谱图
-
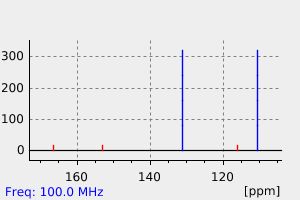
氢谱1HNMR
-
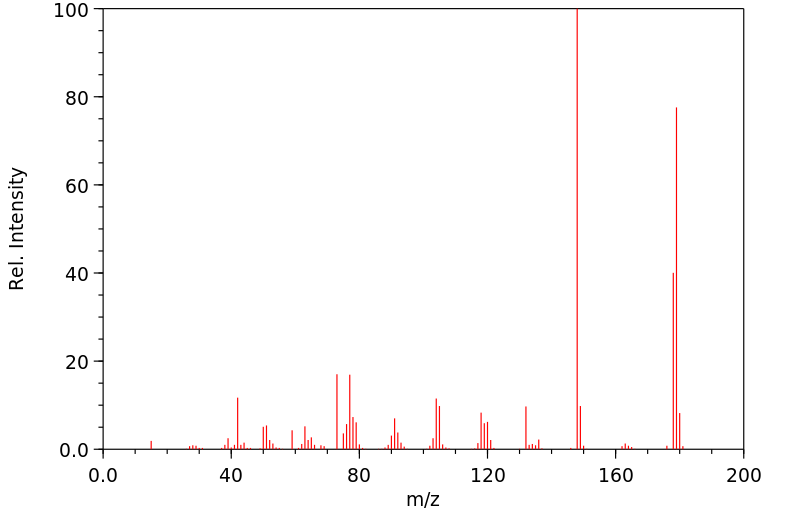
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫