N-(4-氯苯基)-2-氧代甘氨酸乙酯 | 5397-14-8
中文名称
N-(4-氯苯基)-2-氧代甘氨酸乙酯
中文别名
2-((4-氯苯基)氨基)-2-氧代乙酸乙酯
英文名称
ethyl 2-[(4-chlorophenyl)amino]-2-oxoacetate
英文别名
ethyl 4'-chlorooxanilate;ethyl N-(4-chlorophenyl)oxamate;N-(p-Chlor-phenyl)-oxaminsaeureaethylester;Ethyl [(4-chlorophenyl)amino](oxo)acetate;ethyl 2-(4-chloroanilino)-2-oxoacetate
CAS
5397-14-8
化学式
C10H10ClNO3
mdl
MFCD00000611
分子量
227.647
InChiKey
JFAOXNRPSKOLNC-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2.1
-
重原子数:15
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.2
-
拓扑面积:55.4
-
氢给体数:1
-
氢受体数:3
安全信息
-
海关编码:2924299090
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-(4-氯苯胺基)乙酸乙酯 ethyl 2-((4-chlorophenyl)amino)acetate 2521-89-3 C10H12ClNO2 213.664 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 (4-氯苯基)氨基](氧代)乙酸 2-((4-chlorophenyl)amino)-2-oxoacetic acid 17738-71-5 C8H6ClNO3 199.594 —— (4-chloro-phenyl)-oxalamide 17738-73-7 C8H7ClN2O2 198.609 —— NN'-(4,4'-dichlorodiphenyl)oxamide 6333-34-2 C14H10Cl2N2O2 309.152 —— N-p-Chlorphenyl-oxamidsaeurehydrazid 53117-27-4 C8H8ClN3O2 213.623
反应信息
-
作为反应物:描述:N-(4-氯苯基)-2-氧代甘氨酸乙酯 在 水 、 O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate 、 三乙胺 、 sodium hydroxide 作用下, 以 乙醇 为溶剂, 生成 N1-(4-chlorophenyl)-N2-((5-(2-hydroxyethyl)-4-methylthiazol-2-yl)(piperidin-2-yl)methyl)oxalamide formate参考文献:名称:Design, Synthesis, and Antiviral Activity of Entry Inhibitors That Target the CD4-Binding Site of HIV-1摘要:The CD4 binding site on HIV-1 gp120 has been validated as a drug target to prevent HIV-1 entry to cells. Previously, we identified two small molecule inhibitors consisting of a 2,2,6,6-tetramethylpiperidine ring linked by an oxalamide to a p-halide-substituted phenyl group, which target this site, specifically, a cavity termed "Phe43 cavity". Here we use synthetic chemistry, functional assessment, and structure-based analysis to explore variants of each region of these inhibitors for improved antiviral properties. Alterations of the phenyl group and of the oxalamide linker indicated that these regions were close to optimal in the original lead compounds. Design of a series of compounds, where the tetramethylpiperidine ring was replaced with new scaffolds, led to improved antiviral activity. These new scaffolds provide insight into the surface chemistry at the entrance of the cavity and offer additional opportunities by which to optimize further these potential-next-generation therapeutics and microbicides against HIV-1.DOI:10.1021/jm3002247
-
作为产物:描述:N-(p-Chlorphenyl)-aminofumarsaeureethylester 在 叔丁基过氧化氢 作用下, 以 水 、 氯苯 为溶剂, 反应 2.0h, 以80%的产率得到N-(4-氯苯基)-2-氧代甘氨酸乙酯参考文献:名称:叔丁基过氧化氢促进缺电子烯胺的无金属氧化 C=C 键裂解摘要:描述了一种新型的叔丁基氢过氧化物 (TBHP) 促进的烯胺氧化 C=C 双键裂解。在 TBHP 存在下,将缺电子烯胺的氯苯溶液在 80°C 下加热两小时,导致 C=C 键断裂。本研究为利用 TBHP 形成 C=O 双键提供了一种新策略。DOI:10.1055/s-0036-1588990
文献信息
-
Diamine derivatives申请人:Ohta Toshiharu公开号:US20050020645A1公开(公告)日:2005-01-27A compound represented by the general formula (1): Q 1 -Q 2 -T 0 -N(R 1 )-Q 3 -N(R 2 )-T 1 -Q 4 (1) wherein R 1 and R 2 are hydrogen atoms or the like; Q 1 is a saturated or unsaturated, 5- or 6-membered cyclic hydrocarbon group which may be substituted, or the like; Q 2 is a single bond or the like; Q 3 is a group in which Q 5 is an alkylene group having 1 to 8 carbon atoms, or the like; and T 0 and T 1 are carbonyl groups or the like; a salt thereof, a solvate thereof, or an N-oxide thereof. The compound is useful as an agent for preventing and/or treating cerebral infarction, cerebral embolism, myocardial infarction, angina pectoris, pulmonary infarction, pulmonary embolism, Buerger's disease, deep venous thrombosis, disseminated intravascular coagulation syndrome, thrombus formation after valve or joint replacement, thrombus formation and reocclusion after angioplasty, systemic inflammatory response syndrome (SIRS), multiple organ dysfunction syndrome (MODS), thrombus formation during extracorporeal circulation, or blood clotting upon blood drawing.通用式(1)表示的化合物: Q1-Q2-T0-N(R1)-Q3-N(R2)-T1-Q4(1) 其中R1和R2是氢原子或类似物;Q1是饱和或不饱和的、5-或6-成员环烃基,可以被取代,或类似物;Q2是单键或类似物;Q3是一个基团,其中Q5是具有1至8个碳原子的烷基基团,或类似物;T0和T1是羰基团或类似物;其盐、溶剂合物或N-氧化物。 该化合物可用作预防和/或治疗脑梗死、脑栓塞、心肌梗死、心绞痛、肺梗死、肺栓塞、布尔格病、深静脉血栓形成、弥散性血管内凝血综合征、瓣膜或关节置换后的血栓形成、血管成形术后的血栓形成和再闭塞、全身性炎症反应综合征(SIRS)、多器官功能障碍综合征(MODS)、体外循环期间的血栓形成,或抽血时的血液凝结。
-
Design, synthesis and SAR of a novel series of heterocyclic phenylpropanoic acids as GPR120 agonists作者:Xuqing Zhang、Chaozhong Cai、Michael Winters、Michele Wells、Mark Wall、James Lanter、Zhihua Sui、Jingyuan Ma、Aaron Novack、Imad Nashashibi、Yuanping Wang、Wen Yan、Arthur Suckow、Hong Hua、Austin Bell、Peter Haug、Wilma Clapper、Celia Jenkinson、Joseph Gunnet、James Leonard、William V. MurrayDOI:10.1016/j.bmcl.2017.06.028日期:2017.8A novel series of 5-membered heterocycle-containing phenylpropanoic acid derivatives was discovered as potent GPR120 agonists with low clearance, high oral bioavailability and in vivo antidiabetic activity in rodents.
-
New Highlights in the Synthesis of 4-Aryl-1,4-dihydropyrazines作者:Jing-Yu He、Xiu-Qing Song、Hong Yan、Ru-Gang ZhongDOI:10.1002/jhet.989日期:2012.11The 4‐aryl‐1,4‐dihydropyrazines were prepared via the cyclization of N,N‐bisalkylated anilines with ammonium acetate. These reactions were aided by improvements in the synthesis of N,N‐bisalkylated anilines which were alkylated with anilines using ethyl 2‐diazo acetoacetate in a reaction catalyzed by rhodium acetate in the absence of oxygen. A possible mechanistic route is postulated on the basis of
-
Discovery of Cytochrome P450 4F11 Activated Inhibitors of Stearoyl Coenzyme A Desaturase作者:Sarah E. Winterton、Emanuela Capota、Xiaoyu Wang、Hong Chen、Prema L. Mallipeddi、Noelle S. Williams、Bruce A. Posner、Deepak Nijhawan、Joseph M. ReadyDOI:10.1021/acs.jmedchem.8b00052日期:2018.6.28Stearoyl-CoA desaturase (SCD) catalyzes the first step in the conversion of saturated fatty acids to unsaturated fatty acids. Unsaturated fatty acids are required for membrane integrity and for cell proliferation. For these reasons, inhibitors of SCD represent potential treatments for cancer. However, systemically active SCD inhibitors result in skin toxicity, which presents an obstacle to their development
-
Visible light-mediated photocatalytic oxidative cleavage of activated alkynes <i>via</i> hydroamination: a direct approach to oxamates作者:Narenderreddy Katta、Mamata Ojha、Arumugavel Murugan、Sagar Arepally、Duddu S. SharadaDOI:10.1039/c9ra10555g日期:——The direct oxidative cleavage of activated alkynes via hydroamination has been described using organic photocatalyst under visible-light irradiation at room temperature. In this reaction, the single electron oxidation of an in situ formed enamine followed by radical coupling with an oxidant finally delivers the oxamate. The key features of this photocatalytic reaction are the mild reaction conditions
表征谱图
-
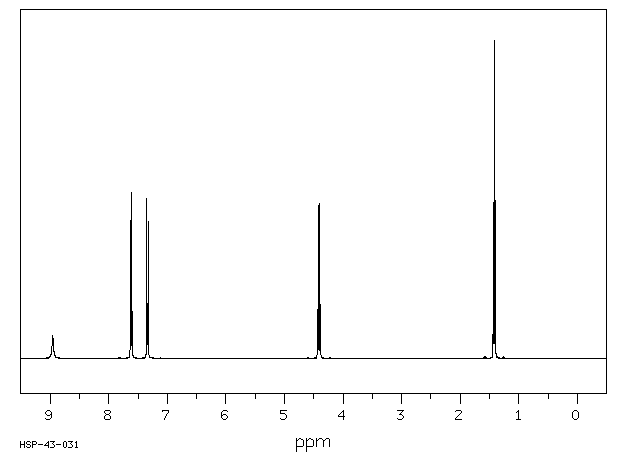
氢谱1HNMR
-
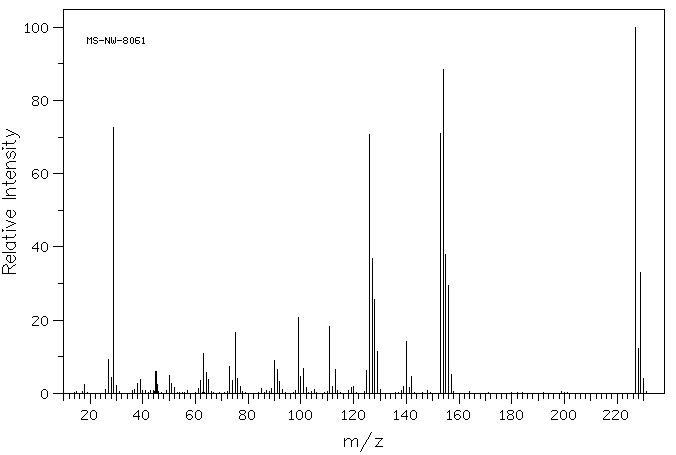
质谱MS
-
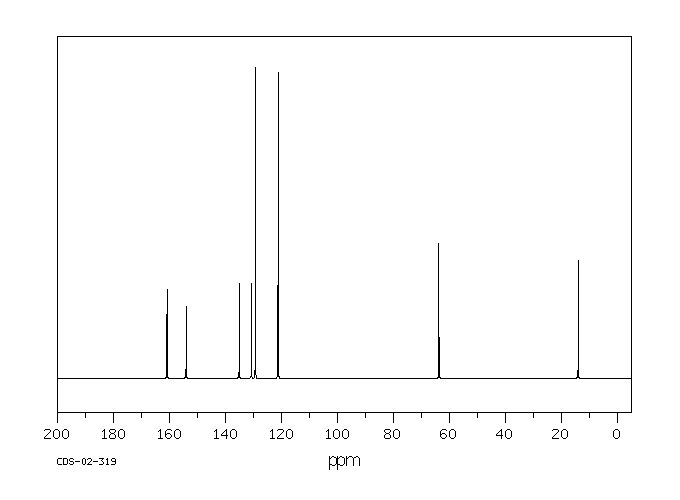
碳谱13CNMR
-
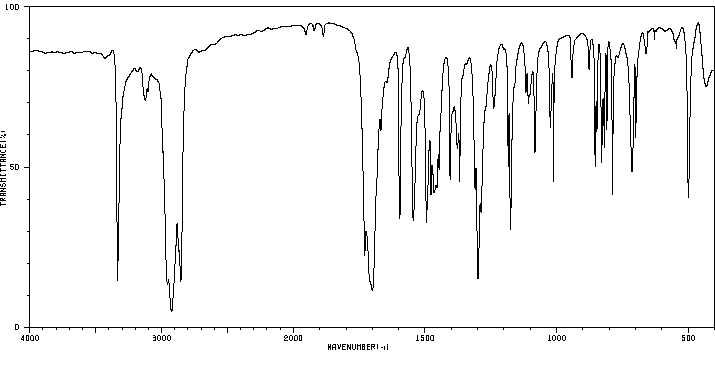
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(甲基3-(二甲基氨基)-2-苯基-2H-azirene-2-羧酸乙酯)
(±)-盐酸氯吡格雷
(±)-丙酰肉碱氯化物
(d(CH2)51,Tyr(Me)2,Arg8)-血管加压素
(S)-(+)-α-氨基-4-羧基-2-甲基苯乙酸
(S)-阿拉考特盐酸盐
(S)-赖诺普利-d5钠
(S)-2-氨基-5-氧代己酸,氢溴酸盐
(S)-2-[[[(1R,2R)-2-[[[3,5-双(叔丁基)-2-羟基苯基]亚甲基]氨基]环己基]硫脲基]-N-苄基-N,3,3-三甲基丁酰胺
(S)-2-[3-[(1R,2R)-2-(二丙基氨基)环己基]硫脲基]-N-异丙基-3,3-二甲基丁酰胺
(S)-1-(4-氨基氧基乙酰胺基苄基)乙二胺四乙酸
(S)-1-[N-[3-苯基-1-[(苯基甲氧基)羰基]丙基]-L-丙氨酰基]-L-脯氨酸
(R)-乙基N-甲酰基-N-(1-苯乙基)甘氨酸
(R)-丙酰肉碱-d3氯化物
(R)-4-N-Cbz-哌嗪-2-甲酸甲酯
(R)-3-氨基-2-苄基丙酸盐酸盐
(R)-1-(3-溴-2-甲基-1-氧丙基)-L-脯氨酸
(N-[(苄氧基)羰基]丙氨酰-N〜5〜-(diaminomethylidene)鸟氨酸)
(6-氯-2-吲哚基甲基)乙酰氨基丙二酸二乙酯
(4R)-N-亚硝基噻唑烷-4-羧酸
(3R)-1-噻-4-氮杂螺[4.4]壬烷-3-羧酸
(3-硝基-1H-1,2,4-三唑-1-基)乙酸乙酯
(2S,4R)-Boc-4-环己基-吡咯烷-2-羧酸
(2S,3S,5S)-2-氨基-3-羟基-1,6-二苯己烷-5-N-氨基甲酰基-L-缬氨酸
(2S,3S)-3-((S)-1-((1-(4-氟苯基)-1H-1,2,3-三唑-4-基)-甲基氨基)-1-氧-3-(噻唑-4-基)丙-2-基氨基甲酰基)-环氧乙烷-2-羧酸
(2S)-2,6-二氨基-N-[4-(5-氟-1,3-苯并噻唑-2-基)-2-甲基苯基]己酰胺二盐酸盐
(2S)-2-氨基-N,3,3-三甲基-N-(苯甲基)丁酰胺
(2S)-2-氨基-3-甲基-N-2-吡啶基丁酰胺
(2S)-2-氨基-3,3-二甲基-N-(苯基甲基)丁酰胺,
(2S)-2-氨基-3,3-二甲基-N-2-吡啶基丁酰胺
(2S,4R)-1-((S)-2-氨基-3,3-二甲基丁酰基)-4-羟基-N-(4-(4-甲基噻唑-5-基)苄基)吡咯烷-2-甲酰胺盐酸盐
(2R,3'S)苯那普利叔丁基酯d5
(2R)-2-氨基-3,3-二甲基-N-(苯甲基)丁酰胺
(2-氯丙烯基)草酰氯
(1S,3S,5S)-2-Boc-2-氮杂双环[3.1.0]己烷-3-羧酸
(1R,5R,6R)-5-(1-乙基丙氧基)-7-氧杂双环[4.1.0]庚-3-烯-3-羧酸乙基酯
(1R,4R,5S,6R)-4-氨基-2-氧杂双环[3.1.0]己烷-4,6-二羧酸
齐特巴坦
齐德巴坦钠盐
齐墩果-12-烯-28-酸,2,3-二羟基-,苯基甲基酯,(2a,3a)-
齐墩果-12-烯-28-酸,2,3-二羟基-,羧基甲基酯,(2a,3b)-(9CI)
黄酮-8-乙酸二甲氨基乙基酯
黄荧菌素
黄体生成激素释放激素(1-6)
黄体生成激素释放激素 (1-5) 酰肼
黄体瑞林
麦醇溶蛋白
麦角硫因
麦芽聚糖六乙酸酯
麦根酸