5-氨磺酰基-2-甲氧基苯甲酸 | 22117-85-7
中文名称
5-氨磺酰基-2-甲氧基苯甲酸
中文别名
2-甲氧基-5-氨磺酰基苯甲酸;2-甲氧基-5-氨磺酰苯甲酸;2-甲氧基-5-磺酰苯甲酸;2-甲氧基-5-磺酰胺基苯甲酸
英文名称
2-Methoxy-5-sulfamoylbenzoic acid
英文别名
5-sulfamoyl-o-anisic acid
CAS
22117-85-7
化学式
C8H9NO5S
mdl
MFCD00129997
分子量
231.229
InChiKey
SQAILWDRVDGLGY-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:220 °C
-
沸点:482.5±55.0 °C(Predicted)
-
密度:1.492±0.06 g/cm3(Predicted)
-
溶解度:可溶于DMSO(少许)、甲醇(少许)
-
稳定性/保质期:
如果按照规格使用和储存,则不会分解。请避免接触氧化物。
计算性质
-
辛醇/水分配系数(LogP):-0.3
-
重原子数:15
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.125
-
拓扑面积:115
-
氢给体数:2
-
氢受体数:6
安全信息
-
危险品标志:Xi
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2935009090
-
危险性防范说明:P280,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:保持贮藏器密封,并将其放入一个紧密封装的容器中。储存时,请确保存放在阴凉、干燥的地方。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
2-Methoxy-5-sulfamoylbenzoic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
2-Methoxy-5-sulfamoylbenzoic acid
Ingredient name:
CAS number: 22117-85-7
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C8H9NO5S
Molecular weight: 231.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
2-Methoxy-5-sulfamoylbenzoic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
2-Methoxy-5-sulfamoylbenzoic acid
Ingredient name:
CAS number: 22117-85-7
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C8H9NO5S
Molecular weight: 231.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
5-氨基磺酰基-2-甲氧基苯甲酸
5-氨磺酰基-2-甲氧基苯甲酸又被称为2-甲氧基-5-氨磺酰基苯甲酸,是舒必利的重要中间体。舒必利属于抗精神病药物。
制备方法 原料及配比- 苯酰:1千克
- 25% 氨水:1.7千克
- 将按比例计算的25%氨水加入氨化反应釜中。
- 打开冷却水进出阀,并在搅拌下缓缓加入苯酰,确保温度保持在20℃以下。加完后立即盖好投料口以防止氨气溢出。
- 关闭进出冷却水阀门,缓慢打开进出蒸汽阀门,使釜内温度维持在70-75℃约1.5小时,反应结束后通过吹拂法排出氨,并回收利用。
- 之后关闭蒸汽进出阀,重新开启冷却水进出阀,将釜内温度降至30±2℃。
- 打开真空泵并注入盐酸,向反应液中加入适量的盐酸以调节pH值至1.0-1.5,并控制加酸速度确保温度不超过50℃。酸析完成后提升温度至98±2℃并保温15分钟,然后继续搅拌使温度降至常温。
- 停止搅拌,关闭冷却水进出阀,在静置过程中(可微开进出冷却水阀门以保持温度在25~35℃)静置2小时。
- 静置结束后进行离心分离,滤饼用水冲洗至pH值为3-4后打碎放入烘箱中,在83-87℃下干燥22小时。最终收率为65-75%。
主要用于有机合成中间体以及作为舒必利的合成原料。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-甲氧基-5-磺酰胺苯甲酸甲酯 2-methoxy-5-sulfamoylbenzoic acid methyl ester 33045-52-2 C9H11NO5S 245.256 5-氯磺酰-2-甲氧基苯甲酸 5-chlorosulfonyl-2-anisic acid 51904-91-7 C8H7ClO5S 250.66 邻甲氧基苯甲酸 2-Methoxybenzoic acid 579-75-9 C8H8O3 152.15 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-甲氧基-5-磺酰胺苯甲酸甲酯 2-methoxy-5-sulfamoylbenzoic acid methyl ester 33045-52-2 C9H11NO5S 245.256 2-羟基-5-氨基磺酰基苯甲酸 2-hydroxy-5-sulfonamidobenzoic acid 5378-41-6 C7H7NO5S 217.202 2-甲氧基-5-氨磺酰基苯甲酸乙酯 2-methoxy-5-sulfamoylbenzoic acid ethyl ester 33045-53-3 C10H13NO5S 259.283 5-(氨基磺酰基)-2-甲氧基苯甲酰氯 2-methoxy-5-sulfamoyl-benzoyl chloride 52542-44-6 C8H8ClNO4S 249.675 —— N-benzyl-5-(aminosulfonyl)-2-methoxybenzamide 113681-63-3 C15H16N2O4S 320.369 —— 3-cyano-4-methoxybenzenesulfonamide 22117-84-6 C8H8N2O3S 212.229 —— N-[1-(dimethylamino)propan-2-yl]-2-methoxy-5-sulfamoylbenzamide 102535-20-6 C13H21N3O4S 315.393 5-(氨基磺酰基)-N-(2,5-二氯戊基)-2-甲氧基苯甲酰胺 N-(2,5-dichloropentyl)-2-methoxy-5-sulfamoylbenzamide 67833-50-5 C13H18Cl2N2O4S 369.269 —— N-[4-[2-(dimethylamino)ethoxy]benzyl]-2-methoxy-5-sulfamoylbenzamide 122892-35-7 C19H25N3O5S 407.491 —— 2-methoxy-5-sulphamoyl-benzoylpiperazine 103054-37-1 C12H17N3O4S 299.351 —— 4-Methoxy-3-(4-methylpiperazine-1-carbonyl)benzenesulfonamide —— C13H19N3O4S 313.37 —— 5-(aminosulfonyl)-2-methoxybenzoic acid 4-benzylpiperazide 57479-93-3 C19H23N3O4S 389.475 —— N-<(2-tetrahydrothienyl)methyl>-2-methoxy-5-sulfamoylbenzamide 118894-84-1 C13H18N2O4S2 330.429 3-(4-异丁基-哌嗪-1-羰基)-4-甲氧基-苯磺酰胺 4-Methoxy-3-[4-(2-methylpropyl)piperazine-1-carbonyl]benzenesulfonamide 102535-28-4 C16H25N3O4S 355.458 —— 4-Methoxy-3-[4-[(4-methoxyphenyl)methyl]piperazine-1-carbonyl]benzenesulfonamide 102535-34-2 C20H25N3O5S 419.502 3-(4-烯丙基-哌嗪-1-羰基)-4-甲氧基-苯磺酰胺 4-Methoxy-3-(4-prop-2-enylpiperazine-1-carbonyl)benzenesulfonamide 102535-27-3 C15H21N3O4S 339.415 —— 4-Methoxy-3-[4-(3-methoxy-benzyl)-piperazine-1-carbonyl]-benzenesulfonamide 102535-35-3 C20H25N3O5S 419.502 舒必利 (-)sulpiride 15676-16-1 C15H23N3O4S 341.431 —— (S)-N-<(1-n-butyl-2-pyrrolidinyl)methyl>-2-methoxy-5-sulfamoylbenzamide 130904-92-6 C17H27N3O4S 369.485 —— (R)-N-<(1-n-butyl-2-pyrrolidinyl)methyl>-2-methoxy-5-sulfamoylbenzamide 130904-93-7 C17H27N3O4S 369.485 —— 2-[4-(2-methoxy-5-sulfamoylbenzoyl)piperazin-1-yl]-N-propan-2-ylacetamide 102535-33-1 C17H26N4O5S 398.483 —— N-(1-Ethyl-2-piperidyl)-2-methoxy-5-sulfamoylbenzamid 51218-14-5 C15H23N3O4S 341.431 —— 3-(4-Cyclohexylmethyl-piperazine-1-carbonyl)-4-methoxy-benzenesulfonamide 102535-29-5 C19H29N3O4S 395.523 —— N-[(4-benzylmorpholin-2-yl)methyl]-2-methoxy-5-sulfamoylbenzamide 112887-21-5 C20H25N3O5S 419.502 —— N-[3,5-bis(trifluoromethyl)phenyl]-2-methoxy-5-sulfamoyl-benzamide 439146-22-2 C16H12F6N2O4S 442.339 - 1
- 2
- 3
反应信息
-
作为反应物:描述:参考文献:名称:Treatment of chronic inflammatory joint disease with arylsulfonamides摘要:公开了一种使用以下结构的芳基磺酰胺来治疗慢性炎性关节疾病的方法:Z--SO.sub.2 NR.sup.1 R.sup.2其中R.sup.1和R.sup.2从氢、低烷基、低烯基、环烷基、苯基、低烷基苯基、2或3吡咯基、2或3-(N-低烷基吡咯基中选择,或者R.sup.1和R.sup.2一起可以形成吡咯基或哌啶基杂环氨基基团,Z是从取代或未取代的四唑基、1,3,4-噻二唑基、1,2,4-三唑基、苯并噻唑基、苯并咪唑基、咪唑基、吡啶基、4,6-二甲基嘧啶基、苯或萘中选择的芳基。公开号:US04990523A1
-
作为产物:描述:参考文献:名称:Synthesis and D2 dopaminergic activity of pyrrolidinium, tetrahydrothiophenium, and tetrahydrothiophene analogs of sulpiride摘要:All of the existing dopamine receptor models recognize the amine nitrogen of agonist and antagonist drugs as playing a crucial role in receptor interactions. However, there has been some controversy as to which molecular form of the amine, charged or uncharged, is most important in these interactions. We have synthesized and examined the biological activity of permanently charged and permanently uncharged analogues of the dopaminergic antagonist, sulpiride. Sulpiride and the permanently charged pyrrolidinium (6,7) and tetrahydrothiophenium (9) analogues were able to antagonize the inhibitory effect of apomorphine on the K+-induced release of [3H]acetylcholine from striatal slices. In contrast, the permanently uncharged tetrahydrothiophene analogue 8 was inactive at concentrations up to 100 microM. Additionally, both sulpiride and the tetrahydrothiophenium analogue were able to displace [3H]spiperone from D2 binding sites, while the tetrahydrothiophene analogue was unable to produce any significant displacement. These results are consistent with our previous observations on permanently charged chlorpromazine analogues and provide further evidence that dopaminergic antagonists bind in their charged molecular forms to anionic sites on the D2 receptor.DOI:10.1021/jm00124a024
文献信息
-
Macrocyclic compounds as inhibitors of viral replication申请人:Blatt M. Lawrence公开号:US20050267018A1公开(公告)日:2005-12-01The embodiments provide compounds of the general formulas I-XIX, as well as compositions, including pharmaceutical compositions, comprising a subject compound. The embodiments further provide treatment methods, including methods of treating flaviviral infection, including hepatitis C virus infection and methods of treating liver fibrosis, the methods generally involving administering to an individual in need thereof an effective amount of a subject compound or composition.该实施例提供了一般式I-XIX的化合物,以及包括药物组合在内的组合物,其中包括一种主体化合物。该实施例还提供了治疗方法,包括治疗黄病毒感染的方法,包括丙型肝炎病毒感染的方法和治疗肝纤维化的方法,这些方法通常涉及向需要的个体施用一种主体化合物或组合物的有效量。
-
Novel benzamides as selective and potent gastric prokinetic agents. 1. Synthesis and structure-activity relationships of N-[(2-morpholinyl)alkyl]benzamides作者:Shiro Kato、Toshiya Morie、Katsuhiko Hino、Tatsuya Kon、Shunsuke Naruto、Naoyuki Yoshida、Tadahiko Karasawa、Junichi MatsumotoDOI:10.1021/jm00167a020日期:1990.5With the purpose of obtaining more potent and selective gastric prokinetic than metoclopramide (1), a new series of N-[(2-morpholinyl)alkyl]benzamides (17-52) were synthesized and their gastric prokinetic activity was evaluated by determining effects on the gastric emptying of phenol red semisolid meal and of resin pellets solid meal in rats and mice. The morpholinyl moiety was newly designed after
-
Benzoyl-piperazine derivatives申请人:Jolidon J. Synese公开号:US20050209241A1公开(公告)日:2005-09-22The invention relates to compounds of formula wherein the substituents are described herein. The compounds may be used in the treatment of illnesses based on the glycine uptake inhibitor, such as psychoses, pain, neurodegenerative disfunction in memory and learning, schizophrenia, dementia and other diseases in which cognitive processes are impaired, such as attention deficit disorders or Alzheimer's disease.本发明涉及具有以下通式的化合物 其中取代基如本文所述。这些化合物可用于治疗基于抑制甘氨酸摄取的疾病,如精神疾病、疼痛、记忆和学习方面的神经退行性功能障碍、精神分裂症、痴呆症以及认知过程受损的其他疾病,例如注意力缺陷障碍或阿尔茨海默病。
-
Sulfonic acid esters申请人:Fujisawa Pharmaceutical Co., Ltd.公开号:US04242507A1公开(公告)日:1980-12-30A novel process for the amidation or esterification which comprises reacting a compound having a carboxy group with a compound having an amino or imino group which can be acylated or with a compound having a hydroxy group in the presence of a sulfonic acid ester of the formula: R.sub.1 --SO.sub.2 --OR.sub.2 wherein R.sub.1 is an organic group and R.sub.2 O-- is a residue of a strongly acidic N-hydroxy compound as a condensation agent, and a novel sulfonic acid ester useful as such a condensation agent and a process for the preparation thereof.
-
Discovery of N-substituted sulfamoylbenzamide derivatives as novel inhibitors of STAT3 signaling pathway based on Niclosamide作者:Xuebao Wang、Kaiqi Wu、Longcheng Fang、Xiaojiao Yang、Nan Zheng、Zongxuan Du、Ying Lu、Zixin Xie、Zhiguo Liu、Zhigui Zuo、Faqing YeDOI:10.1016/j.ejmech.2021.113362日期:2021.6Signal transducer and activator of transcription 3 (STAT3) has been confirmed as an attractive therapeutic target for cancer therapy. Herein, we designed and synthesized a series of N-substituted Sulfamoylbenzamide STAT3 inhibitors based on small-molecule STAT3 inhibitor Niclosamide. Compound B12, the best active compound of this series, was identified as an inhibitor of IL-6/STAT3 signaling with an信号转导和转录激活因子 3 (STAT3) 已被证实是一种有吸引力的癌症治疗靶点。在此,我们在小分子 STAT3 抑制剂氯硝柳胺的基础上,设计并合成了一系列 N 取代磺氨酰苯甲酰胺 STAT3 抑制剂。化合物 B12 是该系列中活性最好的化合物,被鉴定为 IL-6/STAT3 信号传导的抑制剂,IC 50在 STAT3 过表达的 MDA-MB-231、HCT-116 和 SW480 肿瘤细胞系中 0.61–1.11 μM,通过抑制 Tyr705 残基的 STAT3 磷酸化和 STAT3 下游基因的表达,诱导细胞凋亡和抑制癌细胞迁移. 此外,体内研究表明,化合物 B12 在 30 mg/kg (ig) 的剂量下抑制了裸鼠中 MDA-MB-231 异种移植肿瘤的生长,比阳性对照氯硝柳胺具有更好的抗肿瘤活性。更重要的是,B12 是一种口服生物可利用的抗癌剂,是进一步开发的有希望的候选者。
表征谱图
-
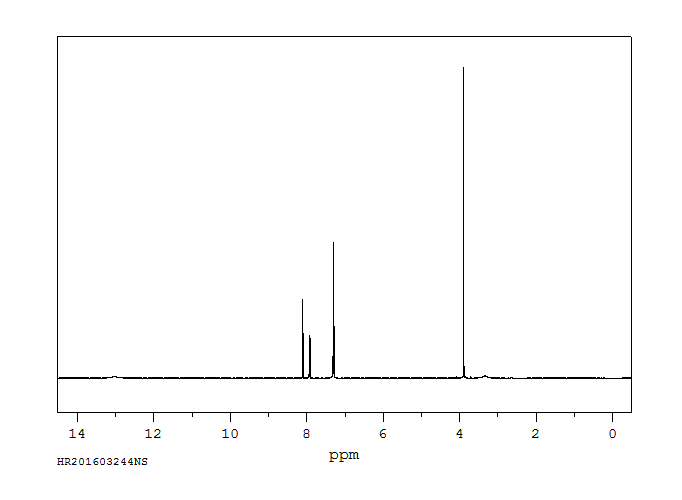
氢谱1HNMR
-
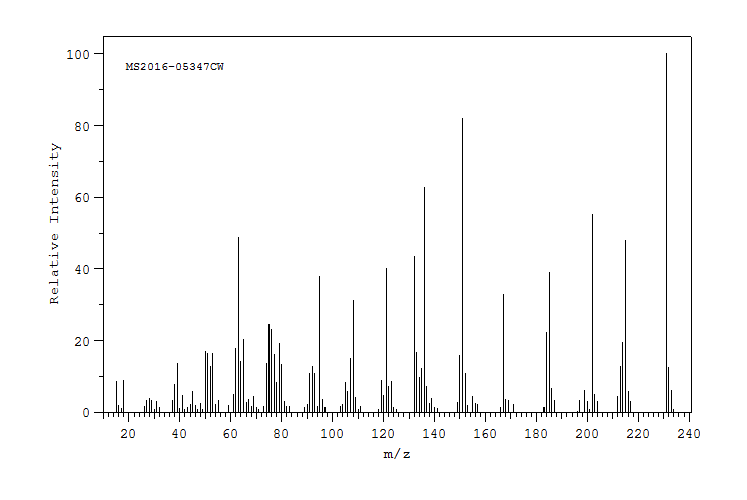
质谱MS
-
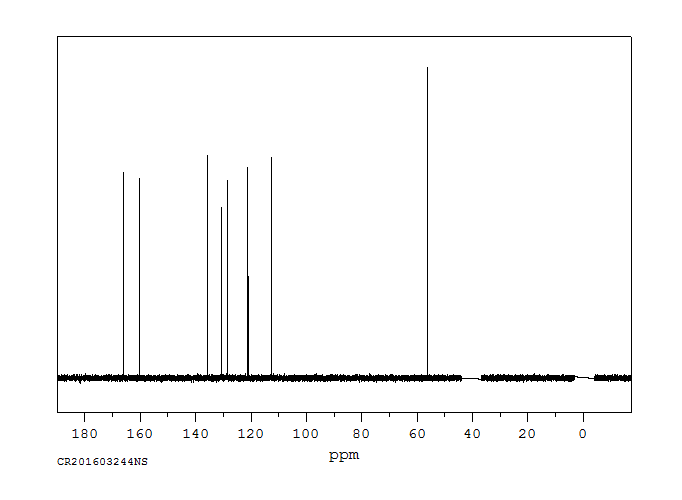
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫