庚醛二甲基缩醛 | 10032-05-0
中文名称
庚醛二甲基缩醛
中文别名
庚醛二甲缩醛;1,1-二甲氧基庚烷;庚醛缩二甲醇
英文名称
1,1-dimethoxyheptane
英文别名
heptanal dimethyl acetal
CAS
10032-05-0
化学式
C9H20O2
mdl
MFCD00014908
分子量
160.257
InChiKey
BBMCNYFBAIUERL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:183 °C760 mm Hg(lit.)
-
密度:0.832 g/mL at 25 °C(lit.)
-
闪点:136 °F
-
LogP:2.74
-
物理描述:colourless oily liquid with a green, herbaceous, somewhat nutty odour
-
溶解度:insoluble in water; soluble in organic solvents, oils
-
折光率:1.405-1.415
-
保留指数:1069;1063
-
稳定性/保质期:
性质为无色液体,具有类似核桃的香气。
计算性质
-
辛醇/水分配系数(LogP):3
-
重原子数:11
-
可旋转键数:7
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:18.5
-
氢给体数:0
-
氢受体数:2
安全信息
-
TSCA:Yes
-
危险等级:3
-
危险品标志:Xn
-
安全说明:S23,S24/25
-
危险类别码:R22
-
WGK Germany:2
-
危险品运输编号:UN 1993 3
-
海关编码:2911000000
-
包装等级:III
-
危险类别:3
SDS
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-羟基辛醛二甲基缩醛 2-hydroxyoctanal dimethyl acetal 65644-37-3 C10H22O3 190.283 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2-Brom-oenanthaldehyd-dimethylacetal 18207-18-6 C9H19BrO2 239.153
反应信息
-
作为反应物:描述:参考文献:名称:Magtrieve™:一种在中性条件下用于乙缩醛和缩酮的脱保护/氧化的新试剂摘要:Magtrieve TM可用于缩醛和缩酮的脱保护或在中性条件下直接氧化为羧酸。DOI:10.1016/s0040-4039(99)01188-0
-
作为产物:描述:参考文献:名称:Shono, Tatsuya; Matsumura, Yoshihiro; Inoue, Kenji, Journal of the Chemical Society. Perkin transactions I, 1986, p. 73 - 78摘要:DOI:
文献信息
-
Two-way homologation of aliphatic aldehydes: Both one-carbon shortening and lengthening via the same intermediate作者:Jae Won Yoo、Youngran Seo、Jong Beom Park、Young Gyu KimDOI:10.1016/j.tet.2019.130883日期:2020.2Aliphatic aldehydes can be homologated to both one-carbon shorter and one-carbon longer homologous carbonyl compounds through the 2–4 steps of reactions via the same intermediates, β,γ-unsaturated α-nitrosulfones, prepared from the proline-catalyzed sequential reactions of several aliphatic aldehydes with phenylsulfonylnitromethane. While the oxidative cleavage of the key intermediates gave one-carbon
-
Synthesis of dimethyl acetals. diethyl acetals. and cyclic acetals catalyzed by aminopropylated silica gel hydrochloride (APSG-HCL)作者:F. Gasparrini、M. Giovannoli、D. Misiti、G. PalmieriDOI:10.1016/s0040-4020(01)91796-x日期:1984.1The aminopropylated Silica-Gel hydrochloride (APSG.HCl) proved to be an efficient catalyst for the rapid conversion of carbonyl compounds in the corresponding acetals with high yields and in mild and selective conditions. In addition to the obvious advantages offered by heterogeneous catalysis, the present method results very useful when the presence of a weakly-acidic function chemically bonded on
-
Er(OTf)<sub>3</sub>as a Mild Cleaving Agents for Acetals and Ketals作者:Antonio Procopio、Renato Dalpozzo、Antonio De Nino、Loredana Maiuolo、Monica Nardi、Antonio TagarelliDOI:10.1055/s-2004-815941日期:——Er(OTf) 3 is proposed as a very gentle Lewis acid catalyst in a chemoselective method for the cleavage of alkyl and cyclic acetals and ketals at room temperature in wet nitromethane is presented.
-
Cyanotrimethylsilane as a versatile reagent for introducing cyanide functionality作者:Kiitiro Utimoto、Yukio Wakabayashi、Takafumi Horiie、Masaharu Inoue、Yuho Shishiyama、Michio Obayashi、Hitosi NozakiDOI:10.1016/s0040-4020(01)88595-1日期:1983.1Cyanotrimethylsilane adds to some ⇌,β-unsaturated ketones in conjugate manner under the catalytic action of Lewis acids such as triethylaluminium, aluminium chloride, and SnCl2. Hydrolysis of the products gives β-cyano ketones which are identical to the hydrocyanated products of the starting enones. The title silicon reagent reacts with acetals and orthoesters under the catalytic action of SnCI2 or BF3-OEt2
-
Electrogenerated Acid-Catalyzed Reactions of Acetals, Aldehydes, and Ketones with Organosilicon Compounds, Leading to Aldol Reactions, Allylations, Cyanations, and Hydride Additions作者:Sigeru Torii、Tsutomu Inokuchi、Sadahito Takagishi、Hirofumi Horike、Hideki Kuroda、Kenji UneyamaDOI:10.1246/bcsj.60.2173日期:1987.6electrogenerated acid (EG acid) in the silicon-mediated acid-catalyzed reactions; e.g., aldol reactions, allylations, cyanations, and hydride additions is described. The aldol reaction of acetals 1 with enol trimethylsilyl ethers 3 and 1,2-bis(trimethylsiloxy)alkenes 4 gives the corresponding adducts 5 and 6, respectively. The reaction proceeds smoothly with EG acid derived from perchlorate salts such
表征谱图
-
氢谱1HNMR
-
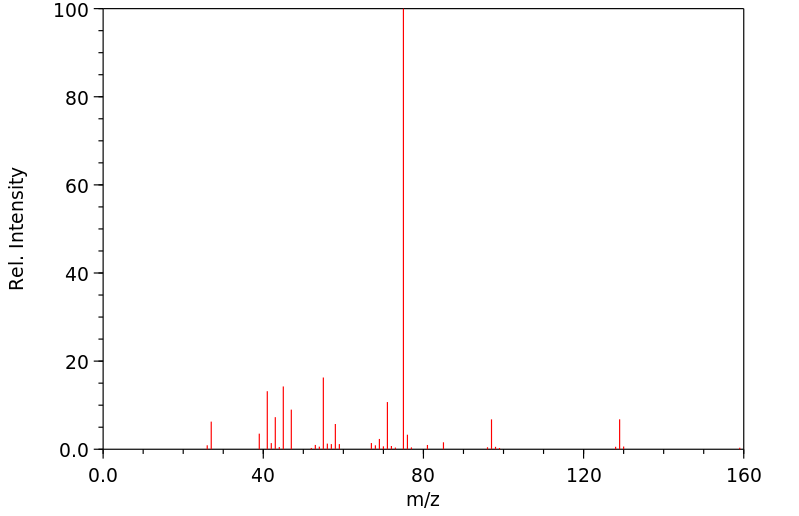
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷