2-(1-哌啶基)苯甲腈 | 72752-52-4
中文名称
2-(1-哌啶基)苯甲腈
中文别名
2-(1-哌啶基)苯腈
英文名称
2-(piperidin-1-yl)benzonitrile
英文别名
2-piperidin-1-ylbenzonitrile;2-piperidino-benzonitril;2-piperidin-1-yl-benzonitrile;2-(1-piperidinyl)benzonitrile;2-piperidino-benzonitrile;2-Piperidinobenzonitrile
CAS
72752-52-4
化学式
C12H14N2
mdl
MFCD00049221
分子量
186.257
InChiKey
MEBVSLLKZSAIGK-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:45 °C
-
沸点:122-125°C 1mm
-
密度:1.09±0.1 g/cm3(Predicted)
-
闪点:122-125°C/1mm
-
稳定性/保质期:
远离氧化物。
计算性质
-
辛醇/水分配系数(LogP):2.6
-
重原子数:14
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.416
-
拓扑面积:27
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:6.1
-
安全说明:S22,S36/37/39
-
危险类别码:R20/21/22
-
海关编码:2933399090
-
包装等级:III
-
危险类别:6.1
-
危险品运输编号:3276
-
危险性防范说明:P280,P305+P351+P338
-
危险性描述:H302
-
储存条件:存放在密封容器中,并放置在阴凉、干燥处。储存地点需上锁,钥匙应由技术专家及其助手保管。同时,储存地点应远离氧化剂。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 2-Piperidinobenzonitrile
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H411: Toxic to aquatic life with long lasting effects
H401: Toxic to aquatic life
P273: Avoid release to the environment
Section 3. Composition/information on ingredients.
Ingredient name: 2-Piperidinobenzonitrile
CAS number: 72752-52-4
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C12 H14 N2
Molecular weight: 186.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 2-Piperidinobenzonitrile
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H411: Toxic to aquatic life with long lasting effects
H401: Toxic to aquatic life
P273: Avoid release to the environment
Section 3. Composition/information on ingredients.
Ingredient name: 2-Piperidinobenzonitrile
CAS number: 72752-52-4
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C12 H14 N2
Molecular weight: 186.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— N-(but-3-enyl)-2-cyanobenzenamine 157428-86-9 C11H12N2 172.23 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-(1-哌啶基)苄胺 (2-(piperidin-1-yl)phenyl)methanamine 72752-54-6 C12H18N2 190.288 2-哌啶基-1-苯甲醛 2-(piperidin-1-yl)benzaldehyde 34595-26-1 C12H15NO 189.257 —— 1-(2-Piperidin-1-ylphenyl)ethanimine 83901-83-1 C13H18N2 202.299 —— 1-(2-piperidino-phenyl)-ethylamine 89606-11-1 C13H20N2 204.315 2-哌啶苯甲酸 2-(piperidin-1-yl)benzoic acid 42093-97-0 C12H15NO2 205.257 2-哌啶基苯甲酰胺 2-Piperidinobenzamid 3430-40-8 C12H16N2O 204.272 —— 1-(2-piperidino-phenyl)-butylamine 99469-97-3 C15H24N2 232.369 —— (S)-1-(2-(1-piperidinyl)phenyl)-butylamine 219922-02-8 C15H24N2 232.369 —— (R)-1-(2-(1-piperidinyl)phenyl)-butylamine 219921-98-9 C15H24N2 232.369 —— 1-(2-(1-piperidinyl)phenyl)pentan-1-ol —— C16H25NO 247.381 —— (Z)-2-Piperidino-benzamidoxim 131269-45-9 C12H17N3O 219.286 —— 1-(2-piperidino-phenyl)-pentylamine 219921-60-5 C16H26N2 246.396 —— Phenyl-(2-piperidin-1-ylphenyl)methanimine 1026540-82-8 C18H20N2 264.37 —— 1-(2-Piperidin-1-ylphenyl)butan-1-imine 89606-19-9 C15H22N2 230.353 瑞格列奈 3-methyl-1-[2-(piperidin-1-yl)phenyl]butan-1-amine 108157-52-4 C16H26N2 246.396 (S)-3-甲基-1-[2-(1-哌啶基)苯基]丁胺 (S)-3-methyl-1-(2-piperidin-1-yl-phenyl)butylamine 147769-93-5 C16H26N2 246.396 瑞格列奈杂质 (R)-3-methyl-1-(2-(1-pieridinyl)phenyl)-butylamine 219921-93-4 C16H26N2 246.396 瑞格列奈杂质P 3-methyl-1-(2-(1-piperidinyl)phenyl)butan-1-ol 332347-70-3 C16H25NO 247.381 —— phenyl-(2-piperidino-phenyl)-methylamine 99469-98-4 C18H22N2 266.386 —— (R)-phenyl-(2-(1-piperidinyl)phenyl)-methylamine 219922-09-5 C18H22N2 266.386 —— (S)-phenyl-(2-(1-piperidinyl)phenyl)-methylamine 219922-10-8 C18H22N2 266.386 —— n-propyl-(2-piperidino-phenyl)-ketone 219921-99-0 C15H21NO 231.338 —— 1-(2-piperidino-phenyl)-hexylamine 219921-61-6 C17H28N2 260.423 —— (±)-2-phenyl-1-[2-(piperidin-1-yl)phenyl]ethylamine 107362-46-9 C19H24N2 280.413 —— 1-(2-piperidino-phenyl)-4-pentenylamine 89606-14-4 C16H24N2 244.38 —— 1-(2-piperidino-phenyl)-heptylamine 219921-62-7 C18H30N2 274.45 —— 1-(2-Piperidin-1-ylphenyl)pentan-1-imine 89606-17-7 C16H24N2 244.38 —— 2-Phenyl-1-(2-piperidin-1-ylphenyl)ethanimine 89606-21-3 C19H22N2 278.397 —— (3-methyl-butyl)-(2-piperidino-phenyl)-ketimine 147769-96-8 C16H24N2 244.38 —— 1-(2-Piperidin-1-ylphenyl)hexan-1-imine 89606-18-8 C17H26N2 258.407 瑞格列奈杂质O 3-methyl-1-(2-piperidin-1-yl-phenyl)-butan-1-one 147770-03-4 C16H23NO 245.365 —— 2-cyclopropyl-1-(2-piperidino-phenyl)ethylamine 219922-85-7 C16H24N2 244.38 —— 1-(2-Piperidin-1-ylphenyl)heptan-1-imine 1027833-78-8 C18H28N2 272.434 —— 3-methyl-1-(2-piperidino-phenyl)-3-butenylamine 219921-63-8 C16H24N2 244.38 - 1
- 2
- 3
- 4
反应信息
-
作为反应物:描述:2-(1-哌啶基)苯甲腈 在 sodium hydroxide 、 sodium tetrahydroborate 、 N-乙酰-L-谷氨酸 、 碘 、 magnesium 、 N,N'-二环己基碳二亚胺 作用下, 以 四氢呋喃 、 甲醇 、 二氯甲烷 、 异丙醇 、 丙酮 为溶剂, 反应 15.0h, 生成 瑞格列奈参考文献:名称:An Improved Process for Repaglinide via an Efficient and One Pot Process of (1S)-3-methyl-1-(2-piperidin-1-ylphenyl)butan-1-amine – A Useful Intermediate摘要:本文描述了(1S)-3-甲基-1-(2-哌啶-1-基苯基)丁-1-胺(S-(+)-1),瑞格列奈(2)的关键中间体的大规模合成的发展。优化并缩短了S-(+)-1的过程条件,包括亲核取代、Grignard反应、还原和分辨。与现有的方法相比,不需要的对映异构体R-(?)-1的消旋具有成本和总产率的显着优势。本文还描述了在S-(+)-1与苯乙酸衍生物3缩合合成2的过程中所得到的DcU副产物的控制。DOI:10.2533/chimia.2006.593
-
作为产物:参考文献:名称:An Improved Process for Repaglinide via an Efficient and One Pot Process of (1S)-3-methyl-1-(2-piperidin-1-ylphenyl)butan-1-amine – A Useful Intermediate摘要:本文描述了(1S)-3-甲基-1-(2-哌啶-1-基苯基)丁-1-胺(S-(+)-1),瑞格列奈(2)的关键中间体的大规模合成的发展。优化并缩短了S-(+)-1的过程条件,包括亲核取代、Grignard反应、还原和分辨。与现有的方法相比,不需要的对映异构体R-(?)-1的消旋具有成本和总产率的显着优势。本文还描述了在S-(+)-1与苯乙酸衍生物3缩合合成2的过程中所得到的DcU副产物的控制。DOI:10.2533/chimia.2006.593
文献信息
-
[EN] SUBSTITUTED PIPERIDINES THAT INCREASE p53 ACTIVITY AND THE USES THEREOF<br/>[FR] PIPÉRIDINES SUBSTITUÉES QUI ACCROISSENT L'ACTIVITÉ DE P53, ET UTILISATIONS DE CES COMPOSÉS申请人:SCHERING CORP公开号:WO2011046771A1公开(公告)日:2011-04-21The present invention provides a compound of Formula (1) as described herein or a pharmaceutically acceptable salt, solvate or ester thereof. The compounds are useful as inhibitors of the HDM2 protein. Also disclosed are pharmaceutical compositions comprising the above compounds and methods of treating cancer using the same.
-
Rhodium(<scp>iii</scp>)-catalyzed olefinic C–H alkynylation of enamides at room temperature作者:Chao Feng、Daming Feng、Teck-Peng LohDOI:10.1039/c4cc04072d日期:——
Rh(
iii )-catalyzed C–H olefinic alkynylation of enamides for the stereospecific construction of synthetically usefulZ -type enynamides is reported. This protocol displays good functionality tolerance and operational simplicity thus providing an alternative synthetic opportunity for the ease of access to specific 1,3-enyne derivatives.报告了Rh(III)催化的烯酰胺的C-H烯烃炔基化反应,用于立体特异性构建具有合成用途的Z型烯炔酰胺。该方案显示出良好的官能团容忍性和操作简便性,从而为轻松获取特定的1,3-炔烯衍生物提供了另一种合成机会。 -
Repaglinide and Related Hypoglycemic Benzoic Acid Derivatives作者:Wolfgang Grell、Rudolf Hurnaus、Gerhart Griss、Robert Sauter、Eckhard Rupprecht、Michael Mark、Peter Luger、Herbert Nar、Helmut Wittneben、Peter MüllerDOI:10.1021/jm9810349日期:1998.12.1carboxy group further increased activity and duration of action in the rat. The most active racemic compound, 6al (R4 = isobutyl; R = ethoxy), turned out to be 12 times more active than the sulfonylurea (SU) glibenclamide (1). Activity was found to reside predominantly in the (S)-enantiomers. Compared with the SUs 1 and 2 (glimepiride), the most active enantiomer, (S)-6al (AG-EE 623 ZW; repaglinide;研究了两个系列的降血糖苯甲酸衍生物(5、6)的构效关系。当2-甲氧基被亚烷基亚氨基残基取代时,系列5由美格替宁(3)产生。用顺式3、5-二甲基哌啶子基(5h)和八亚甲基亚氨基(5l)残基观察到最大活性。当将2-甲氧基,5-氟和α-甲基残基替换为2-哌啶子基,5-氢和较大的α-烷基残基时,具有反向酰胺基功能的meglitinide类似物4产生6系列, 分别。羧基邻位的烷氧基残基进一步增加了大鼠的活性和作用时间。最具活性的外消旋化合物6al(R4 =异丁基; R =乙氧基)的活性比磺酰脲(SU)格列本脲(1)高12倍。发现活性主要存在于(S)-对映异构体中。与SUs 1和2(格列美脲)相比,活性最高的对映异构体(S)-6al(AG-EE 623 ZW;瑞格列奈; ED50 = 10 micro / kg po)活性高25和18倍。瑞格列奈对2型糖尿病患者是一种有用的治疗药物。FDA和EMEA最
-
Substituted compounds derived from N-(benzyl)phenylacetamide, preparation and uses申请人:Masson Christophe公开号:US20060079696A1公开(公告)日:2006-04-13This invention relates to poly-substituted derivatives of the N-(benzyl)phenylacetamide type, pharmaceutical compositions comprising same, therapeutic uses thereof, more particularly in the fields of human and animal health. This invention also relates to a process for the preparation of such derivatives.
-
[EN] HETEROCYCLIC DERIVATIVES AS RORGAMMA MODULATORS<br/>[FR] DÉRIVÉS HÉTÉROCYCLIQUES EN TANT QUE MODULATEURS RORGAMMA申请人:GENFIT公开号:WO2016102633A1公开(公告)日:2016-06-30The present invention provides novel compounds of formula (I) that are modulators of RORgamma. These compounds, and pharmaceutical compositions comprising the same, are suitable means for treating any disease wherein the modulation of RORgamma has therapeutic effects, for instance in autoimmune diseases, autoimmune-related diseases, inflammatory diseases, fibrotic diseases, or cholestatic diseases.本发明提供了一种新颖的公式(I)化合物,该化合物是RORgamma的调节剂。这些化合物以及包含相同化合物的药物组合物,适用于治疗任何一种疾病,其中调节RORgamma具有治疗作用,例如在自身免疫性疾病、自身免疫相关疾病、炎症性疾病、纤维化疾病或胆汁淤积性疾病中。
表征谱图
-
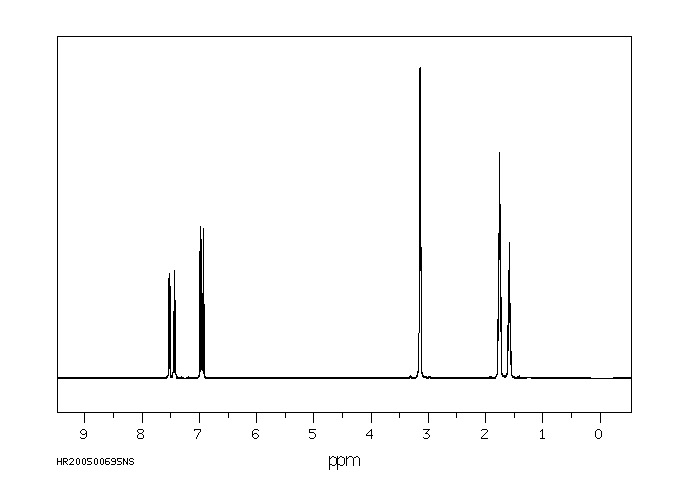
氢谱1HNMR
-
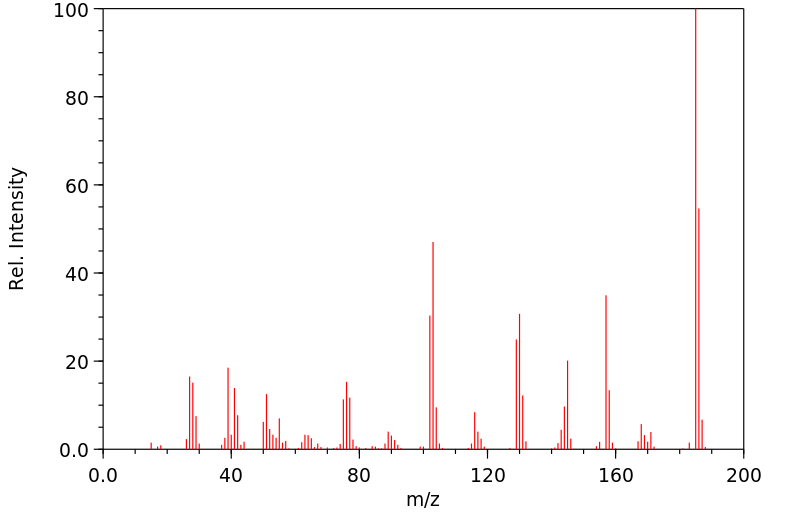
质谱MS
-
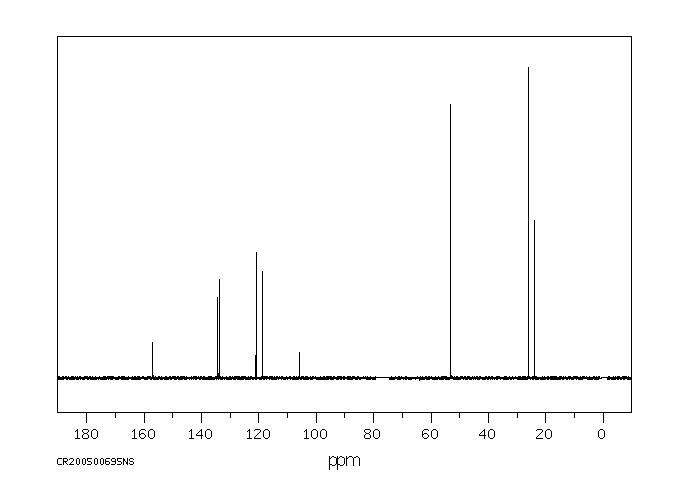
碳谱13CNMR
-
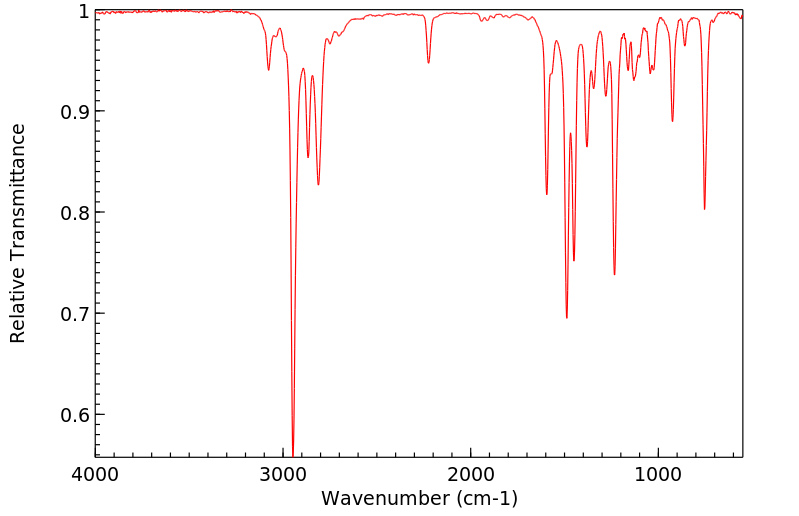
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(R)-3-甲基哌啶盐酸盐;
(R)-2-苄基哌啶-1-羧酸叔丁酯
((3S,4R)-3-氨基-4-羟基哌啶-1-基)(2-(1-(环丙基甲基)-1H-吲哚-2-基)-7-甲氧基-1-甲基-1H-苯并[d]咪唑-5-基)甲酮盐酸盐
高氯酸哌啶
高托品酮肟
马来酸帕罗西汀
颜料红48:4
顺式3-氟哌啶-4-醇盐酸盐
顺式2,6-二甲基哌啶-4-酮
顺式1-苄基-4-甲基-3-甲氨基-哌啶
顺式-叔丁基4-羟基-3-甲基哌啶-1-羧酸酯
顺式-6-甲基-哌啶-1,3-二甲酸1-叔丁酯
顺式-5-(三氟甲基)哌啶-3-羧酸甲酯盐酸盐
顺式-4-叔丁基-2-甲基哌啶
顺式-4-Boc-氨基哌啶-3-甲酸甲酯
顺式-4-(氮杂环丁烷-1-基)-3-氟哌
顺式-3-顺式-4-氨基哌啶
顺式-3-甲氧基-4-氨基哌啶
顺式-3-BOC-3,7-二氮杂双环[4.2.0]辛烷
顺式-3-(1-吡咯烷基)环丁腈
顺式-3,5-哌啶二羧酸
顺式-3,4-二溴-3-甲基吡咯烷盐酸盐
顺式-2,6-二甲基-4-氧代哌啶-1-羧酸叔丁基酯
顺式-1-叔丁氧羰基-4-甲基氨基-3-羟基哌啶
顺式-1-boc-3,4-二氨基哌啶
顺式-1-(4-叔丁基环己基)-4-苯基-4-哌啶腈
顺式-1,3-二甲基-4-乙炔基-6-苯基-3,4-哌啶二醇
顺-4-(4-氟苯基)-1-(4-异丙基环己基)-4-哌啶羧酸
顺-4-(2-氟苯基)-1-(4-异丙基环己基)-4-哌啶羧酸
顺-3-氨基-4-氟哌啶-1-羧酸叔丁酯
顺-1-苄基-4-甲基哌啶-3-氨基酸甲酯盐酸盐
非莫西汀
雷芬那辛
雷拉地尔
阿维巴坦中间体4
阿格列汀杂质
阿尼利定盐酸盐 CII
阿尼利定
阿塔匹酮
阿哌沙班杂质BMS-591455
阿哌沙班杂质87
阿哌沙班杂质52
阿哌沙班杂质51
阿哌沙班杂质5
阿哌沙班杂质
阿哌沙班杂质
阿哌沙班-d3
阿哌沙班
阻聚剂701
间氨基谷氨酰胺