Section I.Chemical Product and Company Identification
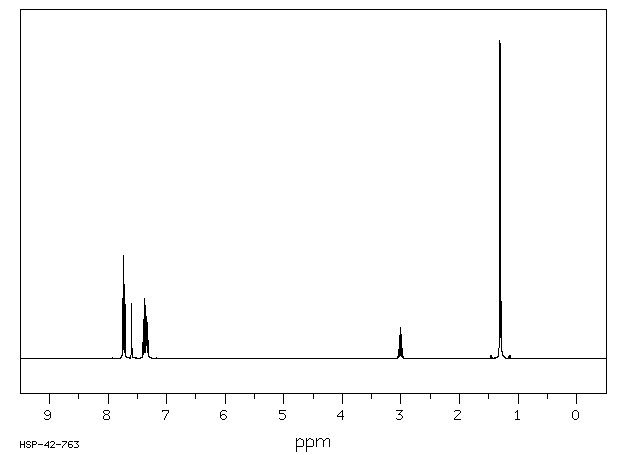
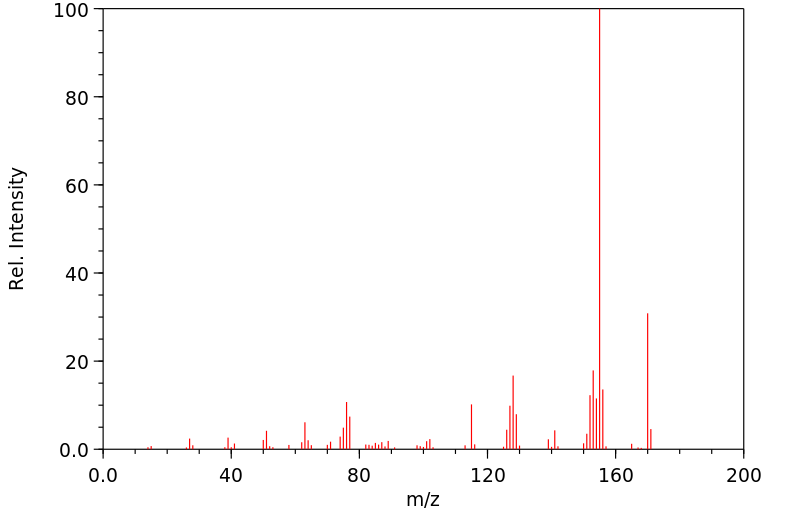
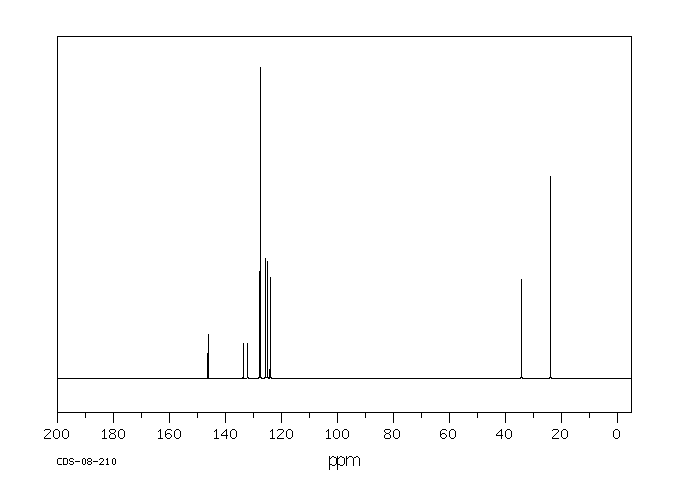
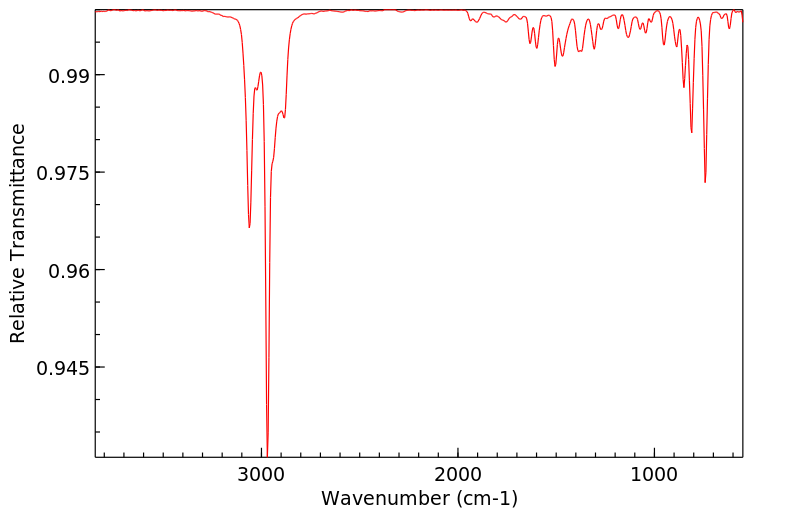
Chemical Name 2-Isopropylnaphthalene (beta-)
Portland OR
Synonym Not available.
Chemical Formula C10H7CH(CH3)2
CAS Number 2027-17-0
Section II. Composition and Information on Ingredients
Chemical Name CAS Number Percent (%) TLV/PEL Toxicology Data
2-Isopropylnaphthalene (beta-) 2027-17-0 Min. 94.0 Not available. Mouse LD50 (oral) 5300 mg/kg
(GC)
Section III. Hazards Identification
Acute Health Effects No specific information is available in our data base regarding the toxic effects of this material for humans. However,
exposure to any chemical should be kept to a minimum. Skin and eye contact may result in irritation. May be harmful if
inhaled or ingested. Always follow safe industrial hygiene practices and wear proper protective equipment when handling
this compound.
CARCINOGENIC EFFECTS : Not available.
Chronic Health Effects
MUTAGENIC EFFECTS : Not available.
TERATOGENIC EFFECTS : Not available.
Toxicity to the reproductive system: Not available.
There is no known effect from chronic exposure to this product. Repeated or prolonged exposure to this compound is
not known to aggravate existing medical conditions.
Section IV. First Aid Measures
Eye Contact Check for and remove any contact lenses. DO NOT use an eye ointment. Flush eyes with running water for a minimum
of 15 minutes, occasionally lifting the upper and lower eyelids. Seek medical attention. Treat symptomatically and
supportively.
Skin Contact If the chemical gets spilled on a clothed portion of the body, remove the contaminated clothes as quickly as possible,
protecting your own hands and body. Place the victim under a deluge shower. If the chemical touches the victim's
exposed skin, such as the hands: Gently and thoroughly wash the contaminated skin with running water and non-abrasive
soap. Be particularly careful to clean folds, crevices, creases and groin. Cover the irritated skin with an emollient. Seek
medical attention. Treat symptomatically and supportively. Wash any contaminated clothing before reusing.
Inhalation If the victim is not breathing, perform artificial respiration. Loosen tight clothing such as a collar, tie, belt or waistband. If
breathing is difficult, oxygen can be administered. Seek medical attention. Treat symptomatically and supportively.
Remove dentures if any. Have conscious person drink several glasses of water or milk. INDUCE VOMITING by sticking
Ingestion
finger in throat. Lower the head so that the vomit will not reenter the mouth and throat. NEVER give an unconscious
person anything to ingest. Seek medical attention. Treat symptomatically and supportively.
Section V. Fire and Explosion Data
Not available.
Flammability Combustible. Auto-Ignition
Flammable Limits
Flash Points Not available.
122°C (251.6°F)
Combustion Products These products are toxic carbon oxides (CO, CO 2).
Fire Hazards
No specific information is available regarding the flammability of this compound in the presence of various materials.
Explosion Hazards Risks of explosion of the product in presence of mechanical impact: Not available.
Risks of explosion of the product in presence of static discharge: Not available.
No additional information is available regarding the risks of explosion.
Fire Fighting Media
SMALL FIRE: Use DRY chemicals, CO 2, water spray or foam.
LARGE FIRE: Use water spray, fog or foam. DO NOT use water jet.
and Instructions
Continued on Next Page
2-Isopropylnaphthalene (beta-)
Section VI. Accidental Release Measures
Spill Cleanup Combustible material.
Instructions Keep away from heat and sources of ignition. Mechanical exhaust required. Stop leak if without risk. Finish cleaning the
spill by rinsing any contaminated surfaces with copious amounts of water.
Section VII. Handling and Storage
Keep away from heat and sources of ignition. Mechanical exhaust required. When not in use, tightly seal the container
Handling and Storage
and store in a dry, cool place. Avoid excessive heat and light. Do not breathe gas, fumes, vapor or spray. In case of
Information
insufficient ventilation, wear suitable respiratory equipment. If you feel unwell, seek medical attention and show the label
when possible. Treat symptomatically and supportively. Avoid contact with skin and eyes.
Always store away from incompatible compounds such as oxidizing agents.
Section VIII. Exposure Controls/Personal Protection
Engineering Controls Provide exhaust ventilation or other engineering controls to keep the airborne concentrations of vapors below their
respective threshold limit value. Ensure that eyewash station and safety shower is proximal to the work-station location.
Personal Protection Splash goggles. Lab coat. Vapor respirator. Boots. Gloves. A MSHA/NIOSH approved respirator must be used to avoid
inhalation of the product. Suggested protective clothing might not be sufficient; consult a specialist BEFORE handling
this product.
Exposure Limits Not available.
Section IX. Physical and Chemical Properties
Physical state @ 20°C Clear, yellowish-brown liquid. Solubility
Insoluble in cold water, hot water.
0.973 (water=1)
Specific Gravity
Molecular Weight 170.25 Partition Coefficient
log Kow = 4.89
Boiling Point 268°C (514.4°F) Vapor Pressure 100 mm of Hg (@ 188.8°C)
Melting Point -16°C (3.2°F) Vapor Density Not available.
Not available. 100% (v/v).
Refractive Index Volatility
Critical Temperature Not available. Odor Sweetish. (Slight.)
Viscosity Not available. Taste Not available.
Section X. Stability and Reactivity Data
Stability
This material is stable if stored under proper conditions. (See Section VII for instructions)
Conditions of Instability
Avoid excessive heat and light.
Incompatibilities
Reactive with strong oxidizing agents.
Section XI. Toxicological Information
RTECS Number QJ8000000
Routes of Exposure Ingestion. Inhalation.
Mouse LD50 (oral) 5300 mg/kg
Toxicity Data
CARCINOGENIC EFFECTS : Not available.
Chronic Toxic Effects
MUTAGENIC EFFECTS : Not available.
TERATOGENIC EFFECTS : Not available.
Toxicity to the reproductive system: Not available.
There is no known effect from chronic exposure to this product. Repeated or prolonged exposure to this compound is not
known to aggravate existing medical conditions.
Acute Toxic Effects No specific information is available in our data base regarding the toxic effects of this material for humans. However,
exposure to any chemical should be kept to a minimum. Skin and eye contact may result in irritation. May be harmful if
inhaled or ingested. Always follow safe industrial hygiene practices and wear proper protective equipment when handling
this compound.
Continued on Next Page
2-Isopropylnaphthalene (beta-)
Section XII. Ecological Information
Ecotoxicity Not available.
2-Isopropylnaphthalene is a component of crude oil and a product of combustion which is produced and released to the
Environmental Fate
environment during natural fires. Emissions from petroleum refining, coal tar distillation, and gasoline and diesel fueled
engines are contributors of 2-isopropylnaphthalene to the environment. 2-Isopropylnaphthalene is also used as chemical
intermediate and a general solvent. Consequently, 2-isopropylnaphthalene is released to the environment via
manufacturing effluents. 2-Isopropylnaphthalene is also released to the environment by municipal waste incinerators.
2-Isopropylnaphthalene should biodegrade in the environment. However, hydrolysis of 2-isopropylnaphthalene should not
be important. In sunlit environmental media, 2-isopropylnaphthalene will undergo direct photolysis. 2-Isopropylnaphthalene
is expected to be slightly mobile to immobile in soil. 2-Isopropylnaphthalene has the potential to bioconcentrate in aquatic
organisms. 2-Isopropylnaphthalene may also partition from the water column to organic matter contained in sediments
and suspended solids. Volatilization of 2-isopropylnaphthalene from environmental waters may be important. The
volatilization half-lives from a model river and a model pond, the latter considers the effect of adsorption, have been
estimated to be 5.2 hr and 93.6 days, respectively. 2-Isopropylnaphthalene is expected to exist entirely in the vapor phase
in ambient air. In the atmosphere, reaction with photochemically produced hydroxyl radicals (half-life of 6.6 hr) is likely to
be important. The most probable human exposure would be occupational exposure, which may occur through dermal
contact or inhalation at places were 2-isopropylnaphthalene is produced or used. Non-occupational exposures would most
likely occur via urban atmospheres, contaminated drinking water supplies and recreational activities at contaminated
waterways. (Excerpted from HSDB, 1996)
Section XIII. Disposal Considerations
Waste Disposal Recycle to process, if possible. Consult your local or regional authorities. You may be able to dissolve or mix material with
a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system. Observe all
federal, state, and local regulations when disposing of this substance.
Section XIV. Transport Information
DOT Classification Not a DOT controlled material (United States).
PIN Number NONE
Proper Shipping Name NONE
Packing Group (PG) NONE
DOT Pictograms
Section XV. Other Regulatory Information and Pictograms
TSCA Chemical Inventory This product is ON the EPA Toxic Substances Control Act (TSCA) inventory.
(EPA)
WHMIS Classification Not controlled under WHMIS (Canada).
(Canada)
EINECS Number (EEC) Not available.
EEC Risk Statements
Not available.
2-Isopropylnaphthalene (beta-)
TCI laboratory chemicals are for research purposes only and are NOT intended for use as drugs, food additives, households, or pesticides. The information herein is believed to be correct, but does not
claim to be all inclusive and should be used only as a guide. Neither the above named supplier nor any of its subsidiaries assumes any liability whatsoever for the accuracy or completeness of the
information contained herein. Final determination of suitability of any material is the sole responsibility of the user. All chemical reagents must be handled with the recognition that their chemical,
physiological, toxicological, and hazardous properties have not been fully investigated or determined. All chemical reagents should be handled only by individuals who are familiar with their potential
hazards and who have been fully trained in proper safety, laboratory, and chemical handling procedures. Although certain hazards are described herein, we can not guarantee that these are the only hazards
which exist. Our MSDS sheets are based only on data available at the time of shipping and are subject to change without notice as new information is obtained. Avoid long storage periods since the
product is subject to degradation with age and may become more dangerous or hazardous. It is the responsibility of the user to request updated MSDS sheets for products that are stored for extended
periods. Disposal of unused product must be undertaken by qualified personnel who are knowledgeable in all applicable regulations and follow all pertinent safety precautions including the use of
appropriate protective equipment (e.g. protective goggles, protective clothing, breathing equipment, facial mask, fume hood). For proper handling and disposal, always comply with federal, state, and local
regulations.
SECTION 16 - ADDITIONAL INFORMATION
N/A