2-甲基-3-戊炔-2-醇 | 590-38-5
中文名称
2-甲基-3-戊炔-2-醇
中文别名
——
英文名称
2-methyl-3-pentyn-2-ol
英文别名
2-methylpent-3-yn-2-ol;3-methyl-1-methyl-1-butyn-3-ol
CAS
590-38-5
化学式
C6H10O
mdl
——
分子量
98.1448
InChiKey
JPKQHUHDYLMRJW-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:81-82 °C(Press: 100 Torr)
-
密度:0.87g/ml
计算性质
-
辛醇/水分配系数(LogP):0.8
-
重原子数:7
-
可旋转键数:1
-
环数:0.0
-
sp3杂化的碳原子比例:0.67
-
拓扑面积:20.2
-
氢给体数:1
-
氢受体数:1
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-甲基-3-丁炔-2-醇 2-methyl-but-3-yn-2-ol 115-19-5 C5H8O 84.1179 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2-Methoxy-2-methyl-pentin-(3) 24642-05-5 C7H12O 112.172 甲基異丙基乙炔 4-methylpent-2-yne 21020-27-9 C6H10 82.1454
反应信息
-
作为反应物:描述:参考文献:名称:Re(V)-氧代配合物催化串联重排-共轭加成反应合成β-硫烷基酮摘要:已经开发了通过串联重排和共轭加成反应合成β-硫烷基酮的方法。通过将炔丙醇重排成相应的烯酮,然后共轭添加未活化的硫醇,该方法学提供了一系列β-硫烷基酮的途径。通过ReOCl 3(OPPh 3)(S(CH 3)2)催化一锅串联转化,可提供高收率的芳基和烷基β-硫烷基酮。DOI:10.1016/j.tetlet.2012.11.047
-
作为产物:描述:参考文献:名称:环加成。第三部分 在共轭烯丙酸酯和腈中添加2-重氮丙烷的立体效应摘要:丁-2-,3-二烯酸及其甲基取代的类似物与2-重氮丙烷发生更多的亲电(αβ)双键反应,但加成的方向取决于γ-碳原子上的取代度。在γ-位被单取代或未被取代的丙二烯得到4-亚烷基-1-吡唑啉(即,电子上优选的加合物)。在反应条件下,丁酸2,3-二烯酸甲酯异构体的初始加合物异构化为3-甲氧基羰基-5,5-二甲基-4-亚甲基-2-吡唑啉,后者易于与第二个分子2-重氮丙烷反应生成斯皮兰。加法的方向与γγ相反-二取代的烯丙酸酯和腈,它们仅产生3-异亚丙基-1-吡唑啉。所述逆转归因于在过渡态中对于正常添加至γγ-二甲基丙烯烯而言高度不利的“楔形”相互作用。讨论了其他几种烯丙基-重氮烷环加成反应中的取向。DOI:10.1039/j39690002443
文献信息
-
Transfert de l'ethoxycarbonylcarbene sur les alcynes substitues en α par un groupe partant作者:M. Vincens、A. Dussauge、M. VidalDOI:10.1016/0040-4020(77)88027-7日期:1977.1Synthesis of ethyl cyclopropenecarboxylates with a leaving group on the position α to the double bond has been realised by photolytic or thermocatalytic decomposition of ethyl diazoacetate with acetylenic substrates. With these adducts allenic, a etylenic or dienic esters are obtained. In the case of photolytic transfer, the proposed intermediate agent is ethoxycarbonyl carbene. If the transfer is
-
Convenient Synthesis of 4-Methylene-2-oxazolidinones and 4-Methylenetetrahydro-1,3-oxazin-2-ones via Transition-Metal Catalyzed Intramolecular Addition of Nitrogen Atom to Acetylenic Triple Bond作者:Yoshinao Tamaru、Masanari Kimura、Shuji Tanaka、Sigeru Kure、Zen-ichi YoshidaDOI:10.1246/bcsj.67.2838日期:1994.102-Propynyl tosylcarbamates 1 undergo cyclization smoothly by the catalysis of CuCl/Et3N or AgNCO/Et3N to furnish 4-methylene-2-oxazolidinones 2 in good yields. The similar cyclization of the N-acyl derivatives of 1 (PhCO, MeCO, EtOCO, etc.) is catalyzed effectively by AgNCO/t-BuOK. These reactions accommodate a variety of substituents at C1 and C3 of 2-propyn-1-ol and provide (Z)-2 as single stereoisomers. The scope of the cyclization of 3-butynyl carbamates is rather limited, and in general only N-tosyl derivatives of terminally unsubstituted 3-butyn-1-ols undergo cyclization to give 4-methylenetetrahydro-1,3-oxazin-2-ones in synthetically useful yields by the catalysis of AgNCO/Et3N or AgNCO/t-BuOK.
-
Synthesis of Isoquinolines through Ir<sup>III</sup> -Catalyzed C-H Activation/Annulation from Benzimidates with Hydroxylisopropylalkynes作者:Mingliang Liu、Wanchun Gong、Erli You、Haizhen Zhang、Lei Shi、Weiguo Cao、Jingjing ShiDOI:10.1002/ejoc.201800410日期:2018.9.30An IrIII‐catalyzed cascade reaction consisting of C–H activation and annulation of benzimidates with hydroxylisopropylalkynes yields a broad range of isoquinolines in one step with good functional‐group tolerance and high efficiency.Ir- III催化的级联反应由C–H活化和苯甲二酸酯与羟基异丙基炔的环化反应组成,可一步合成多种异喹啉,具有良好的官能团耐受性和高效率。
-
Interligand Interactions Dictate the Regioselectivity of<i>trans</i>-Hydrometalations and Related Reactions Catalyzed by [Cp*RuCl]. Hydrogen Bonding to a Chloride Ligand as a Steering Principle in Catalysis作者:Stephan M. Rummelt、Karin Radkowski、Dragoş-Adrian Roşca、Alois FürstnerDOI:10.1021/jacs.5b01475日期:2015.4.29mode in the presence of [Cp*Ru(MeCN)3]PF6 (1) as the catalyst; however, the regioselectivity is often poor with unsymmetrical substrates. This problem can be solved upon switching to a catalyst comprising a [Ru-Cl] bond, provided that the acetylene derivative carries a protic functional group. The R3M unit is then delivered with high selectivity to the alkyne-C atom proximal to this steering substituent在 [Cp*Ru(MeCN)3]PF6 (1) 作为催化剂的存在下,内炔与 R3M-H(M = Si、Ge、Sn)的反应遵循非常规的反式加成模式;然而,不对称底物的区域选择性通常很差。如果乙炔衍生物带有质子官能团,则该问题可以通过改用包含 [Ru-Cl] 键的催化剂来解决。然后,R3M 单元以高选择性传递到该控制取代基附近的炔烃-C 原子。这种导向效应源于极化的 [Ru-Cl] 键与质子取代基进行氢键合的能力,这有助于将炔烃上传、激活和锁定在配位球内。氯化物与 -MR3 中心的额外配位体接触将进入的试剂定位在匹配的方向,从而转化为高区域选择性。所提出的负载催化剂内的二次相互作用与大量制备和光谱数据以及固态新型钌 π 配合物 10 和 11 的结构一致。此外,还提出了 [Ru(σ-锡烷)] 配合物 (12a) 的第一个 X 射线结构,它确实具有外围 Ru-Cl……MR3 接触;该加合物也证实炔烃反式加成化学可能涉及
-
Scope and Mechanistic Analysis of the Enantioselective Synthesis of Allenes by Rhodium-Catalyzed Tandem Ylide Formation/[2,3]-Sigmatropic Rearrangement between Donor/Acceptor Carbenoids and Propargylic Alcohols作者:Zhanjie Li、Vyacheslav Boyarskikh、Jørn H. Hansen、Jochen Autschbach、Djamaladdin G. Musaev、Huw M. L. DaviesDOI:10.1021/ja3061529日期:2012.9.19Rhodium-catalyzed reactions of tertiary propargylic alcohols with methyl aryl- and styryldiazoacetates result in tandem reactions, consisting of oxonium ylide formation followed by [2,3]-sigmatropic rearrangement. This process competes favorably with the standard O-H insertion reaction of carbenoids. The resulting allenes are produced with high enantioselectivity (88-98% ee) when the reaction is catalyzed by the炔丙基叔醇与甲基芳基乙酸酯和苯乙烯基重氮乙酸酯的铑催化反应导致串联反应,包括氧鎓叶立德形成,然后是 [2,3]-σ 重排。该过程与类卡宾的标准 OH 插入反应相得益彰。当反应由四脯氨酸二铑配合物 Rh(2)(S-DOSP)(4) 催化时,生成的丙二烯具有高对映选择性 (88-98% ee)。当外消旋叔炔丙醇用作底物时,可以进行动力学拆分。在动力学拆分条件下,形成的丙二烯具有良好的非对映选择性和对映选择性(高达6.1:1 dr,88-93% ee),未反应的醇被对映体富集到65-95% ee。计算研究表明,高不对称诱导是通过涉及两点连接的有组织的过渡态获得的:醇氧与类卡宾之间的叶立德形成以及醇与羧酸盐配体的氢键合。2,3-sigmatropic 重排通过 OH 键的初始裂解进行,生成具有紧密开壳单线态、三线态和闭壳单线态电子态的中间体。该中间体将具有显着的双自由基特征,这与供体/受体类卡宾和更高度官能化的炔丙醇有利于
表征谱图
-
氢谱1HNMR
-
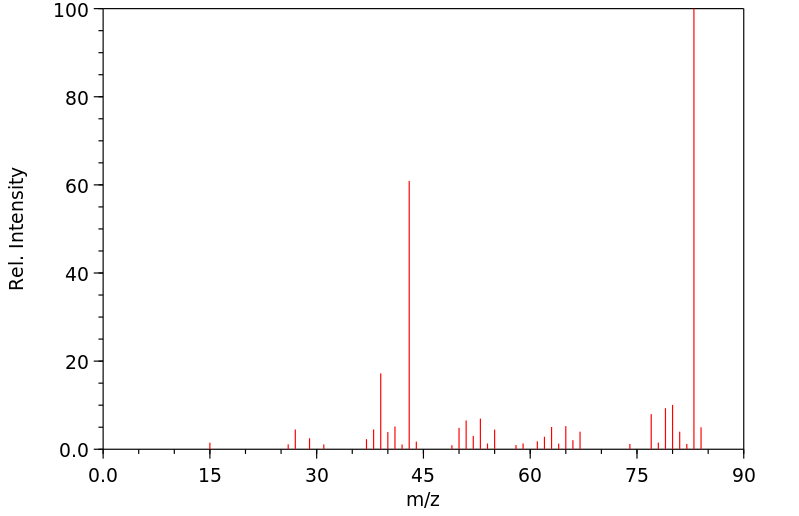
质谱MS
-
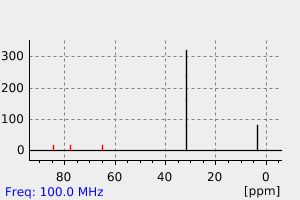
碳谱13CNMR
-
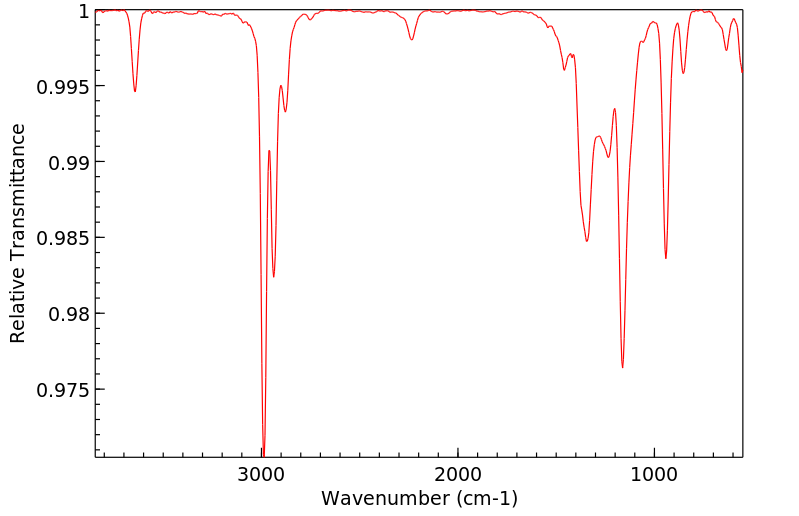
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷