顺式-3,5-二甲基环己酮 | 7214-52-0
中文名称
顺式-3,5-二甲基环己酮
中文别名
——
英文名称
3,5-dimethyl-cyclohexanone
英文别名
cis-3,5-dimethyl-cyclohexanone;cis-3,5-Dimethylcyclohexanone;(3R,5S)-3,5-dimethylcyclohexan-1-one
CAS
7214-52-0
化学式
C8H14O
mdl
——
分子量
126.199
InChiKey
MSANHHHQJYQEOK-KNVOCYPGSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):1.8
-
重原子数:9
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.88
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3,5-二甲基环己酮 3,5-dimethylcyclohexanone 2320-30-1 C8H14O 126.199 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— cis-3,5-Dimethylcycloheptanon —— C9H16O 140.225 —— (+/-)-cis-1.3-dimethyl-cycloheptanone-(5) 24291-91-6 C9H16O 140.225
反应信息
-
作为反应物:描述:顺式-3,5-二甲基环己酮 在 camphor monooxygenase from Cylindrocarpon radicicola ATCC 11011 、 还原型辅酶II(NADPH)四钠盐 作用下, 以97%的产率得到参考文献:名称:首次化学酶促合成(R)-谷口内酯和CAMO和OTEMO(两种新的Baeyer-Villiger单加氧酶)的底物谱。摘要:摘要:这项研究调查了环烷酮单加氧酶和2-氧代-Delta3-4,5,5-三甲基环戊烯基乙酰辅酶A单加氧酶(一种最近被发现的Baeyer-Villiger单加氧酶家族的酶)的底物概况,该酶被用作全细胞生物催化剂。在分析规模上进行了多种酮的生物氧化:取代的前手性环丁酮和环己酮的不对称化,萜酮和双环酮的区域发散性氧化以及外消旋环酮的动力学拆分。我们证明了标题酶在(R)-(-)-谷口内酯的对映选择性合成中的适用性,这是制备各种天然产物类似物如对苯二酚的基础。图形概要:DOI:10.1007/s00706-016-1873-9
-
作为产物:描述:参考文献:名称:Preparation of alicyclic oxygen compounds摘要:公开号:US02514329A1
文献信息
-
Anaerobic Nitroxide-Catalyzed Oxidation of Alcohols Using the NO<sup>+</sup>/NO· Redox Pair作者:Martin Holan、Ullrich JahnDOI:10.1021/ol403016p日期:2014.1.3A new method for alcohol oxidation using TEMPO or AZADO in conjunction with BF3·OEt2 or LiBF4 as precatalysts and tert-butyl nitrite as a stoichiometric oxidant has been developed. The system is based on a NO+/NO· pair for nitroxide reoxidation under anaerobic conditions. This allows the simple, high-yielding conversion of various achiral and chiral alcohols to carbonyl compounds without epimerization
-
Asymmetric Schmidt Reaction of Hydroxyalkyl Azides with Ketones作者:Kiran Sahasrabudhe、Vijaya Gracias、Kelly Furness、Brenton T. Smith、Christopher E. Katz、D. Srinivasa Reddy、Jeffrey AubéDOI:10.1021/ja0348896日期:2003.7.1asymmetric equivalent of the Schmidt reaction permits stereocontrol in ring expansions of symmetrical cyclohexanones. The procedure involves the reaction of chiral 1,2- and 1,3-hydroxyalkyl azides with ketones under acid catalysis; the initial reaction affords an iminium ether that can be subsequently opened with base. A systematic study of this reaction is reported, in which ketone substrates, chiral hydroxyalkyl
-
[EN] NOVEL SPIRO IMIDAZOLONES AS GLUCAGON RECEPTOR ANTAGONISTS, COMPOSITIONS, AND METHODS FOR THEIR USE<br/>[FR] NOUVELLES SPIRO-IMIDAZOLONES EN TANT QU'ANTAGONISTES DE RÉCEPTEUR DE GLUCAGON, COMPOSITIONS ET LEURS PROCÉDÉS D'UTILISATION申请人:SCHERING CORP公开号:WO2011119559A1公开(公告)日:2011-09-29The present invention relates to compounds of the general formula: wherein ring A, ring B, R1, R3, Z, L1, and L2 are selected independently of each other and are as defined herein, to compositions comprising the compounds, and to methods of using the compounds as glucagon receptor antagonists and for the treatment or prevention of type 2 diabetes and conditions related thereto.本发明涉及一般式化合物,其中环A、环B、R1、R3、Z、L1和L2彼此独立选择,并如本文所定义,涉及包含该化合物的组合物,以及使用该化合物作为胰高血糖素受体拮抗剂以及用于治疗或预防2型糖尿病及相关疾病的方法。
-
Palladium-Catalyzed Intramolecular Hydroalkylation of Alkenyl- ?-Keto Esters, ?-Aryl Ketones, and Alkyl Ketones in the Presence of Me3SiCl or HCl作者:Xiaoqing Han、Xiang Wang、Tao Pei、Ross A. WidenhoeferDOI:10.1002/chem.200400459日期:2004.12.17palladium-catalyzed hydroalkylation, but rather served as a source of HCl, which presumably catalyzes enolization of the ketone. Identification of HCl as the active promoter of palladium-catalyzed hydroalkylation led to the development of an effective protocol for the hydroalkylation of alkyl 3-butenyl ketones that employed sub-stoichiometric amounts of 2, HCl, and CuCl2 in a sealed tube at 70 degrees C.
-
A steric tethering approach enables palladium-catalysed C–H activation of primary amino alcohols作者:Jonas Calleja、Daniel Pla、Timothy W. Gorman、Victoriano Domingo、Benjamin Haffemayer、Matthew J. GauntDOI:10.1038/nchem.2367日期:2015.12higher-order nitrogen-containing molecules, commonly found in biologically active compounds and pharmaceutical agents. New methods for the construction of complex amines remain a continuous challenge to synthetic chemists. Here, we outline a general palladium-catalysed strategy for the functionalization of aliphatic C–H bonds within amino alcohols, an important class of small molecule. Central to this strategy
表征谱图
-
氢谱1HNMR
-
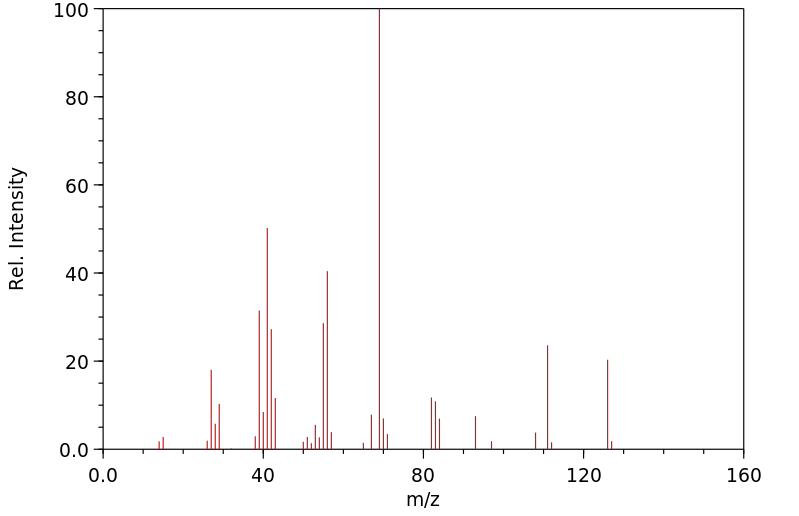
质谱MS
-
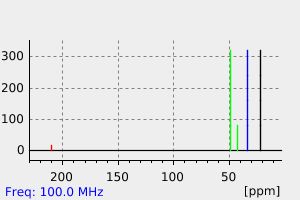
碳谱13CNMR
-
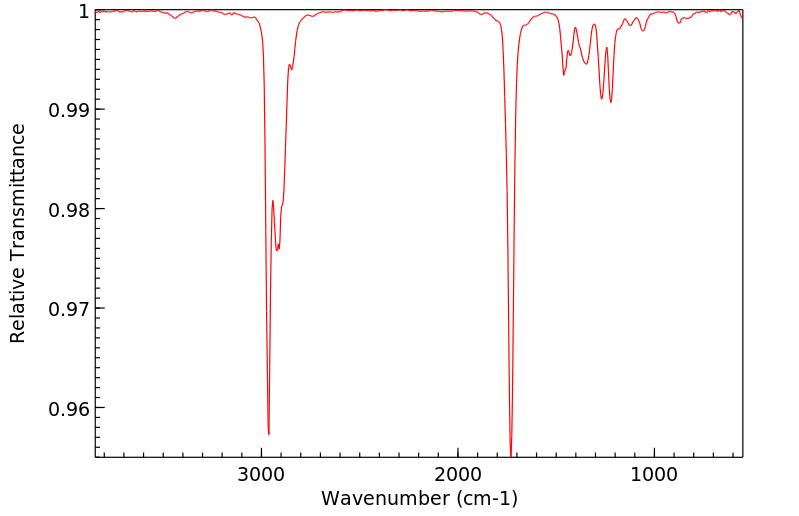
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷