乙酰丙酮酸乙酯 | 615-79-2
中文名称
乙酰丙酮酸乙酯
中文别名
2,4-二酮戊酸乙酯;丙酮草酸乙酯;2,4-二氧戊酸乙酯
英文名称
Ethyl 2,4-dioxopentanoate
英文别名
ethyl 2,4-diketopentanoate;ethyl 2,4-dioxovalerate
CAS
615-79-2
化学式
C7H10O4
mdl
MFCD00009124
分子量
158.154
InChiKey
OYQVQWIASIXXRT-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:16-18 °C (lit.)
-
沸点:101-103 °C/12 mmHg (lit.)
-
密度:1.126 g/mL at 25 °C (lit.)
-
闪点:>230 °F
-
溶解度:可溶于氯仿、甲醇
-
介电常数:16.1(19℃)
-
稳定性/保质期:
性质与稳定性:在常温常压下,不会发生分解反应。
计算性质
-
辛醇/水分配系数(LogP):0.2
-
重原子数:11
-
可旋转键数:5
-
环数:0.0
-
sp3杂化的碳原子比例:0.571
-
拓扑面积:60.4
-
氢给体数:0
-
氢受体数:4
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xn
-
安全说明:S23,S24/25
-
危险类别码:R22
-
WGK Germany:3
-
海关编码:29183000
-
危险品运输编号:NONH for all modes of transport
-
储存条件:贮存: 将密器密封,储存在密封的主容器中,并放置于阴凉、干燥处。
SDS
| Name: | Ethyl 2 4-dioxovalerate 98% Material Safety Data Sheet |
| Synonym: | None |
| CAS: | 615-79-2 |
Synonym:None
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 615-79-2 | Ethyl 2,4-dioxovalerate | 98 | 210-447-5 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 615-79-2: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear very slight yellow
Odor: none reported
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 101 - 103 deg C @ 12.00mmHg
Freezing/Melting Point: 16.00 - 18.00 deg C
Autoignition Temperature: Not applicable.
Flash Point: > 112 deg C (> 233.60 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature: Not available.
Solubility in water: Not available.
Specific Gravity/Density: 1.1260g/cm3
Molecular Formula: C7H10O4
Molecular Weight: 158.15
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, strong oxidants.
Incompatibilities with Other Materials:
Acids, bases, oxidizing agents, reducing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 615-79-2 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
Ethyl 2,4-dioxovalerate - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 615-79-2: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 615-79-2 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 615-79-2 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-氧代戊酸乙酯 ethyl 2-oxovalerate 50461-74-0 C7H12O3 144.17 3-氯-2,4-二氧代戊酸乙酯 ethyl 3-chloro-2,4-dioxopentanoate 34959-81-4 C7H9ClO4 192.599 乙酰丙酮酸 2,4-dioxo-pentanoic acid 5699-58-1 C5H6O4 130.1 —— 3-acetyl-2-oxo-γ-butyrolactone 61203-07-4 C6H6O4 142.111 —— ethyl-4-hydroximino-2-oxopentanoate 13081-00-0 C7H11NO4 173.169
反应信息
-
作为反应物:描述:参考文献:名称:Discovery of α,γ-Diketo Acids as Potent Selective and Reversible Inhibitors of Hepatitis C Virus NS5b RNA-Dependent RNA Polymerase摘要:alpha,gamma-Diketo acids (DKA) were discovered from screening as selective and reversible inhibitors of hepatitis C virus NS5b RNA-dependent RNA polymerase. The diketo acid moiety proved essential for activity, while substitution on the gamma position was necessary for selectivity and potency. Optimization led to the identification of a DKA inhibitor of NS5b polymerase with IC50 = 45 nM, one of the most potent HCV NS5b polymerase inhibitors reported.DOI:10.1021/jm0342109
-
作为产物:参考文献:名称:Synthesis of camptothecin and analogs thereof摘要:使用一种新型含羟基的三环中间体合成紫杉醇和紫杉醇类似物的方法,以及由该过程产生的紫杉醇类似物。这些紫杉醇类似物是有效的拓扑异构酶I抑制剂,并显示出抗白血病和抗肿瘤活性。公开号:US04981968A1
-
作为试剂:参考文献:名称:Van Dormael; Ghys, Bulletin des Societes Chimiques Belges, 1948, vol. 57, p. 24,29摘要:DOI:
文献信息
-
[EN] CALPAIN MODULATORS AND THERAPEUTIC USES THEREOF<br/>[FR] MODULATEURS DE CALPAÏNE ET LEURS UTILISATIONS THÉRAPEUTIQUES申请人:BLADE THERAPEUTICS INC公开号:WO2019190885A1公开(公告)日:2019-10-03Small molecule calpain modulator compounds, including their pharmaceutically acceptable salts, can be included in pharmaceutical compositions. The compounds can be useful in inhibiting calpain, or competitive binding with calpastatin, by contacting them with CAPN1, CAPN2, and/or CAPN9 enzymes residing inside a subject. The compounds and composition can also be administered to a subject in order to treat a fibrotic disease or a secondary disease state or condition of a fibrotic disease.
-
[EN] NEW 6-AMINO-QUINOLINONE COMPOUNDS AND DERIVATIVES AS BCL6 INHIBITORS<br/>[FR] NOUVEAUX COMPOSÉS 6-AMINO-QUINOLINONE ET DÉRIVÉS EN TANT QU'INHIBITEURS DE BCL6申请人:BOEHRINGER INGELHEIM INT公开号:WO2018108704A1公开(公告)日:2018-06-21The present invention encompasses compounds of formula (I), wherein the groups R1 to R5, X, Y and W have the meanings given in the claims and specification, their use as inhibitors of BCL6, pharmaceutical compositions which contain compounds of this kind and their use as medicaments, especially as agents for treatment and/or prevention of oncological diseases.本发明涵盖了式(I)的化合物,其中基团R1至R5、X、Y和W具有权利要求和说明中给定的含义,它们作为BCL6的抑制剂的用途,含有这种化合物的药物组合物以及它们作为药物的用途,特别是作为治疗和/或预防肿瘤疾病的药剂。
-
Design, synthesis, antifungal activity and 3D-QSAR study of novel pyrazole carboxamide and niacinamide derivatives containing benzimidazole moiety作者:Wei-Jie Si、Xiao-Bin Wang、Min Chen、Meng-Qi Wang、Ai-Min Lu、Chun-Long YangDOI:10.1039/c8nj05150j日期:——series of novel pyrazole carboxamide and niacinamide derivatives containing a benzimidazole moiety were designed and synthesized as antifungal candidate agents. All target compounds were characterized by FTIR, 1H NMR, 13C NMR, HRMS and elemental analysis techniques. The structure of compound T1 was further confirmed by single crystal X-ray diffraction analysis. The antifungal activities of the target设计并合成了一系列含有苯并咪唑部分的新型吡唑羧酰胺和烟酰胺衍生物,作为抗真菌候选药物。所有目标化合物均通过FTIR,1 H NMR,13 C NMR,HRMS和元素分析技术进行了表征。化合物T1的结构通过单晶X射线衍射分析进一步确认。在体外评估了目标化合物对四种植物病原真菌(灰葡萄孢,立枯根瘤菌,禾谷镰刀菌和黑斑病菌)的抗真菌活性。)通过菌丝体生长抑制方法。生物测定结果表明,一些化合物对显示出良好的抗真菌活性灰霉病以100μgML -1相比其他三个真菌。为了更好地探索结构-活性关系(SAR),测定并评估了目标化合物对灰葡萄孢的EC 50值。随后,基于被测化合物对灰葡萄孢的抑制活性,使用比较分子场分析(CoMFA)技术进行了3D定量结构-活性关系(3D-QSAR)研究。分子建模结果显示具有交叉验证的q 2的良好预测能力和非交叉验证的r 2值分别为0.578和0.850。
-
[EN] 1H-PYRAZOLO[4,3-B]PYRIDINES AS PDE1 INHIBITORS<br/>[FR] 1H-PYRAZOLO [4,3-B] PYRIDINES EN TANT QU'INHIBITEURS DE PDE1申请人:H LUNDBECK AS公开号:WO2018007249A1公开(公告)日:2018-01-11The present invention provides 1H-pyrazolo[4,3-b]pyridin-7-amines of formula (I) as PDE1 inhibitors and their use as a medicament, in particular for the treatment of neurodegenerative disorders and psychiatric disorders.
-
MACROCYCLES AS PDE1 INHIBITORS申请人:H. Lundbeck A/S公开号:US20190185489A1公开(公告)日:2019-06-20The present invention provides macrocycles of formula (I) as PDE1 inhibitors and their use as a medicament, in particular for the treatment of neurodegenerative disorders and psychiatric disorders.本发明提供了式(I)的大环化合物作为PDE1抑制剂,并将其用作药物,特别用于治疗神经退行性疾病和精神疾病。
表征谱图
-
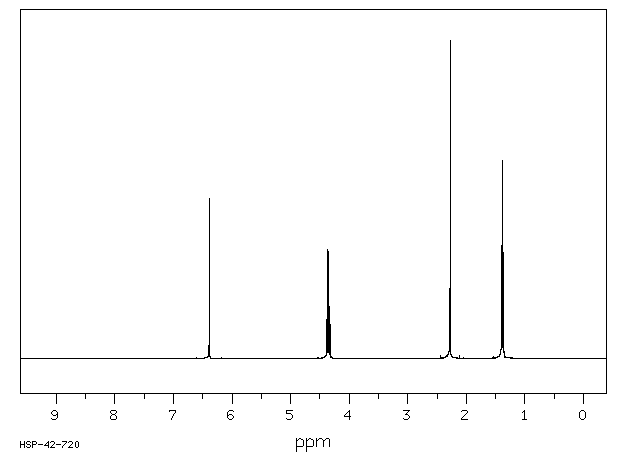
氢谱1HNMR
-
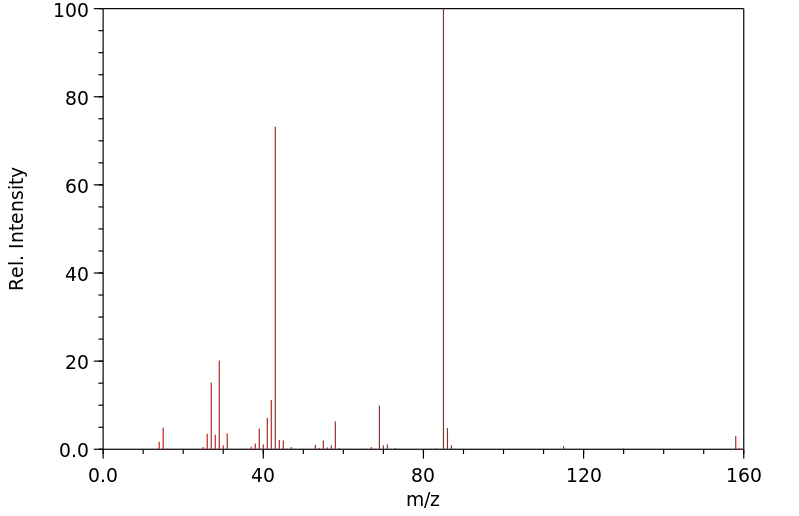
质谱MS
-
碳谱13CNMR
-
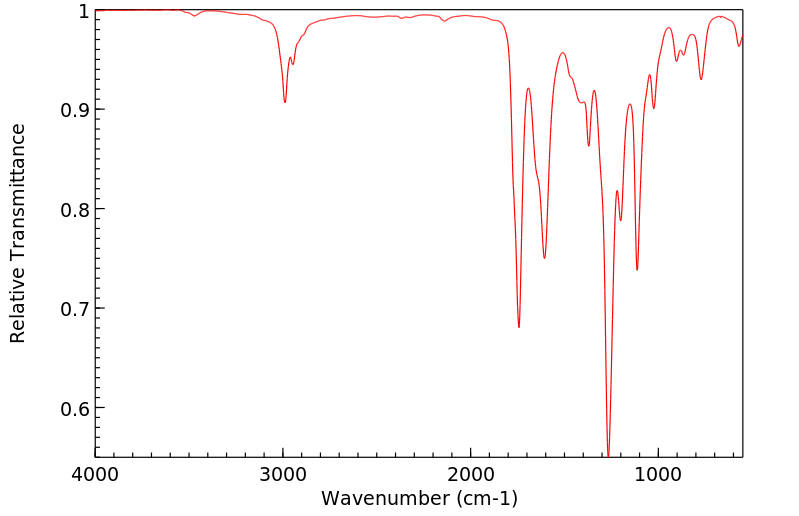
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
马来酰基乙酸
顺-3-己烯-1-丙酮酸
青霉酸
钠氟草酰乙酸二乙酯
醚化物
酮霉素
辛酸,2,4-二羰基-,乙基酯
草酸乙酯钠盐
草酰乙酸二乙酯钠盐
草酰乙酸二乙酯
草酰乙酸
草酰丙酸二乙酯
苯乙酰丙二酸二乙酯
苯丁酸,b-羰基-,2-丙烯基酯
聚氧化乙烯
羟基-(3-羟基-2,3-二氧代丙基)-氧代鏻
磷酸二氢2-{(E)-2-[4-(二乙胺基)-2-甲基苯基]乙烯基}-1,3,3-三甲基-3H-吲哚正离子
碘化镝
硬脂酰乙酸乙酯
甲氧基乙酸乙酯
甲氧基乙酰乙酸酯
甲基氧代琥珀酸二甲盐
甲基4-环己基-3-氧代丁酸酯
甲基4-氯-3-氧代戊酸酯
甲基4-氧代癸酸酯
甲基4-氧代月桂酸酯
甲基4-(甲氧基-甲基磷酰)-2,2,4-三甲基-3-氧代戊酸酯
甲基3-羰基-2-丙酰戊酸酯
甲基3-氧代十五烷酸酯
甲基2-氟-3-氧戊酯
甲基2-氟-3-氧代己酸酯
甲基2-氟-3-氧代丁酸酯
甲基2-乙酰基环丙烷羧酸酯
甲基2-乙酰基-4-甲基-4-戊烯酸酯
甲基2-乙酰基-2-丙-2-烯基戊-4-烯酸酯
甲基2,5-二氟-3-氧代戊酸酯
甲基2,4-二氟-3-氧代戊酸酯
甲基2,4-二氟-3-氧代丁酸酯
甲基1-异丁酰基环戊烷羧酸酯
甲基1-乙酰基环戊烷羧酸酯
甲基1-乙酰基环丙烷羧酸酯
甲基1-乙酰基-2-乙基环丙烷羧酸酯
甲基(2Z,4E,6E)-2-乙酰基-7-(二甲基氨基)-2,4,6-庚三烯酸酯
甲基(2S)-2-甲基-4-氧代戊酸酯
甲基(1S,2R)-2-乙酰基环丙烷羧酸酯
甲基(1R,2R)-2-乙酰基环丙烷羧酸酯
瑞舒伐他汀杂质
瑞舒伐他汀杂质
环氧乙烷基甲基乙酰乙酸酯
环戊戊烯酸,Β-氧代,乙酯