二氢芳樟醇 | 18479-51-1
中文名称
二氢芳樟醇
中文别名
3,7-二甲基-6-辛烯-3-醇,2,6-二甲基-2-辛烯-6-醇;3,7-二甲基-1-辛烯-3-醇;3,7-二甲基-6-辛烯-3-醇
英文名称
dihydrolinalool
英文别名
1,2-dihydrolinalool;3,7-dimethyl-6-octen-3-ol;3,7-dimethyloct-6-en-3-ol
CAS
18479-51-1
化学式
C10H20O
mdl
MFCD00059140
分子量
156.268
InChiKey
JRTBBCBDKSRRCY-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:-4.05°C (estimate)
-
沸点:200 °C
-
密度:0.86
-
闪点:178°C(lit.)
-
LogP:2.9 at 23℃ and pH7
-
表面张力:27.126mN/m at 1.2g/L and 20℃
-
保留指数:1119;1125;1121;1121
计算性质
-
辛醇/水分配系数(LogP):3
-
重原子数:11
-
可旋转键数:4
-
环数:0.0
-
sp3杂化的碳原子比例:0.8
-
拓扑面积:20.2
-
氢给体数:1
-
氢受体数:1
安全信息
-
危险性防范说明:P501,P261,P273,P272,P202,P210,P201,P271,P264,P280,P302+P352,P370+P378,P312,P337+P313,P305+P351+P338,P362+P364,P333+P313,P304+P340+P312,P403+P233,P403+P235,P405
-
危险性描述:H302,H315,H319,H361,H317,H336,H412,H227
-
储存条件:室温
SDS
Dihydrolinalool Revision number: 6
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Dihydrolinalool
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Dihydrolinalool
Percent: >97.0%(GC)
CAS Number: 18479-51-1
Synonyms: 3,7-Dimethyl-6-octen-3-ol
Chemical Formula: C10H20O
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Dihydrolinalool
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Very pale yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:5°C
200°C
Boiling point/range:
Flash point: 178°C
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
0.86
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Dihydrolinalool
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
No data available
Log Pow:
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Dihydrolinalool
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Dihydrolinalool
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Dihydrolinalool
Percent: >97.0%(GC)
CAS Number: 18479-51-1
Synonyms: 3,7-Dimethyl-6-octen-3-ol
Chemical Formula: C10H20O
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Dihydrolinalool
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Very pale yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:5°C
200°C
Boiling point/range:
Flash point: 178°C
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
0.86
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Dihydrolinalool
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
No data available
Log Pow:
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Dihydrolinalool
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 芳樟醇 3,7-dimethylocta-1,6-dien-3-ol 78-70-6 C10H18O 154.252 3,7-二甲基-6-辛烯-1-炔基-3-醇 3,7-dimethyloct-6-en-1-yn-3-ol 29171-20-8 C10H16O 152.236 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 6-(allyloxy)-2,6-dimethyloct-2-ene 1044135-68-3 C13H24O 196.333 —— dihydrolinalool acetate 50373-60-9 C12H22O2 198.305
反应信息
-
作为反应物:参考文献:名称:Escourrou, Bulletin de la Societe Chimique de France, 1928, vol. <4>43, p. 1108摘要:DOI:
-
作为产物:参考文献:名称:使用锰/水作为氢气源对不饱和化合物进行温和选择性氢化摘要:描述了使用锰/水温和且高选择性地还原烯烃和炔烃。高度受控的H 2生成允许在不稳定官能团存在下在温和且环境可接受的条件下选择性还原这些化合物。DOI:10.1021/acs.orglett.3c03664
文献信息
-
Lanthanum(III) Isopropoxide Catalyzed Chemoselective Transesterification of Dimethyl Carbonate and Methyl Carbamates作者:Manabu Hatano、Sho Kamiya、Katsuhiko Moriyama、Kazuaki IshiharaDOI:10.1021/ol102754y日期:2011.2.4carbonate and much less reactive methyl carbamates with primary (1°), secondary (2°), and tertiary (3°) alcohols was established with the use of a lanthanum(III) complex, which was prepared in situ from lanthanum(III) isopropoxide (3 mol %) and 2-(2-methoxyethoxy)ethanol (6 mol %). In particular, corresponding carbonates and carbamates obtained were of synthetic utility from the viewpoint of the selective protection
-
Fragrance compositions and compounds
-
Rethinking Basic Concepts—Hydrogenation of Alkenes Catalyzed by Bench-Stable Alkyl Mn(I) Complexes作者:Stefan Weber、Berthold Stöger、Luis F. Veiros、Karl KirchnerDOI:10.1021/acscatal.9b03963日期:2019.11.1intermediate which undergoes rapid hydrogenolysis to form the active 16e Mn(I) hydride catalyst [Mn(dippe)(CO)2(H)]. A range of mono- and disubstituted alkenes were efficiently converted into alkanes in good to excellent yields. The hydrogenation of 1-alkenes and 1,1-disubstituted alkenes proceeds at 25 °C, while 1,2-disubstituted alkenes require a reaction temperature of 60 °C. In all cases, a catalyst loading描述了一种有效的无添加剂锰催化的分子氢将烯烃氢化为烷烃的方法。该反应是原子经济的,实现了廉价的,富含地球的非贵金属催化剂。最有效的前催化剂是长凳稳定的烷基双膦Mn(I)配合物fac- [Mn(dippe)(CO)3(CH 2 CH 2 CH 3)]。催化过程是通过将CO配体迁移插入M n-烷基键中而产生的酰基中间体,该中间体经过快速氢解后形成活性16e Mn(I)氢化物催化剂[Mn(dippe)(CO)2(H)]。各种单取代和二取代的烯烃以良好或优异的收率有效地转化为烷烃。1-烯烃和1,1-二取代烯烃的氢化反应在25°C进行,而1,2-二取代烯烃的反应温度为60°C。在所有情况下,均施加2mol%的催化剂负载和50bar的氢气压力。提出了一种基于DFT计算的机制,并得到了初步的实验研究的支持。
-
Palladium nanoparticles in situ generated in metal–organic films for catalytic applications
-
Silicon–Carbon Bond Formation via Nickel-Catalyzed Cross-Coupling of Silicon Nucleophiles with Unactivated Secondary and Tertiary Alkyl Electrophiles作者:Crystal K. Chu、Yufan Liang、Gregory C. FuDOI:10.1021/jacs.6b03465日期:2016.5.25cross-coupling methods for the formation of C-C bonds from unactivated alkyl electrophiles have been described in recent years. In contrast, progress in the development of methods for the construction of C-heteroatom bonds has lagged; for example, there have been no reports of metal-catalyzed cross-couplings of unactivated secondary or tertiary alkyl halides with silicon nucleophiles to form C-Si bonds. In this
表征谱图
-
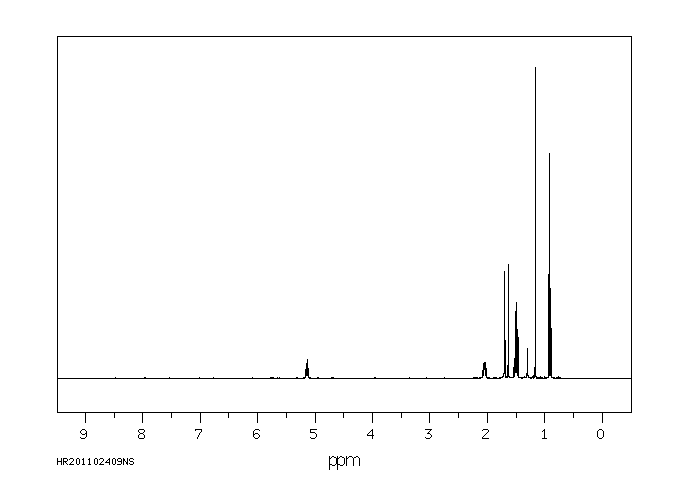
氢谱1HNMR
-
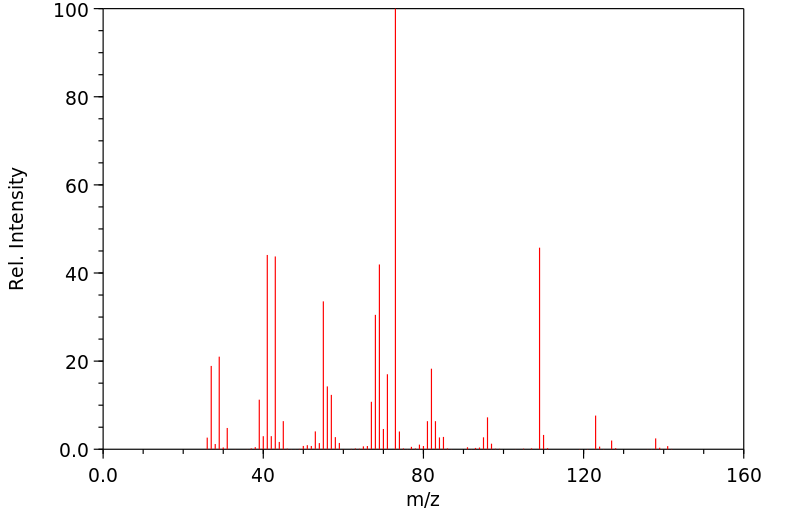
质谱MS
-
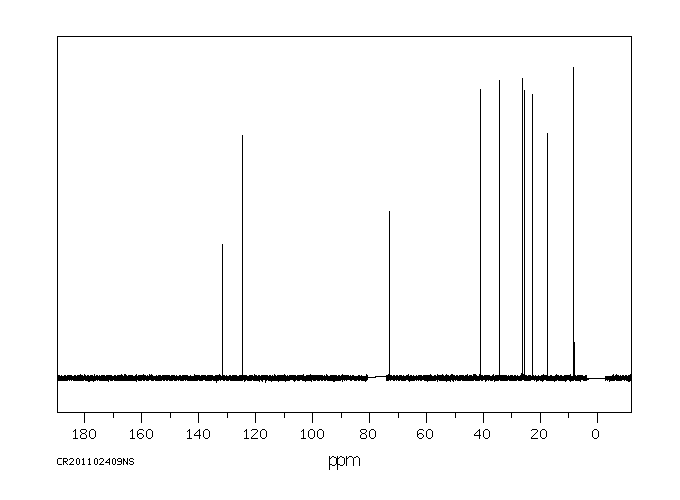
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(5β,6α,8α,10α,13α)-6-羟基-15-氧代黄-9(11),16-二烯-18-油酸
(3S,3aR,8aR)-3,8a-二羟基-5-异丙基-3,8-二甲基-2,3,3a,4,5,8a-六氢-1H-天青-6-酮
(2Z)-2-(羟甲基)丁-2-烯酸乙酯
(2S,4aR,6aR,7R,9S,10aS,10bR)-甲基9-(苯甲酰氧基)-2-(呋喃-3-基)-十二烷基-6a,10b-二甲基-4,10-dioxo-1H-苯并[f]异亚甲基-7-羧酸盐
(1aR,4E,7aS,8R,10aS,10bS)-8-[((二甲基氨基)甲基]-2,3,6,7,7a,8,10a,10b-八氢-1a,5-二甲基-氧杂壬酸[9,10]环癸[1,2-b]呋喃-9(1aH)-酮
(+)顺式,反式-脱落酸-d6
龙舌兰皂苷乙酯
龙脑香醇酮
龙脑烯醛
龙脑7-O-[Β-D-呋喃芹菜糖基-(1→6)]-Β-D-吡喃葡萄糖苷
龙牙楤木皂甙VII
龙吉甙元
齿孔醇
齐墩果醛
齐墩果酸苄酯
齐墩果酸甲酯
齐墩果酸溴乙酯
齐墩果酸二甲胺基乙酯
齐墩果酸乙酯
齐墩果酸3-O-alpha-L-吡喃鼠李糖基(1-3)-beta-D-吡喃木糖基(1-3)-alpha-L-吡喃鼠李糖基(1-2)-alpha-L-阿拉伯糖吡喃糖苷
齐墩果酸 beta-D-葡萄糖酯
齐墩果酸 beta-D-吡喃葡萄糖基酯
齐墩果酸 3-乙酸酯
齐墩果酸 3-O-beta-D-葡吡喃糖基 (1→2)-alpha-L-吡喃阿拉伯糖苷
齐墩果酸
齐墩果-12-烯-3b,6b-二醇
齐墩果-12-烯-3,24-二醇
齐墩果-12-烯-3,21,23-三醇,(3b,4b,21a)-(9CI)
齐墩果-12-烯-3,21,23-三醇,(3b,4b,21a)-(9CI)
齐墩果-12-烯-3,11-二酮
齐墩果-12-烯-2α,3β,28-三醇
齐墩果-12-烯-29-酸,3,22-二羟基-11-羰基-,g-内酯,(3b,20b,22b)-
齐墩果-12-烯-28-酸,3-[(6-脱氧-4-O-b-D-吡喃木糖基-a-L-吡喃鼠李糖基)氧代]-,(3b)-(9CI)
齐墩果-12-烯-28-酸,3,7-二羰基-(9CI)
齐墩果-12-烯-28-酸,3,21,29-三羟基-,g-内酯,(3b,20b,21b)-(9CI)
鼠特灵
鼠尾草酸醌
鼠尾草酸
鼠尾草酚酮
鼠尾草苦内脂
黑蚁素
黑蔓醇酯B
黑蔓醇酯A
黑蔓酮酯D
黑海常春藤皂苷A1
黑檀醇
黑果茜草萜 B
黑五味子酸
黏黴酮
黏帚霉酸