双硫氧吡啶 | 3696-28-4
中文名称
双硫氧吡啶
中文别名
奥麦丁二硫化物;双吡硫翁;2,2'-二硫代二(吡啶-1-氧化物);2,2'-二硫双(吡啶)-1.1'-二氧化物;二硫双(吡啶-N-氧化物);2,2’-二硫代二(吡啶-1-氧化物);双吡硫氧;2,2-二硫代双(吡啶-N-氧化物;双吡啶硫酮;2,2′-二甲基苯甲酰苯胺;2,2"-二硫代二(吡啶-1-氧化物)
英文名称
2,2'-dipyridyl disulfide bis-N-oxide
英文别名
2,2'-dithiobis(pyridine-N-oxide);dipyrithione;2,2′-dithiobis(pyridine-N-oxide);1-oxido-2-[(1-oxidopyridin-1-ium-2-yl)disulfanyl]pyridin-1-ium
CAS
3696-28-4
化学式
C10H8N2O2S2
mdl
MFCD00006197
分子量
252.318
InChiKey
ZHDBTKPXEJDTTQ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:200.5°C (rough estimate)
-
沸点:582.8±25.0 °C(Predicted)
-
密度:1.4423 (rough estimate)
-
溶解度:甲醇(微溶)、水(微溶、加热、超声处理)
计算性质
-
辛醇/水分配系数(LogP):0.6
-
重原子数:16
-
可旋转键数:3
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:102
-
氢给体数:0
-
氢受体数:4
安全信息
-
危险品运输编号:OTH
-
海关编码:2933399090
-
安全说明:S24/25
-
危险性防范说明:P261,P280,P301+P312,P302+P352,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:室温和干燥环境
SDS
| Name: | 2 2 -Dithiobis(Pyridine-N-Oxide) 97% Material Safety Data Sheet |
| Synonym: | Pyridine, 2,2'-Dithiobis-, 1,1'-Dioxide; Omadine Disulfide |
| CAS: | 3696-28-4 |
Synonym:Pyridine, 2,2'-Dithiobis-, 1,1'-Dioxide; Omadine Disulfide
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 3696-28-4 | Pyridine, 2,2'-Dithiobis-, 1,1'-Dioxid | 97 | 223-024-5 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Hygroscopic (absorbs moisture from the air).
Potential Health Effects
Eye:
Causes severe eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
Adverse reproductive effects have been reported in animals.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances. Store protected from moisture.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 3696-28-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: beige
Odor: none reported
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 205 deg C
Autoignition Temperature: Not applicable.
Flash Point: Not applicable.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C10H8N2O2S2
Molecular Weight: 252.31
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants, exposure to moist air or water.
Incompatibilities with Other Materials:
Moisture, strong oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, oxides of sulfur, irritating and toxic fumes and gases, carbon dioxide, hydrogen sulfide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 3696-28-4: UT2965000 LD50/LC50:
CAS# 3696-28-4: Draize test, rabbit, eye: 100 uL/24H Severe.
Carcinogenicity:
Pyridine, 2,2'-Dithiobis-, 1,1'-Dioxide - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 3696-28-4: No information available.
Canada
CAS# 3696-28-4 is listed on Canada's NDSL List.
CAS# 3696-28-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 3696-28-4 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 2,2’-disulfanediyldipyridine-1-oxide 132352-99-9 C10H8N2OS2 236.318 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2,2’-disulfanediyldipyridine-1-oxide 132352-99-9 C10H8N2OS2 236.318 —— phenyl 1-oxo-2-pyridyl disulfide 64826-51-3 C11H9NOS2 235.331 —— 4-chlorophenyl 1-oxo-pyridin-2-yl disulfide 64826-48-8 C11H8ClNOS2 269.776 —— 2-n-propyldithiopyridine-N-oxide 136852-78-3 C8H11NOS2 201.313 —— 2-carboxyethyl (1-oxo-2-pyridyl) disulfide 72632-25-8 C8H9NO3S2 231.296 —— 2-carbohydroxymethyldithiopyridine-N-oxide 72632-44-1 C7H7NO3S2 217.269
反应信息
-
作为反应物:参考文献:名称:使SF5组更易接近:芳烃四氟λ6-硫烷基氯的无气体试剂方法摘要:现代五氟硫烷基(SF 5)化学的致命弱点是:合成材料的可及性。本文中,我们介绍了解决芳基-SF 4 Cl化合物(最新的芳基-SF 5合成中的关键中间体)的第一种方法,该方法克服了对有害氟化试剂和/或气体试剂(例如Cl 2)的依赖使用易于处理的三氯异氰尿酸,氟化钾和催化量的酸。这些简单,温和的条件使您可以直接接触到尚未使用或无法通过以前的方法证明的芳基-SF 4 Cl中间体。此外,相同的方法提供了访问芳-SF 3和芳基- SEF 3化合物,将这种化学的应用范围扩展到了芳烃SF 5官能化以外,并证明了其解决更普遍的氧化氟化问题的能力。DOI:10.1002/anie.201812356
-
作为产物:描述:2-巯基吡啶-N-氧化物 在 Porcine Pancreas Lipase 作用下, 以 水 为溶剂, 反应 30.0h, 以75%的产率得到双硫氧吡啶参考文献:名称:二芳基二硫化物的生物催化合成及其作为耐药金黄色葡萄球菌抑制剂的生物评价。摘要:金黄色葡萄球菌是世界卫生组织(WHO)的II级优先病原体,具有引起急性至慢性感染和抵抗抗生素的能力,从而严重影响了全球医疗系统。在这种情况下,迫切需要发现新的分子以阻止持续出现的抗药性。含有二硫化物的小分子作为抗菌剂已受到关注。由于它们的常规合成需要繁琐的合成程序,有时还需要有毒的试剂,因此已开发出一种绿色环保的合成方案,可通过该方案获得一系列分子并评估其对ESKAPE病原体的抗菌活性。测试了该化合物对Vero细胞的细胞毒性,以确定其选择性指数并确定了时间杀灭动力学。金黄色葡萄球菌多药耐药临床分离株。此外,还测试了其与FDA批准药物协同作用的能力以及减少生物膜的能力。我们确定双(2-溴苯基)二硫化物(2t)具有对金黄色葡萄球菌(包括MRSA和VRSA菌株)的等效抗菌活性。此外,2t的选择性指数为25,具有浓度依赖性的杀菌活性,与所有测试的药物均具有协同作用,并显着减少了预先形成的生物膜。综DOI:10.1002/ddr.21507
-
作为试剂:描述:4-(甲氧羰基)双环[2.2.2]辛烷-1-羧酸 在 盐酸 、 lithium aluminium tetrahydride 、 草酰氯 、 三丁基膦 、 双硫氧吡啶 、 水 、 氢气 、 palladium diacetate 、 potassium carbonate 、 二甲基亚砜 、 sodium hydroxide 、 四甲基胍 作用下, 以 四氢呋喃 、 1,4-二氧六环 、 甲醇 、 二氯甲烷 、 水 、 乙酸乙酯 为溶剂, -70.0~20.0 ℃ 、607.99 kPa 条件下, 反应 39.25h, 生成 (S)-2-(((benzyloxy)carbonyl)amino)-3-(bicyclo[2.2.2]octan-1-yl)propanoic acid 、 (R)-2-(((benzyloxy)carbonyl)amino)-3-(bicyclo[2.2.2]octan-1-yl)propanoic acid参考文献:名称:WO2020131282A5摘要:公开号:WO2020131282A5
文献信息
-
[EN] MICROBIOCIDAL OXADIAZOLE DERIVATIVES<br/>[FR] DÉRIVÉS D'OXADIAZOLE MICROBIOCIDES申请人:SYNGENTA PARTICIPATIONS AG公开号:WO2017157962A1公开(公告)日:2017-09-21Compounds of the formula (I) wherein the substituents are as defined in claim 1, useful as a pesticides, especially fungicides.式(I)的化合物,其中取代基如权利要求1所定义,作为杀虫剂特别是杀菌剂有用。
-
Thieno-pyrimidine compounds having fungicidal activity
-
[EN] INSECTICIDAL TRIAZINONE DERIVATIVES<br/>[FR] DÉRIVÉS DE TRIAZINONE INSECTICIDES申请人:SYNGENTA PARTICIPATIONS AG公开号:WO2013079350A1公开(公告)日:2013-06-06Compounds of the formula (I) or (I'), wherein the substituents are as defined in claim 1, are useful as pesticides.式(I)或(I')的化合物,其中取代基如权利要求1所定义的那样,可用作杀虫剂。
-
[EN] IMIDAZOLIUM REAGENT FOR MASS SPECTROMETRY<br/>[FR] RÉACTIF D'IMIDAZOLIUM POUR SPECTROMÉTRIE DE MASSE申请人:HOFFMANN LA ROCHE公开号:WO2021234004A1公开(公告)日:2021-11-25The present invention relates to compounds which are suitable to be used in mass spectrometry as well as methods of mass spectrometric determination of analyte molecules using said compounds.本发明涉及适用于质谱的化合物,以及利用该化合物进行分析物分子的质谱测定方法。
-
[EN] SYNTHETIC OLIGOGLUCOSAMINES FOR IMPROVEMENT OF PLANT GROWTH AND YIELD<br/>[FR] OLIGOGLUCOSAMINES SYNTHÉTIQUES POUR L'AMÉLIORATION DE LA CROISSANCE ET DU RENDEMENT DE VÉGÉTAUX申请人:DU PONT公开号:WO2015130893A1公开(公告)日:2015-09-03The disclosure provides formulations comprising synthetic oligoglucosamines and methods for improving plant growth and crop yield therewith. These formulations may be applied to propagating materials, including seeds and other regenerable plant parts, including cuttings, bulbs, rhizomes and tubers. They may also be applied to foliage, or soil either prior to or following planting of propagating materials. Such applications may be made alone or in combination with fungicides, insecticides, nematicides and other agricultural agents used to improve plant growth and crop yield.
表征谱图
-
氢谱1HNMR
-
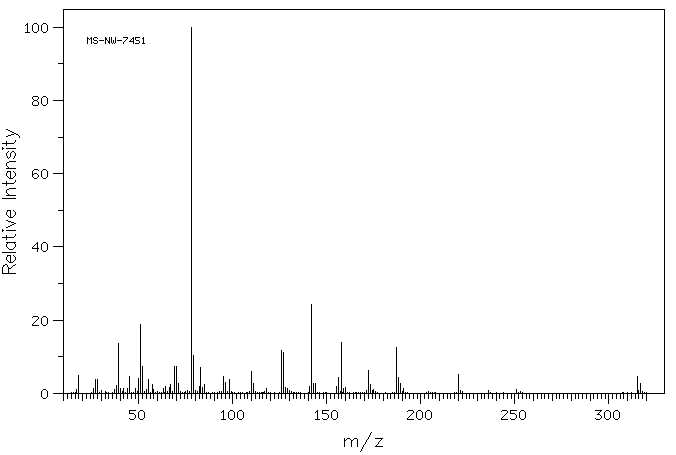
质谱MS
-
碳谱13CNMR
-
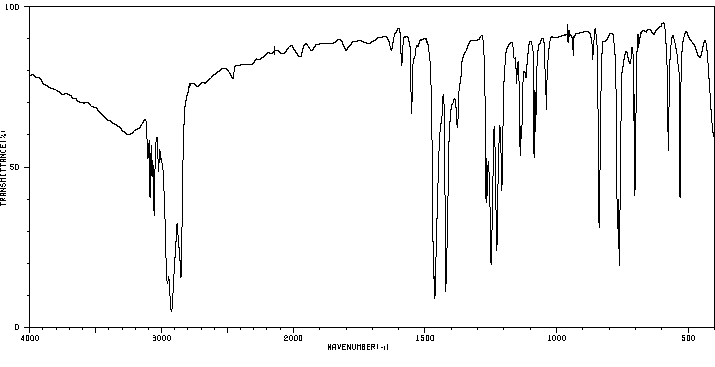
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-氨氯地平-d4
(R,S)-可替宁N-氧化物-甲基-d3
(R)-(+)-2,2'',6,6''-四甲氧基-4,4''-双(二苯基膦基)-3,3''-联吡啶(1,5-环辛二烯)铑(I)四氟硼酸盐
(R)-N'-亚硝基尼古丁
(R)-DRF053二盐酸盐
(5E)-5-[(2,5-二甲基-1-吡啶-3-基-吡咯-3-基)亚甲基]-2-亚磺酰基-1,3-噻唑烷-4-酮
(5-溴-3-吡啶基)[4-(1-吡咯烷基)-1-哌啶基]甲酮
(5-氨基-6-氰基-7-甲基[1,2]噻唑并[4,5-b]吡啶-3-甲酰胺)
(2S,2'S)-(-)-[N,N'-双(2-吡啶基甲基]-2,2'-联吡咯烷双(乙腈)铁(II)六氟锑酸盐
(2S)-2-[[[9-丙-2-基-6-[(4-吡啶-2-基苯基)甲基氨基]嘌呤-2-基]氨基]丁-1-醇
(2R,2''R)-(+)-[N,N''-双(2-吡啶基甲基)]-2,2''-联吡咯烷四盐酸盐
(1'R,2'S)-尼古丁1,1'-Di-N-氧化物
黄色素-37
麦斯明-D4
麦司明
麝香吡啶
鲁非罗尼
鲁卡他胺
高氯酸N-甲基甲基吡啶正离子
高氯酸,吡啶
高奎宁酸
马来酸溴苯那敏
马来酸氯苯那敏-D6
马来酸左氨氯地平
顺式-双(异硫氰基)(2,2'-联吡啶基-4,4'-二羧基)(4,4'-二-壬基-2'-联吡啶基)钌(II)
顺式-二氯二(4-氯吡啶)铂
顺式-二(2,2'-联吡啶)二氯铬氯化物
顺式-1-(4-甲氧基苄基)-3-羟基-5-(3-吡啶)-2-吡咯烷酮
顺-双(2,2-二吡啶)二氯化钌(II) 水合物
顺-双(2,2'-二吡啶基)二氯化钌(II)二水合物
顺-二氯二(吡啶)铂(II)
顺-二(2,2'-联吡啶)二氯化钌(II)二水合物
韦德伊斯试剂
非那吡啶
非洛地平杂质C
非洛地平
非戈替尼
非布索坦杂质66
非尼拉朵
非尼拉敏
雷索替丁
阿雷地平
阿瑞洛莫
阿扎那韦中间体
阿培利司N-6
阿伐曲波帕杂质40
间硝苯地平
间-硝苯地平
镉,二碘四(4-甲基吡啶)-
锌,二溴二[4-吡啶羧硫代酸(2-吡啶基亚甲基)酰肼]-