反-1,2-二苯酰乙烯 | 959-28-4
中文名称
反-1,2-二苯酰乙烯
中文别名
反-1,2-二苯甲酰乙烯;反式-1,4-二苯基-2-丁烯-1,4-二酮;反-1,4-二苯基-2-丁烯-1,4-二酮;1,2-联苯甲酰乙烯
英文名称
1,4-diphenylbut-2-ene-1,4-dione
英文别名
trans-1,2-dibenzoylethylene;(E)-1,4-diphenylbut-2-ene-1,4-dione
CAS
959-28-4
化学式
C16H12O2
mdl
——
分子量
236.27
InChiKey
WYCXGQSQHAXLPK-VAWYXSNFSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:109-112 °C(lit.)
-
沸点:338.73°C (rough estimate)
-
密度:1.1223 (rough estimate)
-
稳定性/保质期:
在常温常压下保持稳定,应避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):3.2
-
重原子数:18
-
可旋转键数:4
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:34.1
-
氢给体数:0
-
氢受体数:2
安全信息
-
TSCA:Yes
-
安全说明:S22,S24/25
-
WGK Germany:3
-
海关编码:29143900
-
RTECS号:EM7000000
-
储存条件:应储存在冷干燥环境中,并放置于通风良好、远离加热源及不相容物质的地方,密封保存。
SDS
| Name: | Trans-1 2-Dibenzoylethylene 97% Material Safety Data Sheet |
| Synonym: | 1, 4-Diphenyl-2-Butene-1, 4 Dione |
| CAS: | 959-28-4 |
Synonym:1, 4-Diphenyl-2-Butene-1, 4 Dione
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 959-28-4 | 2-Butene-1,4-Dione, 1,4-Diphenyl-, (e) | 97 | 213-498-1 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation. The toxicological properties of this material have not been fully investigated.
Skin:
May cause skin irritation. The toxicological properties of this material have not been fully investigated.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 959-28-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Crystalline powder
Color: yellow
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 108.00 - 111.00 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C16H12O2
Molecular Weight: 236.27
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 959-28-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-Butene-1,4-Dione, 1,4-Diphenyl-, (e)- - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 959-28-4: No information available.
Canada
CAS# 959-28-4 is listed on Canada's NDSL List.
CAS# 959-28-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 959-28-4 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 (Z)-1,4-二苯基-2-丁烯-1,4-二酮 (Z)-1,4-diphenylbut-2-ene-1,4-dione 959-27-3 C16H12O2 236.27 1-苯基-2-丙烯基-1-酮 PVK 768-03-6 C9H8O 132.162 苯乙酮 acetophenone 98-86-2 C8H8O 120.151 —— (3E)-1,4-diphenylbut-3-en-1-one —— C16H14O 222.287 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 (Z)-1,4-二苯基-2-丁烯-1,4-二酮 (Z)-1,4-diphenylbut-2-ene-1,4-dione 959-27-3 C16H12O2 236.27 —— (E)-4-hydroxy-1,4-diphenyl-2-buten-1-one 300346-82-1 C16H14O2 238.286 —— (Z)-2-amino-1,4-diphenyl-2-butene-1,4-dione 58940-87-7 C16H13NO2 251.285 (E)-2-氨基-1,4-二苯基-丁-2-烯-1,4-二酮 1-amino-1,2-dibenzoylethylene 43013-73-6 C16H13NO2 251.285 —— (Z)-2-(methylthio)-1,4-diphenylbut-2-ene-1,4-dione —— C17H14O2S 282.363 —— 2-(methylthio)-1,4-diphenylbut-2-ene-1,4-dione 63113-56-4 C17H14O2S 282.363 —— 1-Methoxy-1,2-dibenzoyl-ethylen 33193-65-6 C17H14O3 266.296
反应信息
-
作为反应物:描述:参考文献:名称:A New Synthesis of 1,3-Diarylisobenzofurans摘要:DOI:10.1055/s-1980-28946
-
作为产物:描述:参考文献:名称:Diazoketones as Reagents for the Identification of Organic Acids摘要:DOI:10.1021/ja01155a086
文献信息
-
Proton-Coupled Electron Transfer: Transition-Metal-Free Selective Reduction of Chalcones and Alkynes Using Xanthate/Formic Acid作者:Ramanathan Prasanna、Somraj Guha、Govindasamy SekarDOI:10.1021/acs.orglett.9b00635日期:2019.4.19transfer (PCET). Mechanistic experiments and DFT calculations support the possibility of a concerted proton electron-transfer (CPET) pathway. This Birch-type reduction demonstrates that a small nucleophilic organic molecule can be used as a single electron-transfer (SET) reducing agent with a proper proton source.
-
AlCl<sub>3</sub>-Promoted Conjugate Reduction of α,β Unsaturated Carbonyl Compounds with 1,3-Dimethyl-2-phenylbenzimidazoline作者:Hidenori Chikashita、Kazuyoshi ItohDOI:10.1246/bcsj.59.1747日期:1986.6substrates. The catalytic efficiency of Lewis acids was found to be proportional to the efficiency to form a complex with a carbonyl group. The reduction of 2-cinnamoylpyridine with 2-deuterated DMBI revealed that in the reduction product, a deuterium atom was located at the β-position with respect to the carbonyl group. On the other hand, the reduction of the same substrate with DMBI followed by quenching
-
A Broad‐Spectrum Synthesis of Tetravinylethylenes作者:Kelsey L. Horvath、Christopher G. Newton、Kimberley A. Roper、Jas S. Ward、Michael S. SherburnDOI:10.1002/chem.201900550日期:2019.3.15compounds of the tetravinylethylene (TVE) family is reported. Ramirez‐type dibromo‐olefination of readily accessible penta‐1,4‐dien‐3‐ones generates 3,3‐dibromo[3]dendralenes, which undergo twofold Negishi, Suzuki–Miyaura or Mizoroki–Heck reactions with a wide variety of olefinic coupling partners. This route delivers a broad range of unsymmetrically substituted tetravinylethylenes with up to three
-
Organocatalyzed Enantioselective Michael Addition of 2‐Hydroxypyridines and α,β‐Unsaturated 1,4‐Dicarbonyl Compounds作者:Yu‐Chun Wu、Yi Jhong、Hui‐Jie Lin、Sharada Prasanna Swain、Hui‐Hsu Gavin Tsai、Duen‐Ren HouDOI:10.1002/adsc.201900997日期:2019.11.5prevalent in biologically and medicinally important molecules. Here we report that chiral N‐substituted 2‐pyridones were prepared by enantioselective, organocatalytic aza‐Michael additions of halogenated 2‐hydroxypyridines (pyridin‐2(1H)‐ones) to α,β‐unsaturated‐1,4‐dketones or 1,4‐ketoesters. The reactions were optimized by the choice of solvents and systematic screening of Cinchona alkaloid‐based bifunctional
-
Reduction of<i>α</i>-Halo Carbonyl Compounds with NaBH<sub>4</sub>-SbBr<sub>3</sub>
表征谱图
-
氢谱1HNMR
-
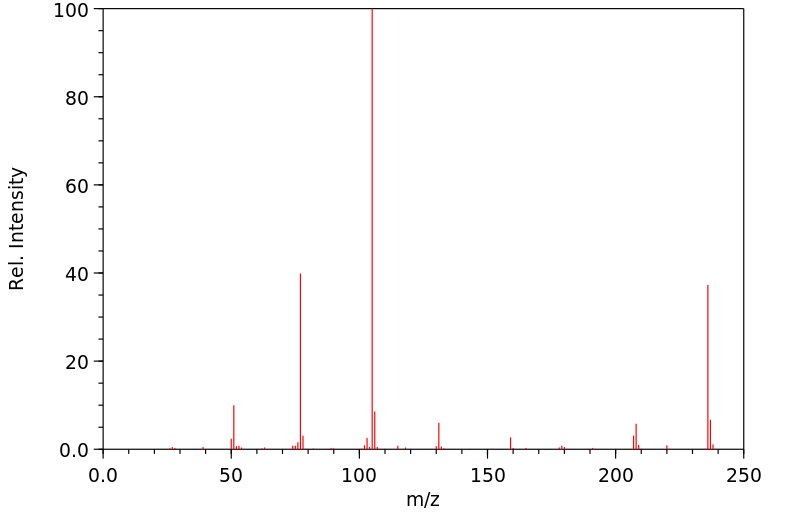
质谱MS
-
碳谱13CNMR
-
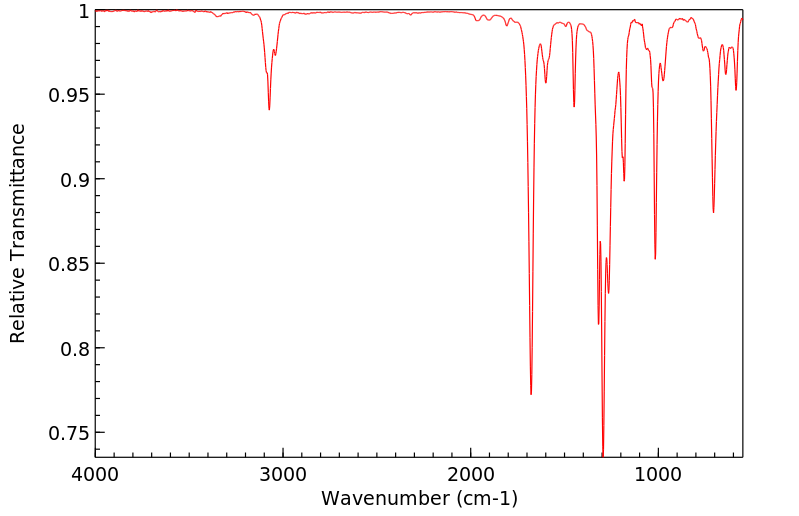
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫