2-乙氧基-3-异丙基吡嗪 | 72797-16-1
中文名称
2-乙氧基-3-异丙基吡嗪
中文别名
——
英文名称
2-Ethoxy-3-isopropylpyrazine
英文别名
2-ethoxy-3-propan-2-ylpyrazine
CAS
72797-16-1
化学式
C9H14N2O
mdl
MFCD00038029
分子量
166.22
InChiKey
LTAUBPVQMBOANV-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
反应信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:230.5±40.0 °C(Predicted)
-
密度:0.99
-
LogP:2.62
-
物理描述:Clear colourless to yellow liquid; Slightly roasted nut aroma
-
溶解度:Slightly soluble in water
-
折光率:1.500-1.510
-
保留指数:1143
-
稳定性/保质期:
正常条件下稳定,远离氧化物。
计算性质
-
辛醇/水分配系数(LogP):2.6
-
重原子数:12
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.555
-
拓扑面积:35
-
氢给体数:0
-
氢受体数:3
安全信息
-
危险品标志:Xn
-
危险类别码:R22
-
海关编码:2933990090
文献信息
-
BITTER TASTE MODIFIERS INCLUDING SUBSTITUTED 1-BENZYL-3-(1-(ISOXAZOL-4-YLMETHYL)-1H-PYRAZOL-4-YL)IMIDAZOLIDINE-2,4-DIONES AND COMPOSITIONS THEREOF申请人:SENOMYX, INC.公开号:US20160376263A1公开(公告)日:2016-12-29The present invention includes compounds and compositions known to modify the perception of bitter taste, and combinations of said compositions and compounds with additional compositions, compounds, and products. Exemplary compositions comprise one or more of the following: cooling agents; inactive drug ingredients; active pharmaceutical ingredients; food additives or foodstuffs; flavorants, or flavor enhancers; food or beverage products; bitter compounds; sweeteners; bitterants; sour flavorants; salty flavorants; umami flavorants; plant or animal products; compounds known to be used in pet care products; compounds known to be used in personal care products; compounds known to be used in home products; pharmaceutical preparations; topical preparations; cannabis-derived or cannabis-related products; compounds known to be used in oral care products; beverages; scents, perfumes, or odorants; compounds known to be used in consumer products; silicone compounds; abrasives; surfactants; warming agents; smoking articles; fats, oils, or emulsions; and/or probiotic bacteria or supplements.本发明涵盖已知用于改变苦味感知的化合物和组合物,以及所述组合物和化合物与额外的组合物、化合物和产品的组合。示例组合物包括以下一种或多种:冷却剂;无活性药物成分;活性药用成分;食品添加剂或食品;调味剂或调味增强剂;食品或饮料产品;苦味化合物;甜味剂;苦味剂;酸味调味剂;咸味调味剂;鲜味调味剂;植物或动物产品;已知用于宠物护理产品中的化合物;已知用于个人护理产品中的化合物;已知用于家用产品中的化合物;制药制剂;局部制剂;大麻衍生或与大麻相关的产品;已知用于口腔护理产品中的化合物;饮料;香味、香水或除臭剂;已知用于消费品中的化合物;硅化合物;磨料;表面活性剂;发热剂;吸烟物品;脂肪、油脂或乳化剂;和/或益生菌或补充剂。
-
COMPOUNDS USEFUL AS MODULATORS OF TRPM8申请人:Senomyx, Inc.公开号:US20170096418A1公开(公告)日:2017-04-06The present disclosure relates to compounds which are useful as cooling sensation compounds.本公开涉及作为冷感化合物有用的化合物。
-
[EN] STEVIOL GLYCOSIDE ESTERS<br/>[FR] ESTERS DE GLYCOSIDES DE STÉVIOL申请人:CHROMOCELL CORP公开号:WO2017156432A1公开(公告)日:2017-09-14The present disclosure provides edible compositions comprising Formulas and compounds of steviol glycoside esters, or combinations thereof, which are suitable for use as sweeteners, for example, by incorporation into edible compositions. Also provided herein are methods of preparing steviol glycoside ester compounds.本公开提供了包含史蒂维奥甙醇酯化合物的配方或其组合的可食用组合物,这些组合物适用于作为甜味剂,例如通过将其纳入可食用组合物中。本文还提供了制备史蒂维奥甙醇酯化合物的方法。
-
7-Substituted bicyclic pyrazolidinones申请人:ELI LILLY AND COMPANY公开号:EP0202046A1公开(公告)日:1986-11-207-Substituted bicyclic pyrazolidinone compounds as antimicrobials and the corresponding intermediates, are discussed or provided. The use of the antimicrobial compounds in pharmaceutical compositions and in methods for treating bacterial infections is set forth. The pyrazolidinone compounds are of the formula wherein one of R1 and R2 is hydrogen, halo, C1 to C6 alkyl, C1 to C6 substituted alkyl, perfluoro C2 to C4 alkyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl, a heterocyclic ring, nitro or cyano; a group of the formula wherein X is fluoro, chloro, bromo or iodo; a group of the formula wherein z is 0, 1 or 2 and R7 is C1 to C6 alkyl, C1 to C6 substituted alkyl, phenyl, substituted phenyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl or a heterocyclic ring; a group of the formula wherein R8 is hydrogen, C1 to C6 alkyl, C, to C6 substituted alkyl, perfluoro C2 to C4 alkyl, trihalomethyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl, amino, (monosubstituted)amino, or (disubstituted)amino; a group of the formula wherein R9 is hydrogen, an organic or inorganic cation, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl, a carboxy-protecting group, or a non-toxic, metabolically-labile, ester-forming group; a group of the formula wherein R10 is hydrogen, an organic or inorganic cation, C1 to C5 alkyl, C, to G6 aubatituted allcyl, C7 to C12 arylalkyl, C7 to C12 aubatltuted arylalkyl, phenyl, or substituted phenyl; a group of the formula ⊕ wherein -N≡Q is a quaternary ammonium group; or a group of the formula -CH2-S-Heterocycllc ring; a group of the formula wherein R11 is hydrogen, C1 to Ce alkyl, C, to Ce substituted alkyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl or C1 to C7 acyl; or a group of the formula wherein R12 and R13 are the same or different and are hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl, C1 to C7 acyl, or a group of the formula; wherein Rq is C, to C6 alkyl, C7 to C12 arylalkyl or phenyl; and the other of R, and R2 is a group of the formula wherein R14 is hydrogen, an organic or inorganic cation, a carboxy-protecting group or a non-toxic, metabolically-labile ester-forming group; and R3 and R4 are the same or different and are hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 arylalkyl, C7 to C12 substituted arylalkyl, phenyl, substituted phenyl or a group of the formula wherein R15 has the same definition as R9; R5 and R6 are: 1) each hydrogen, an amino protecting group; or acyl group derived from a C, to C30 carboxylic acid; provided that at least one of Rs and R6 must be hydrogen; or 2) taken together to form a phthalimido group; or a pharmaceutically-acceptable salt thereof.讨论或提供了作为抗菌剂的 7-取代双环吡唑烷酮化合物及相应的中间体。阐述了抗菌化合物在药物组合物和治疗细菌感染方法中的用途。吡唑烷酮化合物的结构式如下 其中 R1 和 R2 之一为氢、卤素、C1 至 C6 烷基、C1 至 C6 取代烷基、全氟 C2 至 C4 烷基、C7 至 C12 芳烷基、C7 至 C12 取代芳烷基、苯基、取代苯基、杂环、硝基或氰基; 式中的基团 其中 X 是氟、氯、溴或碘; 式中的基团 其中 z 是 0、1 或 2,R7 是 C1 至 C6 烷基、C1 至 C6 取代的烷基、苯基、取代的苯基、C7 至 C12 芳烷基、C7 至 C12 取代的芳烷基或杂环; 式中的基团 其中 R8 是氢、C1 至 C6 烷基、C, 至 C6 取代的烷基、全氟 C2 至 C4 烷基、三卤甲基、C7 至 C12 芳烷基、C7 至 C12 取代的芳烷基、苯基、取代的苯基、氨基、(单取代)氨基或(二取代)氨基; 式中的基团 其中 R9 是氢、有机或无机阳离子、C1~C6 烷基、C1~C6 取代的烷基、C7~C12 芳烷基、C7~C12 取代的芳烷基、苯基、取代的苯基、羧基保护基团或无毒、代谢上易腐烂的酯形成基团; 式中的基团 其中 R10 是氢、有机或无机阳离子、C1 至 C5 烷基、C, 至 G6 亚氨基全烷基、C7 至 C12 芳烷基、C7 至 C12 亚氨基芳烷基、苯基或取代苯基; 式中的基团 ⊕ 其中-N≡Q 是季铵基团;或式中-CH2-S-杂环的基团; 式中的基团 其中 R11 是氢、C1 至 Ce 烷基、C1 至 Ce 取代的烷基、C7 至 C12 芳烷基、C7 至 C12 取代的芳烷基、苯基、取代的苯基或 C1 至 C7酰基;或式中的基团 其中 R12 和 R13 相同或不同,并且是氢、C1 至 C6 烷基、C1 至 C6 取代的烷基、C7 至 C12 芳烷基、C7 至 C12 取代的芳烷基、苯基、取代的苯基、C1 至 C7酰基或式中的基团; 其中 Rq 是 C 至 C6 烷基、C7 至 C12 芳烷基或苯基;以及 R 和 R2 中的另一个是式中的基团 其中 R14 是氢、有机或无机阳离子、羧基保护基团或无毒、可代谢的成酯基团; R3 和 R4 相同或不同,并且是氢、C1 至 C6 烷基、C1 至 C6 取代的烷基、C7 至 C12 芳烷基、C7 至 C12 取代的芳烷基、苯基、取代的苯基或式中的一个基团。 其中 R15 的定义与 R9 相同; R5 和 R6 为 1) 各为氢、氨基保护基;或由 C 至 C30 羧酸衍生的酰基; 但 Rs 和 R6 中至少有一个必须是氢;或 2) 结合在一起形成邻苯二甲酰亚胺基团;或其药学上可接受的盐。
-
Bicyclic pyrazolidinones申请人:ELI LILLY AND COMPANY公开号:EP0203722A1公开(公告)日:1986-12-03Bicyclic pyrazolidinones which have antimicrobial and/or herbicidal properties are discussed. The use of these compounds in pharmaceutical compositions, herbicidal compositions, and methods for treating bacterial infections and controlling undesired plants is set forth. The bicyclic pyrazolidinones have the formula wherein: either R1 or R2 is hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, perfluoro C2 to C4 alkyl, C7 to C12 alkylaryl, C7 to C12 substituted alkylaryl, phenyl, substituted phenyl, nitro br cyano; a group of the formula wherein X is fluoro, chloro, bromo or iodo; a group of the formula wherein Z is 0, 1 or 2 and Rs is C1 to C6 alkyl, C1 to C6 substituted alkyl, phenyl, substituted phenyl, C7 to C12 alkylaryl, C7 to C12 substituted alkylaryl or a heterocyclic ring; a group of the formula wherein R6 is hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, perfluoro C2 to C4 alkyl, trihalomethyl, C7 to C12 alkylaryl, C7 to C12 substituted alkylaryl, phenyl or substituted phenyl; a group of the formula wherein R7 is hydrogen, an organic or inorganic cation, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 alkylaryl, C7 to C12 substituted alkylaryl, phenyl, substituted phenyl, a carboxy protecting group, or a non-toxic, metabolically-labile ester-forming group; or a group of the formula wherein R8 is hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 substituted alkylaryl, phenyl, or substituted phenyl; a group of the formula wherein -N≡Q is a quaternary ammonium group; or a group of the formula -CH2-S-Heterocyclic-ring and the other of R1 or R2 is a group of the formula wherein R9 is hydrogen, an organic or inorganic cation, a carboxy protecting group, or a non-toxic, metabolically-labile ester-forming group; and R3 and R4 are the same or different and are hydrogen, C1 to C6 alkyl, C1 to C6 substituted alkyl, C7 to C12 alkylaryl, C7 to C12 substituted alkylaryl, phenyl, substituted phenyl or a group of the formula wherein R10 has the same definition as R7; with the exception that, when R1 and R2 are a group of the formula R3 and R4 are not methyl; or a pharmaceutically-acceptable salt thereof.本文讨论了具有抗菌和/或除草特性的双环吡唑烷酮。阐述了这些化合物在药物组合物、除草组合物中的用途,以及治疗细菌感染和控制有害植物的方法。双环吡唑烷酮的化学式为 其中 R1 或 R2 是氢、C1 至 C6 烷基、C1 至 C6 取代烷基、全氟 C2 至 C4 烷基、C7 至 C12 烷芳基、C7 至 C12 取代烷芳基、苯基、取代苯基、硝基溴氰基; 式中的基团 其中 X 为氟、氯、溴或碘; 式中的基团 其中 Z 是 0、1 或 2,Rs 是 C1 至 C6 烷基、C1 至 C6 取代的烷基、苯基、取代的苯基、C7 至 C12 烷芳基、C7 至 C12 取代的烷芳基或杂环; 式中的基团 其中 R6 是氢、C1 至 C6 烷基、C1 至 C6 取代的烷基、全氟 C2 至 C4 烷基、三卤甲基、C7 至 C12 烷芳基、C7 至 C12 取代的烷芳基、苯基或取代的苯基; 式中的基团 其中 R7 是氢、有机或无机阳离子、C1-C6 烷基、C1-C6 取代烷基、C7-C12 烷芳基、C7-C12 取代烷芳基、苯基、取代苯基、羧基保护基团或无毒、代谢上易形成酯的基团; 或一个式中的基团 其中 R8 是氢、C1 至 C6 烷基、C1 至 C6 取代的烷基、C7 至 C12 取代的烷芳基、苯基或取代的苯基; 式中的基团 其中 -N≡Q 是季铵基团; 或式中的基团 -CH2-S-杂环,而 R1 或 R2 中的另一个是式中的基团 其中 R9 是氢、有机或无机阳离子、羧基保护基团或无毒、可代谢的成酯基团;以及 R3 和 R4 相同或不同,并且是氢、C1 至 C6 烷基、C1 至 C6 取代的烷基、C7 至 C12 烷芳基、C7 至 C12 取代的烷芳基、苯基、取代的苯基或式中的基团 其中 R10 的定义与 R7 相同;但当 R1 和 R2 为式中的基团时 R3 和 R4 不是甲基;或其药学上可接受的盐。
表征谱图
-
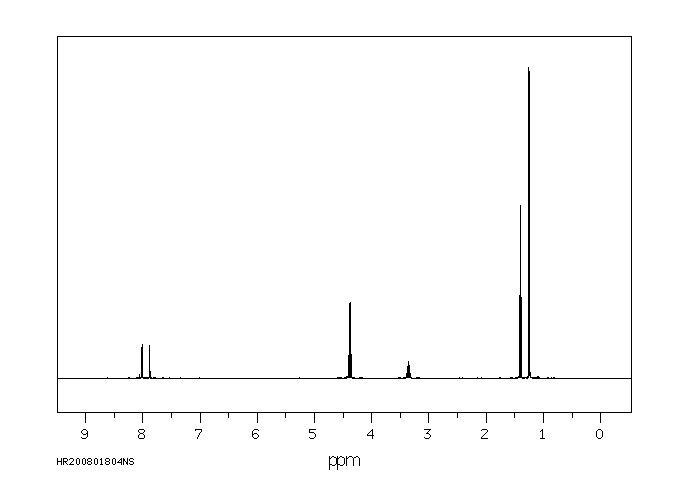
氢谱1HNMR
-
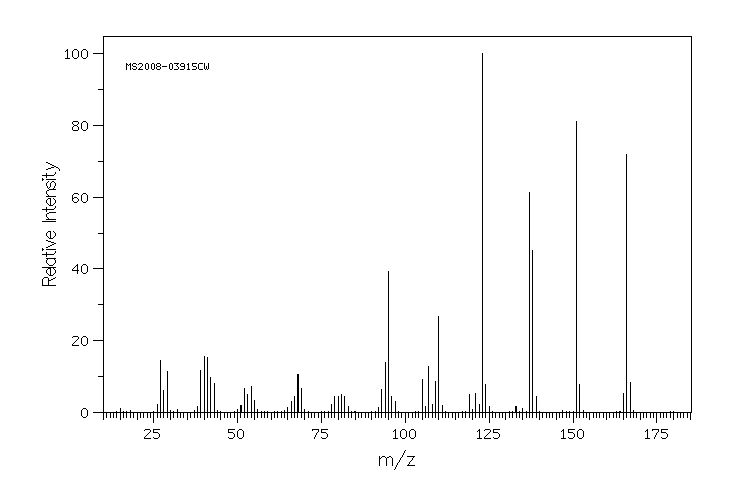
质谱MS
-
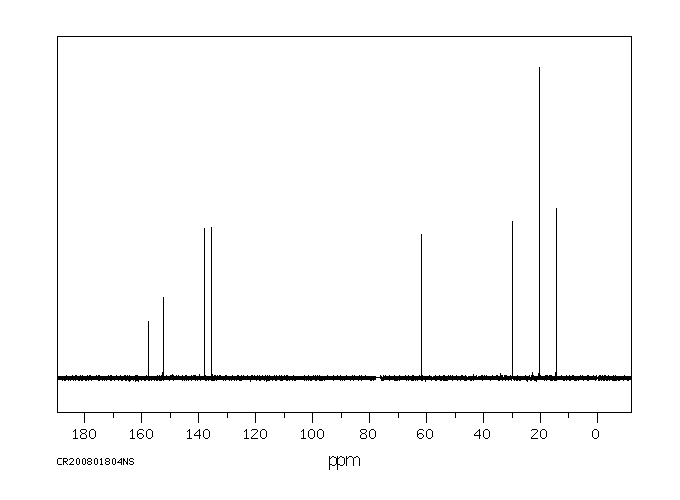
碳谱13CNMR
-
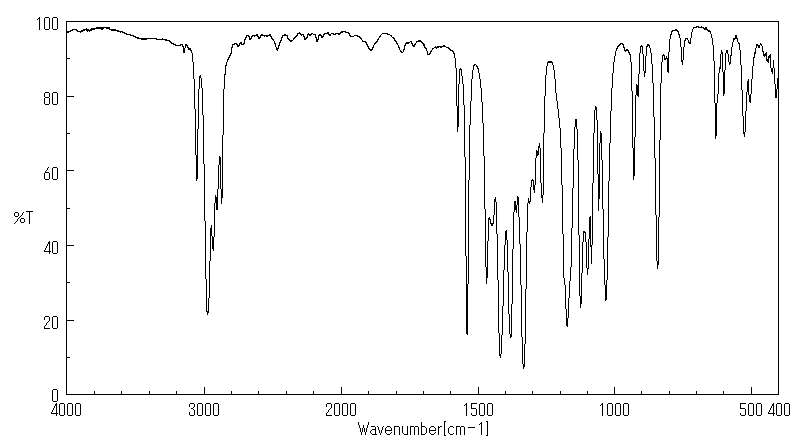
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷