3,5-二(三氟甲基)苯-1-碳酰肼 | 26107-82-4
中文名称
3,5-二(三氟甲基)苯-1-碳酰肼
中文别名
3,5-二(三氟甲基)苯甲酰肼;3,5-双(三氟甲基)苯酰肼;3,5-二(三氟甲基)苯酰肼
英文名称
3,5-bis(trifluoromethyl)benzohydrazide
英文别名
3,5-bis(trifluoromethyl)benzhydrazide;3,5-bis(trifluoromethyl)benzoyl hydrazide;3,5-bis(trifluoromethyl)benzoylhydrazine
CAS
26107-82-4
化学式
C9H6F6N2O
mdl
MFCD00051848
分子量
272.15
InChiKey
GBBRFBSFWKFTMZ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:131-135 °C
-
密度:1.479±0.06 g/cm3(Predicted)
-
稳定性/保质期:
如果按照规格使用和储存,则不会分解。请避免接触氧化物。
计算性质
-
辛醇/水分配系数(LogP):2.2
-
重原子数:18
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.222
-
拓扑面积:55.1
-
氢给体数:2
-
氢受体数:8
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
危险类别码:R36/37/38
-
海关编码:2928000090
-
安全说明:S26,S37/39
-
储存条件:保持贮藏器密封,并将其放入一个紧密封装的容器中。建议存放在阴凉、干燥处。
SDS
| Name: | 3 5-Bis(trifluoromethyl)benzhydrazide Material Safety Data Sheet |
| Synonym: | |
| CAS: | 26107-82-4 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 26107-82-4 | 3,5-Bis(trifluoromethyl)benzhydrazide | 247-462-1 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Avoid generating dusty conditions.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 26107-82-4: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: Not available.
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 132 - 134 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C9H6F6N2O
Molecular Weight: 272.15
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents, strong bases.
Hazardous Decomposition Products:
Hydrogen cyanide, carbon monoxide, oxides of nitrogen, carbon dioxide, hydrogen fluoride gas, nitric acid.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 26107-82-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
3,5-Bis(trifluoromethyl)benzhydrazide - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 37/39 Wear suitable gloves and eye/face
protection.
WGK (Water Danger/Protection)
CAS# 26107-82-4: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 26107-82-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 26107-82-4 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3,5-双三氟甲基苯甲酸 3,5-bistrifluoromethylbenzoic acid 725-89-3 C9H4F6O2 258.12 3,5-双三氟甲基苯甲酰氯 3,5-bis(trifluoromethyl)phenyl carboxylic acid chloride 785-56-8 C9H3ClF6O 276.566 3,5-双(三氟甲基)苯甲酸甲酯 methyl 3,5-bis(trifluoromethyl)benzoate 26107-80-2 C10H6F6O2 272.147 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— N'-(but-3-enyl)-3,5-bis(trifluoromethyl)benzohydrazide 1263000-61-8 C13H12F6N2O 326.241 —— 3,5-bis(trifluoromethyl)-N'-(hex-5-en-2-yl)benzohydrazide 1263000-32-3 C15H16F6N2O 354.295 —— N'-(bicyclo[2.2.1]heptan-2-yl)-3,5-bis(trifluoromethyl)benzohydrazide 1263000-46-9 C16H16F6N2O 366.306
反应信息
-
作为反应物:描述:3,5-二(三氟甲基)苯-1-碳酰肼 在 盐酸 、 sodium cyanoborohydride 作用下, 以 叔丁醇 为溶剂, 生成 N'-benzyl-3,5-bis(trifluoromethyl)benzohydrazide参考文献:名称:Improved Cope-type hydroamination reactivity of hydrazine derivatives摘要:报道了对无金属、Cope型偶氮胺反应性的肼羧酸及其类似物的系统研究。优化肼羧酸结构使分子内反应更加容易,并实现了烯烃的分子间反应,从而提供了一种直接制备多取代肼羧酸的方法。DOI:10.1039/c0cc02403a
-
作为产物:描述:参考文献:名称:双极性电荷传输嵌段共聚物的合成及有机发光二极管的表征摘要:通过三氧化胺介导的自由基聚合反应制备了一系列由三苯胺部分作为空穴传输单元和恶二唑部分作为电子传输单元的空穴和电子传输无规和嵌段共聚物。具有叔丁基或三氟甲基2和7的恶二唑单体分别用于共聚。聚合物的光致发光测量表明,三苯胺和恶二唑单元之间的激基复合物的形成倾向于以无规共聚物,嵌段共聚物和聚合物共混物的顺序发生,这意味着嵌段或共混体系中的相分离形态。该聚合物被用于OLED器件,我们发现聚合物层的形态严重影响了器件的性能。包含空穴和电子传输单元且组成为14/86的嵌段共聚物显示出超过10%的最高外部量子效率。©2010 Wiley Periodicals,Inc. J Polym Sci A部分:Polym Chem 48:1461-1468,2010年DOI:10.1002/pola.23853
文献信息
-
Triazole derivatives申请人:Hoffmann-La Roche Inc.公开号:US06265426B1公开(公告)日:2001-07-24The present invention relates to triazole and imidazole derivatives of formula I and to their pharmaceutically acceptable acid addition salts. These compounds are NMDA receptor subtype blockers and are useful for the treatment of diseases related to the NMDA receptor.
-
AMINO ALCOHOL DERIVATIVE, PHARMACEUTICAL COMPOSITION AND APPLICATION THEREOF申请人:Beijing Foreland Pharma Co., Ltd.公开号:US20210347745A1公开(公告)日:2021-11-11The present invention belongs to the field of medicine, and specifically discloses an amino alcohol derivative represented by Formula I, a pharmaceutically acceptable salt, solvate, polymorph or prodrug thereof. In addition, the present invention also discloses a pharmaceutical composition comprising the above substances, and a use of the substance in the preparation of a medicament for the prevention and treatment of an immune inflammatory disease, or a disease or condition associated with immunological competence such as multiple sclerosis, ALS, CIDP, systemic lupus erythematosus, rheumatoid arthritis, ulcerative colitis, psoriasis, polymyositis, etc.
-
Polycyclic N-Benzamido Imides with Potent Activity against Vaccinia Virus作者:Eva Torres、María D. Duque、Pelayo Camps、Lieve Naesens、Teresa Calvet、Mercè Font-Bardia、Santiago VázquezDOI:10.1002/cmdc.201000306日期:2010.12.3The synthesis and antiviral activity of a series of novel polycyclic analogues of the orthopoxvirus egress inhibitor tecovirimat (ST-246) is presented. Several of these compounds display sub-micromolar activity against vaccinia virus, and were more potent than cidofovir (CDV). The more active compounds were about 10-fold more active than CDV, with minimum cytotoxic concentrations above 100 μM. Chemical
-
Design, Synthesis, and Study of the Insecticidal Activity of Novel Steroidal 1,3,4-Oxadiazoles作者:Shichuang Ma、Weiqi Jiang、Qi Li、Tian Li、Wenjun Wu、Hangyu Bai、Baojun ShiDOI:10.1021/acs.jafc.1c00088日期:2021.10.6was designed and synthesized, and the target compounds were evaluated for their insecticidal activity against five aphid species. Most of the tested compounds exhibited potent insecticidal activity against Eriosoma lanigerum (Hausmann), Myzus persicae, and Aphis citricola. Compounds 20g and 24g displayed the highest activity against E. lanigerum, showing LC50 values of 27.6 and 30.4 μg/mL, respectively设计并合成了一系列具有取代的1,3,4-恶二唑结构的新型甾体衍生物,并评估了目标化合物对五种蚜虫的杀虫活性。大多数测试的化合物表现出对鼠尾草(Hausmann)、桃蚜和柑橘蚜的有效杀虫活性。化合物20g和24g对E. lanigerum 的活性最高,LC 50值分别为 27.6 和 30.4 μg/mL。E. lanigerum中肠细胞的超微结构变化通过透射电子显微镜检测,表明这些甾体恶唑衍生物可能通过破坏昆虫中肠细胞的线粒体和核膜来发挥其杀虫活性。此外,田间试验表明,化合物20g表现出与阳性对照毒死蜱和噻虫嗪对E. lanigerum的效果相似,在200 μg/mL剂量下21天后达到89.5%的控制率。我们还通过测定三种杀虫剂解毒酶的活性研究了E. lanigerum 中目标化合物的水解和代谢。复合物20g50 μg/mL 对羧酸酯酶的抑制作用类似于已知的抑制剂磷酸三苯酯。上述结果证明了
-
Synthesis, fungicidal activity, and 3D-QSAR of tetrazole derivatives containing phenyloxadiazole moieties作者:Yi-Tao Li、Wen-Qiang Yao、Si Zhou、Jun-Xing Xu、Hui Lu、Jian Lin、Xiao-Yun Hu、Shao-Kai ZhangDOI:10.1016/j.bmcl.2020.127762日期:2021.2In an effort to discover new agents with good fungicidal activities against CDM (cucumber downy mildew), a series of tetrazole derivatives containing phenyloxadiazole moieties were designed and synthesized. The EC50 values for fungicidal activities against CDM were determined. Bioassay results indicated that most synthesized compounds exhibited potential in vivo fungicidal activity against CDM. A CoMFA
表征谱图
-
氢谱1HNMR
-
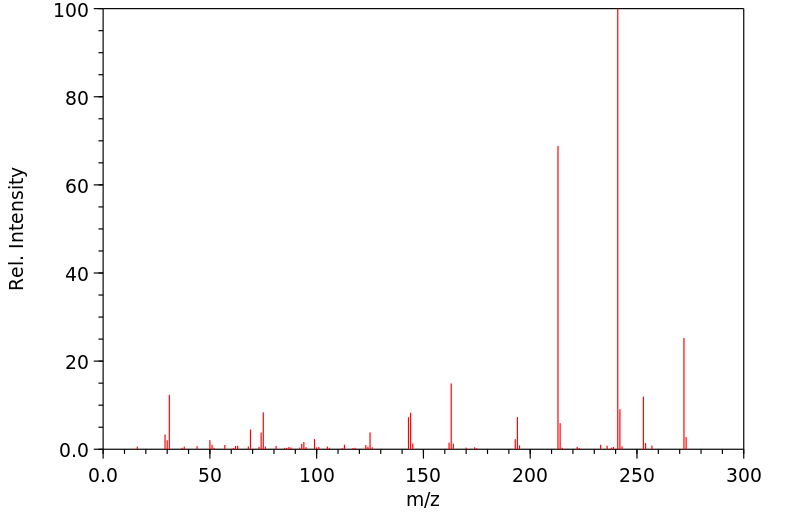
质谱MS
-
碳谱13CNMR
-
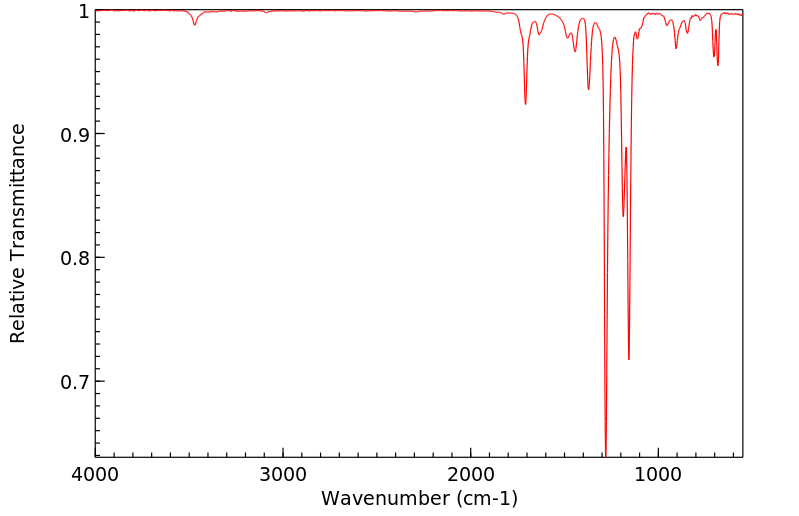
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫