(E)-3-dodecene | 7206-14-6
中文名称
——
中文别名
——
英文名称
(E)-3-dodecene
英文别名
3-Dodecene;(E)-dodec-3-ene
CAS
7206-14-6
化学式
C12H24
mdl
——
分子量
168.323
InChiKey
WLTSXAIICPDFKI-FNORWQNLSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:212.2±7.0 °C(Predicted)
-
密度:0.764±0.06 g/cm3(Predicted)
-
LogP:6.613 (est)
-
保留指数:1193;1193;1193;1185
计算性质
-
辛醇/水分配系数(LogP):5.8
-
重原子数:12
-
可旋转键数:8
-
环数:0.0
-
sp3杂化的碳原子比例:0.83
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 十二碳-1,3-二烯 (E)-1,3-dodecadiene 87921-45-7 C12H22 166.307 正癸烯 1-Decene 872-05-9 C10H20 140.269 —— (E)-1-chlorodec-1-ene 66291-48-3 C10H19Cl 174.714
反应信息
-
作为产物:描述:参考文献:名称:Stereoselective transformation of 1-alkeny ether (R1CH=CHOMe) into alkene (R1CH=CHR2) based on stereospecific elimination of the vicinal iodo(methoxy)alkane摘要:Treatment of alkenyl methyl ether with ICl rapidly gave 1-chloro-2-iodo-1-methoxyalkane quantitatively. Without isolation, this dihalide was treated with Et3Al to afford anti-iodo(methoxy)alkane in good yield with high stereoselectivity. The stereochemistry of the starting alkenyl ether did not affect the stereochemical outcome of the product. The use of alkynylaluminum ((RC=C)(2)AlEt) resulted in alkynylation of the alpha-chloro-beta-iodoether. Anti-iodo(methoxy)alkane was converted into (Z)-alkene upon treatment with n-BuLi in hexane-ether at -78 degrees C. On the other hand, the reaction of the same anti-iodo(methoxy)alkane with allylsilane-TiCl4 provided (E)-alkene. (C) 1998 Elsevier Science Ltd. All rights reserved.DOI:10.1016/s0040-4020(98)01066-7
文献信息
-
Control of Selectivity through Synergy between Catalysts, Silanes, and Reaction Conditions in Cobalt-Catalyzed Hydrosilylation of Dienes and Terminal Alkenes作者:Balaram Raya、Stanley Jing、Vagulejan Balasanthiran、T. V. RajanBabuDOI:10.1021/acscatal.6b03373日期:2017.4.7with 2 equiv of NaEt3BH at −78 °C in toluene to generate a catalyst that effects highly selective anti-Markovnikov hydrosilylation of the terminal double bond in 1,3- and 1,4-dienes. Primary and secondary silanes such as PhSiH3, Ph2SiH2, and PhSi(Me)H2 react with a broad spectrum of terminal dienes without affecting the configuration of the other double bond. When dienes conjugated to an aromatic ring易获得的(i- Pr PDI)CoCl 2 [ i- Pr PDI = 2,6-双(2,6-二异丙基苯基亚氨基乙基)吡啶]在-78°C下与2当量的NaEt 3 BH在甲苯中反应生成催化剂,该催化剂在1,3-和1,4-二烯中的末端双键的高选择性抗Markovnikov氢化硅烷化作用。伯硅烷和仲硅烷,例如PhSiH 3,Ph 2 SiH 2和PhSi(Me)H 2与多种末端二烯发生反应,而不会影响另一个双键的构型。当涉及与芳族环共轭的二烯时,既形成马尔可夫尼可夫产物又形成反马尔可夫尼可夫产物。该反应可耐受各种官能团,例如芳基溴化物,芳基碘化物,受保护的醇,甚至甲硅烷基烯醇醚。1-烯烃在相似条件下的反应干净地导致了马尔可夫尼可夫和抗马尔可夫尼可夫氢化硅烷化产物的混合物,其中产物的比例随着亚氨基芳基中2,6-取代基的尺寸变大而越来越有利于后者。配合物(i- Pr PDI)CoCl 2仅提供用于各种
-
Multiple Mechanistic Pathways for Zirconium-Catalyzed Carboalumination of Alkynes. Requirements for Cyclic Carbometalation Processes Involving C−H Activation作者:Ei-ichi Negishi、Denis Y. Kondakov、Danièle Choueiry、Kayoko Kasai、Tamotsu TakahashiDOI:10.1021/ja9538039日期:1996.1.1hydrometalation, have been observed, and the courses of these reactions significantly depend on (i) the nature and number of alkyl groups in organoalanes, (ii) their amounts, and (iii) solvents. The reaction of alkynes with Et3Al in the presence of 0.1 equiv of Cp2ZrCl2 in nonpolar solvents, e.g., hexanes, proceeds via C−H activation to give the corresponding aluminacyclopentenes. Investigation of the reaction of
-
Stereoselective formation of (E)-olefins by hydrolytic desulphinylation of some substituted 4-(2′-alkenesulphinyl)-morpholines作者:Jean-Bernard Baudin、Sylvestre A. JuliaDOI:10.1016/s0040-4039(00)99626-6日期:1989.1The boron trifluoride-etherate catalysed hydrolysis of the title sulphinamides provide olefins with (E:Z)-ratios depending on the nature of the substituents on the allylic chain.
-
Transformation of carbon-oxygen into carbon-carbon bonds mediated by low-valent nickel species作者:Ernest Wenkert、Enrique L. Michelotti、Charles S. Swindell、Marco TingoliDOI:10.1021/jo00199a030日期:1984.12
-
Kondo; Negishi; Tunemoto, Angewandte Chemie, 1974, vol. 86, # 11, p. 415 - 415作者:Kondo、Negishi、TunemotoDOI:——日期:——
表征谱图
-
氢谱1HNMR
-
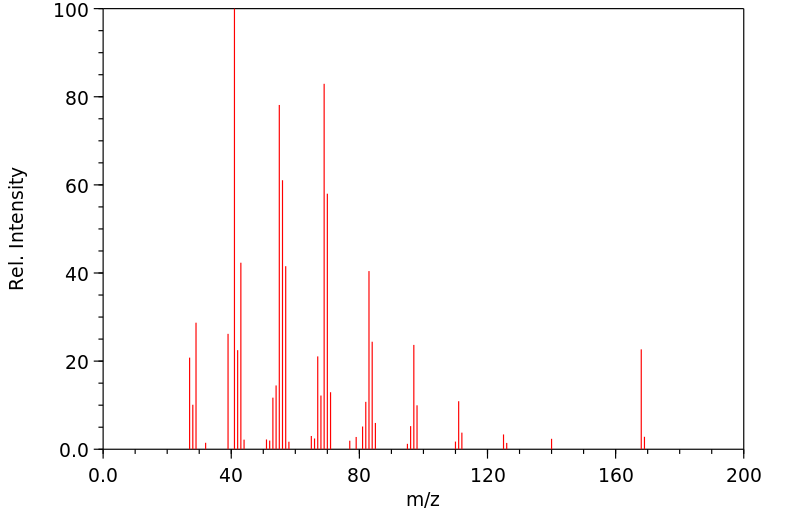
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
高密聚乙烯
香叶醇
顺式3-甲基-2-己烯
顺式-5-癸烯
顺式-5-甲基-2-己烯
顺式-5-庚烯-1-炔
顺式-4-癸烷
顺式-4-甲基-2-戊烯
顺式-4-甲基-2-戊烯
顺式-3-癸烯
顺式-3-甲基-3-己烯
顺式-3-甲基-2-庚烯
顺式-3-戊烯-1-炔
顺式-3,4-二甲基-3-己烯
顺式-3,4-二甲基-2-戊烯
顺式-3,4-二甲基-2-戊烯
顺式-2-甲基-3-己烯
顺式-2-壬烯
顺式-2-丁烯-D1
顺式-1.1.1-三甲基-2-丁烯
顺式-1-甲基-2-环丙基乙烯
顺式-1-甲基-2-乙烯基环戊烷
顺式-1-环戊基-1-辛烯
顺式-1-氘代-3-甲基-1-丁烯
顺式-(9ci)-2,3,3a,7a-四氢-4-(1-甲基乙基)-1H-茚
顺式-(2-丁烯基)环丙烷
顺式,顺式-2,4-己二烯
顺-环辛烯
顺-9-二十一碳烯
顺-6-十三碳烯
顺-5-甲基-1,3,6-庚三烯
顺-4-辛烯
顺-4-壬烯
顺-3-辛烯
顺-3-甲基-2-戊烯
顺-3-壬烯
顺-3-十三碳烯
顺-2-辛烯
顺-2-癸烯
顺-2-戊烯
顺-2-庚烯
顺-2-己烯
顺-2-丁烯
顺-2,2-二甲基-3-己烯
顺-1,3-戊二烯
顺,顺-1,9-环十六烷二烯
顺,顺,顺-环癸-1,3,5-三烯
间戊二烯
间二(4-吡啶基)苯
镁,二-2-丁烯基-