N,N-二异丙基乙醇胺 | 96-80-0
中文名称
N,N-二异丙基乙醇胺
中文别名
2-(二异丙氨基)乙醇;二异丙胺基乙醇;二异丙基乙醇胺
英文名称
2-(diisopropylamino)ethanol
英文别名
2-(bis(1-methylethyl)amino)-ethanol;diisopropyl ethanolamine;2-[di(propan-2-yl)amino]ethanol
CAS
96-80-0
化学式
C8H19NO
mdl
MFCD00008869
分子量
145.245
InChiKey
ZYWUVGFIXPNBDL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
稳定性/保质期:
- 常温常压下稳定。
- 本品易燃、低毒。生产设备需密闭,车间要保持良好通风,注意防火。操作人员应穿戴防护用具。
计算性质
-
辛醇/水分配系数(LogP):1.2
-
重原子数:10
-
可旋转键数:4
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:23.5
-
氢给体数:1
-
氢受体数:2
安全信息
-
储存条件:常温下应存放在阴凉通风处。 产品应采用玻璃瓶外包裹木箱并内衬塑料进行包装,并需储存在阴凉、干燥、通风良好的仓库中。同时,要将此物品与苛性碱和氧化剂隔离存放。
制备方法与用途
上下游信息
反应信息
-
作为反应物:描述:N,N-二异丙基乙醇胺 在 氯化亚砜 、 potassium carbonate 作用下, 以 氯仿 、 丙酮 为溶剂, 反应 4.0h, 生成 S-(2-(diisopropylamino)ethyl) ethanethioate参考文献:名称:一种多用途且可回收的分子印迹聚合物,可作为硫衍生物的氧化催化剂:芥子气和V神经毒剂净化的新方法†摘要:含卟啉单元的分子印迹聚合物已开发为仿生异质催化剂,用于氧化硫衍生物。它在温和条件下的催化效率和易于回收的特性,对于设计用于钇铁矿和VX的新型去污工具而言,是一笔巨大的财富。DOI:10.1039/c9cc04928b
-
作为产物:描述:参考文献:名称:基于黄cut素代谢机制的黄cut素衍生物的设计,合成及生物学评价摘要:根据黄rin苷(1)在体内的代谢机理,设计和合成了3个系列的黄re苷衍生物。通过凝血酶原时间(PT),活化的部分凝血活酶时间(APTT),凝血酶时间(TT)和纤维蛋白原(FIB)的分析测试了它们的凝血酶抑制活性。这些目标产物的抗氧化活性通过1,1-二苯基-2-吡啶并肼基自由基(DPPH)测定进行了评估,并具有保护PC12细胞免受H 2 O 2诱导的细胞毒性的能力,并通过紫外线(UV)评估了它们的溶解度。分光光度计。结果表明,两个异丙基取代衍生物(18c)与黄cut苷(2)相比,显示出更强的抗凝活性,更好的水溶性和良好的抗氧化活性,因此有必要进一步开发18c作为治疗缺血性脑血管疾病的有希望的药物。DOI:10.1111/cbdd.12727
-
作为试剂:描述:苯乙烯 、 2’-溴-4-氯联苯 在 (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride 、 tetrakis(actonitrile)copper(I) hexafluorophosphate 、 4,4'-二甲氧基-2,2'-联吡啶 、 N,N-二异丙基乙醇胺 、 1-allyl-3-methylimidazolium tetrafluoroborate 作用下, 反应 12.0h, 以95.3%的产率得到2-chloro-10-phenylphenanthrene参考文献:名称:一种碳酸氢钾环境下合成医药中间体菲化合物的方法摘要:本发明涉及一种碳酸氢钾环境下合成下式(I)所示菲化合物的方法,所述方法包括:惰性气氛下,在催化剂、有机配体和碳酸氢钾存在下,于溶剂中,下式(II)化合物与式(III)化合物发生反应,从而得到式(I)化合物;其中,R1、R2各自独立地为H、C1?C6烷基、C1?C6烷氧基或卤素;R3为C6?C10芳基或C5?C8杂芳基,所述C6?C10芳基或C4?C8杂芳基任选被1?3个取代基取代,所述取代基为C1?C6烷基或卤素。所述方法通过合适催化剂、有机配体、碱和溶剂的选择而取得了良好的效果,具备广泛的工业应用前景。公开号:CN105777462A
文献信息
-
[EN] TRIAZINES SUITABLE FOR USE IN FABRIC TREATMENT COMPOSITIONS<br/>[FR] TRIAZINES POUVANT ETRE UTILISEES DANS DES COMPOSITIONS DE TRAITEMENT DE TISSUS申请人:UNILEVER PLC公开号:WO2005123699A1公开(公告)日:2005-12-29A water-soluble, triazine-based, non-dye, cellulose cross-linking agent that has a highly flexible linking group between at least two, mono-reactive cross-linking moieties and further hydrophilic or non-hydrophilic substituents, being preferrably represented by the general formula (I): (R1)(X1)T-L1-B-T(X2)(R2) wherein: R1 et R2 are cellulose-unreactive substituent groups on the s-triazine (T) and may be the same or different, X1 and X2 are leaving groups on the s-triazine which are lost on reaction with cellulose and may be the same or different, L1 et L2 are linking groups, an may be the same or different or absent, B is the bridging group comprising or consisting of at least one aliphatic polyoalkylene chain.
-
[EN] NEW THIENOPYRIMIDINE DERIVATIVES, A PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM<br/>[FR] NOUVEAUX DÉRIVÉS DE THIÉNOPYRIMIDINE, PROCÉDÉ POUR LEUR PRÉPARATION ET COMPOSITIONS PHARMACEUTIQUES LES CONTENANT申请人:SERVIER LAB公开号:WO2015097123A1公开(公告)日:2015-07-02Compounds of formula (I): wherein R1, R2, R3, R4, R5, R6, R7, R12, X, A and n are as defined in the description.式(I)的化合物:其中R1、R2、R3、R4、R5、R6、R7、R12、X、A和n的定义如描述中所述。
-
Studies on Cognitive Enhancing Agents. III. Antiamnestic and Antihypoxic Activities of a Series of l-Bicycloaryl-2-(.OMEGA.-aminoalkoxy)ethanols.作者:Satoshi ONO、Tetsuo YAMAFUJI、Hisaaki CHAKI、Hajime MORITA、Yozo TODO、Naomi OKADA、Mutsuko MAEKAWA、Kazunori KITAMURA、Masaru TAI、Hirokazu NARITADOI:10.1248/cpb.43.1492日期:——2-(2-Aminoethoxy)-1-hydroxyethyl derivatives of bicyclic arenes) naphthalene, thianaphthene, benzofuran, and indole) were prepared and screened for antiamnestic (AA) and antihypoxic (AH) activities which were evaluated by measuring the reversing potency in electroconvulsion-induced amnesia and the protective effect against hypoxia, respectively, in mice. Compound 3o, 1-(benzo[b]thiophen-5-yl)-2-(2-diethylaminoethoxy)ethanol, showed the best AA and AH activity profile, being superior to our prototype compound, 2-(2-dimethylaminoethoxy)-1-phenylethanol (1). Elongation of the ethylene linkage in the side chain of 3o to 3- and 4-carbon moiethies brought about a significant decrease in AH activity. Compound 3o was further investigated for its protective effect against Co2-induced memory impairment and for acute toxicity in mice. It is ten-fold more potent than tacrine in the amnesia-reversal assay and is considerably less toxic than tacrine.
-
[EN] ARYLAMINE SUBSTUTUTED BICYCLIC HETEROAROMATIC COMPOUNDS AS P38 KINASE INHIBITORS<br/>[FR] COMPOSES BICYCLIQUES HETEROAROMATIQUES ARYLAMINE SUBSTITUEE UTILISES EN TANT QU'INHIBITEURS DE KINASE P38申请人:CELLTECH R&D LTD公开号:WO2004000846A1公开(公告)日:2003-12-31Bicyclic heteroaromatic derivatives of formula (1) are described: F (1) where: the dashed line joining A and C(Ra) is present and represents a bond and A is a -N= atom or a -C(Rb)= group, or the dashed line is absent and A is a -N(Rb)-, or -C(Rb)(Rc)- group; X is an -O-, -S- or substituted nitrogen atom or a -S(O)-, -S(O2)- or -NH-group; Y is a nitrogen or substituted carbon atom or a -CH= group; n is zero or the integer 1; Alk1 is an optionally substituted aliphatic or heteroaliphatic chain L1 is a covalent bond or a linker atom or group; Cy1 is a hydrogen atom or an optionally substituted cycloaliphatic, polycycloaliphatic, heterocycloaliphatic, polyheterocycloaliphatic, aromatic or heteroaromatic group; Ar is an optionally substituted aromatic or heteroaromatic group; and the remaining substituents are defined in the specification. The compounds are potent and selective inhibitors of p38 kinase and are of use in the prophylaxis and treatment of immune or inflammatory disorders.公式(1)描述了具有双环杂芳衍生物:F(1)其中:连接A和C(Ra)的虚线存在且表示键,A是一个-N=原子或一个-C(Rb)=基团,或者虚线不存在且A是一个-N(Rb)-或-C(Rb)(Rc)-基团;X是一个-O-,-S-或取代的氮原子或一个-S(O)-,-S(O2)-或-NH-基团;Y是一个氮或取代的碳原子或一个-CH=基团;n为零或整数1;Alk1是一个可选择取代的脂肪或杂原脂肪链L1是一个共价键或连接原子或基团;Cy1是一个氢原子或一个可选择取代的环脂肪,多环脂肪,杂环脂肪,多杂环脂肪,芳香或杂芳基团;Ar是一个可选择取代的芳香或杂芳基团;其余取代基在规范中定义。这些化合物是p38激酶的有效和选择性抑制剂,并可用于预防和治疗免疫或炎症性疾病。
-
[EN] THIENOPYRIDONE DERIVATIVES AS KINASE INHIBITORS<br/>[FR] DERIVES DE THIENOPYRIDONE EN TANT QU'INHIBITEURS DE KINASE申请人:CELLTECH R&D LTD公开号:WO2004113347A1公开(公告)日:2004-12-29A series of thieno[2,3-b]pyridin-6(7H)-one derivatives, substituted in the 2-position by a carbonyl- or sulfonyl-linked pyrrolidin-1-yl or related moiety, being inhibitors of p38 MAP kinase, are accordingly of use in medicine, for example in the treatment and/or prevention of immune or inflammatory disorders.
表征谱图
-
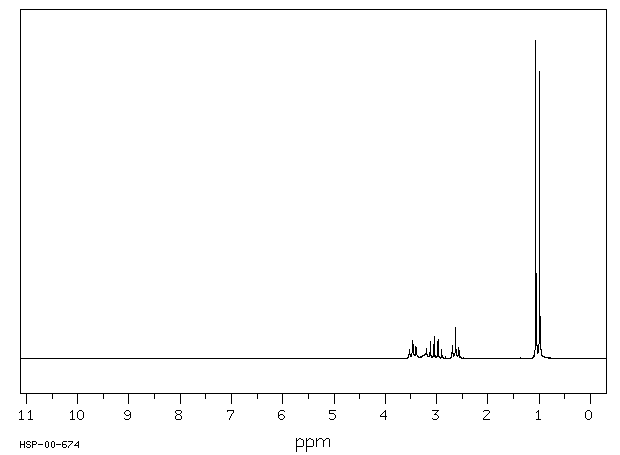
氢谱1HNMR
-
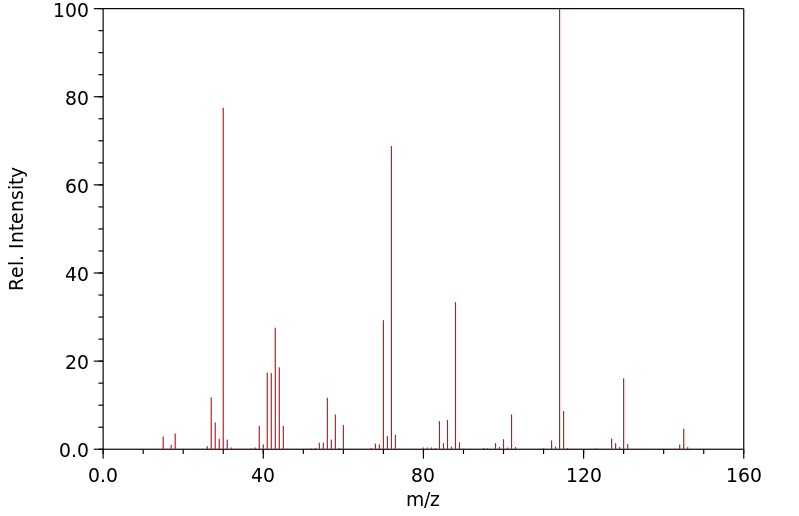
质谱MS
-
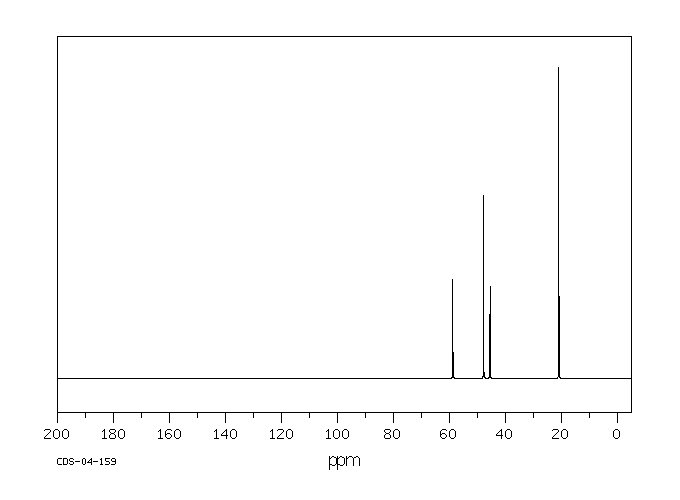
碳谱13CNMR
-
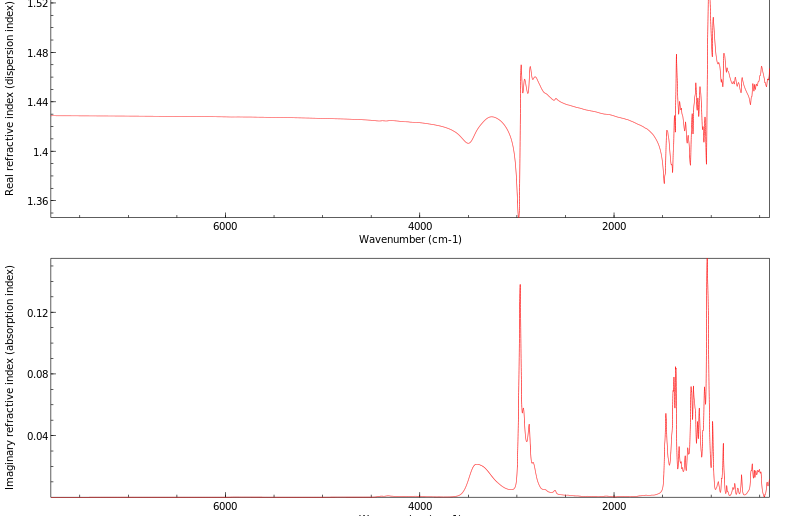
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(乙腈)二氯镍(II)
(R)-(-)-α-甲基组胺二氢溴化物
(N-(2-甲基丙-2-烯-1-基)乙烷-1,2-二胺)
(4-(苄氧基)-2-(哌啶-1-基)吡啶咪丁-5-基)硼酸
(11-巯基十一烷基)-,,-三甲基溴化铵
鼠立死
鹿花菌素
鲸蜡醇硫酸酯DEA盐
鲸蜡硬脂基二甲基氯化铵
鲸蜡基胺氢氟酸盐
鲸蜡基二甲胺盐酸盐
高苯丙氨醇
高箱鲀毒素
高氯酸5-(二甲氨基)-1-({(E)-[4-(二甲氨基)苯基]甲亚基}氨基)-2-甲基吡啶正离子
高氯酸2-氯-1-({(E)-[4-(二甲氨基)苯基]甲亚基}氨基)-6-甲基吡啶正离子
高氯酸2-(丙烯酰基氧基)-N,N,N-三甲基乙铵
马诺地尔
马来酸氢十八烷酯
马来酸噻吗洛尔EP杂质C
马来酸噻吗洛尔
马来酸倍他司汀
顺式环己烷-1,3-二胺盐酸盐
顺式氯化锆二乙腈
顺式吡咯烷-3,4-二醇盐酸盐
顺式双(3-甲氧基丙腈)二氯铂(II)
顺式3,4-二氟吡咯烷盐酸盐
顺式1-甲基环丙烷1,2-二腈
顺式-二氯-反式-二乙酸-氨-环己胺合铂
顺式-二抗坏血酸(外消旋-1,2-二氨基环己烷)铂(II)水合物
顺式-N,2-二甲基环己胺
顺式-4-甲氧基-环己胺盐酸盐
顺式-4-环己烯-1.2-二胺
顺式-4-氨基-2,2,2-三氟乙酸环己酯
顺式-3-氨基环丁烷甲腈盐酸盐
顺式-2-羟基甲基-1-甲基-1-环己胺
顺式-2-甲基环己胺
顺式-2-(苯基氨基)环己醇
顺式-2-(苯基氨基)环己醇
顺式-2-(氨基甲基)-1-苯基环丙烷羧酸盐酸盐
顺式-1,3-二氨基环戊烷
顺式-1,2-环戊烷二胺二盐酸盐
顺式-1,2-环戊烷二胺
顺式-1,2-环丁腈
顺式-1,2-双氨甲基环己烷
顺式--N,N'-二甲基-1,2-环己二胺
顺式-(R,S)-1,2-二氨基环己烷铂硫酸盐
顺式-(2-氨基-环戊基)-甲醇
顺-2-戊烯腈
顺-1,3-环己烷二胺
顺-1,3-双(氨甲基)环己烷