4-戊基苯硼酸 | 121219-12-3
中文名称
4-戊基苯硼酸
中文别名
4-正戊(烷)基苯硼酸;4-正戊基苯硼酸;对戊基苯硼酸
英文名称
(4-pentylphenyl)boronic acid
英文别名
4-n-Pentylphenylboronic acid;4-pentylbenzene boronic acid
CAS
121219-12-3
化学式
C11H17BO2
mdl
MFCD00995151
分子量
192.066
InChiKey
UZRMPSOGFATLJE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:82-84°C
-
沸点:328.0±35.0 °C(Predicted)
-
密度:1.01±0.1 g/cm3(Predicted)
-
稳定性/保质期:
如果遵照规格使用和储存,则不会分解,未有已知危险反应。请避免接触氧化物。
计算性质
-
辛醇/水分配系数(LogP):3.69
-
重原子数:14
-
可旋转键数:5
-
环数:1.0
-
sp3杂化的碳原子比例:0.454
-
拓扑面积:40.5
-
氢给体数:2
-
氢受体数:2
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S26,S36
-
危险类别码:R36/37/38
-
海关编码:2931900090
-
危险类别:IRRITANT
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
SDS
4-Amylphenylboronic Acid (contains varying amounts Revision number: 6
of Anhydride)
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 4-Amylphenylboronic Acid (contains varying amounts of Anhydride)
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Skin corrosion/irritation Category 2
Category 2A
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Warning
Signal word
Hazard statements Causes skin irritation
Causes serious eye irritation
Precautionary statements:
Wash hands thoroughly after handling.
[Prevention]
Wear protective gloves/eye protection/face protection.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
[Response]
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
4-Amylphenylboronic Acid (contains varying amounts of Anhydride)
Components:
Percent: ....
121219-12-3
CAS Number:
Synonyms: 4-Amylbenzeneboronic Acid (contains varying amounts of Anhydride) , 4-
Pentylphenylboronic Acid (contains varying amounts of Anhydride) , 4-
Pentylbenzeneboronic Acid (contains varying amounts of Anhydride)
Chemical Formula: C11H17BO2
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Comply with laws.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Safety glasses. A face-shield, if the situation requires.
Eye protection:
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Form: Crystal- Powder
Colour: White - Almost white
Odour: No data available
pH: No data available
Melting point/freezing point:84°C
Boiling point/range: No data available
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: No data available
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Boron oxides
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
No data available
Fish:
Crustacea: No data available
No data available
Algae:
Persistence / degradability: No data available
No data available
Bioaccumulative
potential(BCF):
Mobility in soil
Log Pow: No data available
No data available
Soil adsorption (Koc):
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 14. TRANSPORT INFORMATION
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
of Anhydride)
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: 4-Amylphenylboronic Acid (contains varying amounts of Anhydride)
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS
Skin corrosion/irritation Category 2
Category 2A
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Warning
Signal word
Hazard statements Causes skin irritation
Causes serious eye irritation
Precautionary statements:
Wash hands thoroughly after handling.
[Prevention]
Wear protective gloves/eye protection/face protection.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
[Response]
if present and easy to do. Continue rinsing.
If eye irritation persists: Get medical advice/attention.
IF ON SKIN: Gently wash with plenty of soap and water.
If skin irritation occurs: Get medical advice/attention.
Take off contaminated clothing and wash before reuse.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
4-Amylphenylboronic Acid (contains varying amounts of Anhydride)
Components:
Percent: ....
121219-12-3
CAS Number:
Synonyms: 4-Amylbenzeneboronic Acid (contains varying amounts of Anhydride) , 4-
Pentylphenylboronic Acid (contains varying amounts of Anhydride) , 4-
Pentylbenzeneboronic Acid (contains varying amounts of Anhydride)
Chemical Formula: C11H17BO2
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Comply with laws.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Safety glasses. A face-shield, if the situation requires.
Eye protection:
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Form: Crystal- Powder
Colour: White - Almost white
Odour: No data available
pH: No data available
Melting point/freezing point:84°C
Boiling point/range: No data available
Flash point: No data available
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Relative density: No data available
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Boron oxides
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
No data available
Fish:
Crustacea: No data available
No data available
Algae:
Persistence / degradability: No data available
No data available
Bioaccumulative
potential(BCF):
Mobility in soil
Log Pow: No data available
No data available
Soil adsorption (Koc):
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
4-Amylphenylboronic Acid (contains varying
amounts of Anhydride)
Section 14. TRANSPORT INFORMATION
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
反应信息
-
作为反应物:描述:4-戊基苯硼酸 在 四(三苯基膦)钯 、 仲丁基锂 、 碳酸氢钠 、 potassium carbonate 作用下, 以 四氢呋喃 、 N,N-二甲基乙酰胺 、 水 为溶剂, 反应 27.8h, 生成 4-(2,3-difluoro-(4-(4-pentylphenyl))phenyl)phenol参考文献:名称:JP6136589摘要:公开号:
-
作为产物:描述:4-硝基苯酚三氟甲基磺酸酯 在 2-双环己基膦-2',6'-二甲氧基联苯 、 盐酸 、 potassium phosphate monohydrate 、 potassium hydrogen fluoride 、 palladium on activated charcoal 、 氢气 、 palladium diacetate 、 三乙胺 、 sodium nitrite 作用下, 以 1,4-二氧六环 、 甲醇 、 乙醇 、 水 、 甲苯 为溶剂, 反应 1.5h, 生成 4-戊基苯硼酸参考文献:名称:Design and synthesis of boronic acid inhibitors of endothelial lipase摘要:Endothelial lipase (EL) and lipoprotein lipase (LPL) are homologous lipases that act on plasma lipoproteins. EL is predominantly a phospholipase and appears to be a key regulator of plasma HDL-C. LPL is mainly a triglyceride lipase regulating (V)LDL levels. The existing biological data indicate that inhibitors selective for EL over LPL should have anti-atherogenic activity, mainly through increasing plasma HDL-C levels. We report here the synthesis of alkyl, aryl, or acyl-substituted phenylboronic acids that inhibit EL. Many of the inhibitors evaluated proved to be nearly equally potent against both EL and LPL, but several exhibited moderate to good selectivity for EL. (C) 2011 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmcl.2011.12.043
文献信息
-
Facile one-pot nanocatalysts encapsulation of palladium–NHC complexes for aqueous Suzuki–Miyaura couplings作者:Chao Chen、Qing Zheng、Shengliang Ni、Hangxiang WangDOI:10.1039/c7nj04836j日期:——nanoparticle encapsulation to fabricate water-soluble nanocatalysts (NCs) for aqueous cross-coupling reactions. To achieve this goal, three types of N-heterocyclic carbene (NHC)–palladium (Pd) complexes (i.e., 1b, 2b, and 3b) with the chemical formula [PdL(CH3CN)Cl]PF6 (where L is a bidentate pyridine- or pyrimidine-bearing NHC ligand) have been synthesized, and their structures were characterized by NMR spectroscopy使用水作为溶剂的有机转化已成为广泛研究的焦点。我们在此提出了一种策略,该策略使用简单的纳米颗粒封装来制造水溶性纳米催化剂(NC),以进行水交叉偶联反应。为了实现这一目标,三种类型的N-杂环卡宾的(NHC) -钯(Pd)的复合物(即,图1b,图2b,和图3b具有化学式)[PDL(CH 3 CN)CL] PF 6(其中L是含二齿吡啶或嘧啶的NHC配体)已被合成,并且它们的结构通过NMR光谱和X射线晶体学表征。利用一种简便的一锅封装方法,水不溶性Pd-NHC复合物可以整合到由两亲共聚物组成的胶束的疏水核中。这些水分散的纳米颗粒在Suzuki-Miyaura水溶液偶联中表现出出色的催化活性。在这些NC中,发现在其NHC配体上带有异氰酸酯部分的3b -NC在测试的反应条件下是活性最高的催化剂。更令人印象深刻的是,3b -NC可以重复使用至少五个循环而不会显着降低催化活性。
-
一种铜催化的芳基硼酸与二氧化碳的羧化反 应方法
-
[EN] G PROTEIN-COUPLED RECEPTOR MODULATORS AND A PHARMACEUTICAL COMPOSITION<br/>[FR] MODULATEURS DU RÉCEPTEUR COUPLÉ À LA PROTÉINE G ET COMPOSITION PHARMACEUTIQUE申请人:UNIV COPENHAGEN公开号:WO2021123394A1公开(公告)日:2021-06-24The invention concerns a compound of formula I, a pharmaceutical formulation comprising the compound of formula I and use of the pharmaceutical composition for treating inflammatory or diabetic disease.这项发明涉及一种I式化合物,包括该I式化合物的药物配方以及将该药物组合物用于治疗炎症性或糖尿病疾病的用途。
-
Palladium Membrane-Installed Microchannel Devices for Instantaneous Suzuki-Miyaura Cross-Coupling作者:Yoichi M. A. Yamada、Toshihiro Watanabe、Tomohiko Beppu、Naoshi Fukuyama、Kaoru Torii、Yasuhiro UozumiDOI:10.1002/chem.201000511日期:——Instantaneous catalytic carbon–carbon bond‐forming reactions were achieved in catalytic membrane‐installed microchannel devices that have a polymeric palladium‐complex membrane. The catalytic membrane‐installed microchannel devices were provided inside the microchannels by means of coordinative and ionic molecular convolution at the interface between the organic and aqueous phases flowing laminarly
-
N<sub>2</sub>Phos – an easily made, highly effective ligand designed for ppm level Pd-catalyzed Suzuki–Miyaura cross couplings in water作者:Nnamdi Akporji、Ruchita R. Thakore、Margery Cortes-Clerget、Joel Andersen、Evan Landstrom、Donald H. Aue、Fabrice Gallou、Bruce H. LipshutzDOI:10.1039/d0sc00968g日期:——A new biaryl phosphine-containing ligand from an active palladium catalyst for ppm level Suzuki–Miyaura couplings, enabled by an aqueous micellar reaction medium. A wide array of functionalized substrates including aryl/heteroaryl bromides are amenable, as are, notably, chlorides. The catalytic system is both general and highly effective at low palladium loadings (1000–2500 ppm or 0.10–0.25 mol%).
表征谱图
-
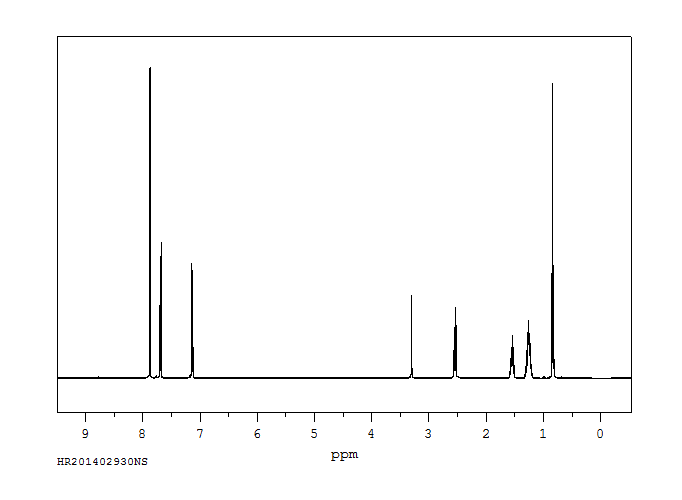
氢谱1HNMR
-
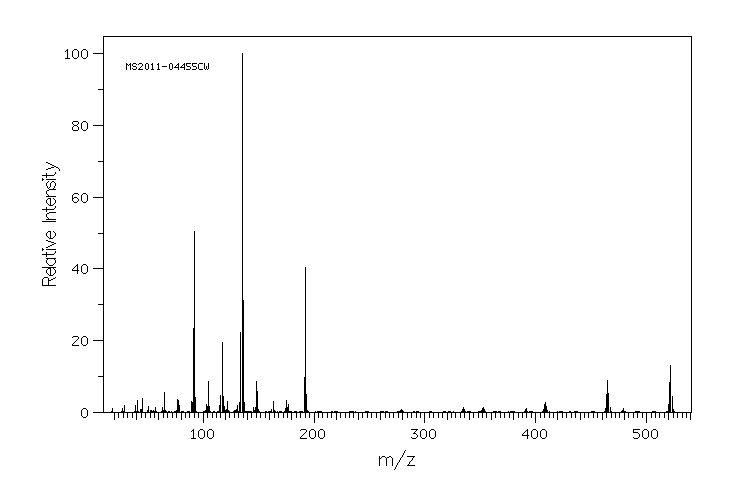
质谱MS
-
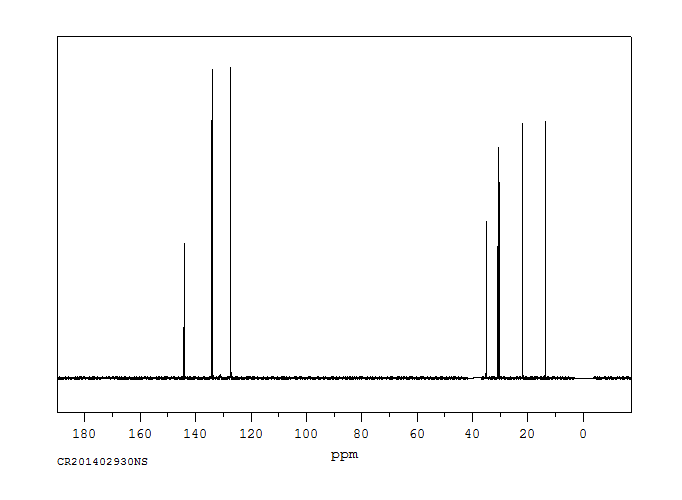
碳谱13CNMR
-
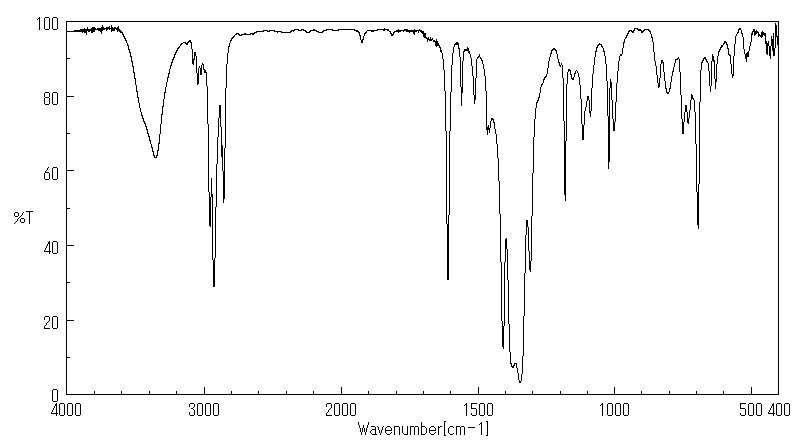
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫