2,4,6-三甲基吡啶-3,5-二羧酸二乙基酯 | 1150-55-6
中文名称
2,4,6-三甲基吡啶-3,5-二羧酸二乙基酯
中文别名
二乙基-2,4,6三甲基-3,5-吡啶
英文名称
2,4,6-trimethylpyridine-3,5-dicarboxylic acid diethyl ester
英文别名
diethyl 2,4,6-trimethylpyridine-3,5-dicarboxylate;diethyl 2,4,6-trimethyl-3,5-pyridinedicarboxylate;diethyl 2,6-dimethyl-4-methylpyridine-3,5-dicarboxylate;3,5-Pyridinedicarboxylic acid, 2,4,6-trimethyl-, diethyl ester
CAS
1150-55-6
化学式
C14H19NO4
mdl
——
分子量
265.309
InChiKey
IJHKOUDBKFONBB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2.5
-
重原子数:19
-
可旋转键数:6
-
环数:1.0
-
sp3杂化的碳原子比例:0.5
-
拓扑面积:65.5
-
氢给体数:0
-
氢受体数:5
SDS
SECTION 1: Identification of the substance/mixture and of the company/undertaking
Product identifiers
Product name : Diethyl 2,4,6-trimethyl-3,5-pyridinedicarboxylate
REACH No. : A registration number is not available for this substance as the substance
or its uses are exempted from registration, the annual tonnage does not
require a registration or the registration is envisaged for a later
registration deadline.
CAS-No. : 1150-55-6
Relevant identified uses of the substance or mixture and uses advised against
Identified uses : Laboratory chemicals, Manufacture of substances
SECTION 2: Hazards identification
Classification of the substance or mixture
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
Label elements
Other hazards
This substance/mixture contains no components considered to be either persistent, bioaccumulative and
toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher.
SECTION 3: Composition/information on ingredients
Substances
Formula : C14H19NO4
Molecular weight : 265,30 g/mol
CAS-No. : 1150-55-6
No components need to be disclosed according to the applicable regulations.
SECTION 4: First aid measures
Description of first aid measures
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration.
In case of skin contact
Wash off with soap and plenty of water.
In case of eye contact
Flush eyes with water as a precaution.
If swallowed
Never give anything by mouth to an unconscious person. Rinse mouth with water.
Most important symptoms and effects, both acute and delayed
The most important known symptoms and effects are described in the labelling (see section 2.2) and/or in
section 11
Indication of any immediate medical attention and special treatment needed
No data available
SECTION 5: Firefighting measures
Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
Special hazards arising from the substance or mixture
Carbon oxides, Nitrogen oxides (NOx)
Advice for firefighters
Wear self-contained breathing apparatus for firefighting if necessary.
Further information
No data available
SECTION 6: Accidental release measures
Personal precautions, protective equipment and emergency procedures
Avoid dust formation. Avoid breathing vapours, mist or gas.
For personal protection see section 8.
Environmental precautions
No special environmental precautions required.
Methods and materials for containment and cleaning up
Sweep up and shovel. Keep in suitable, closed containers for disposal.
Reference to other sections
For disposal see section 13.
SECTION 7: Handling and storage
Precautions for safe handling
Provide appropriate exhaust ventilation at places where dust is formed.
For precautions see section 2.2.
Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place.
Storage class (TRGS 510): Non Combustible Solids
Specific end use(s)
Apart from the uses mentioned in section 1.2 no other specific uses are stipulated
SECTION 8: Exposure controls/personal protection
Control parameters
Components with workplace control parameters
Exposure controls
Appropriate engineering controls
General industrial hygiene practice.
Personal protective equipment
Eye/face protection
Use equipment for eye protection tested and approved under appropriate government standards
such as NIOSH (US) or EN 166(EU).
Skin protection
The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and
the standard EN 374 derived from it.
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique
(without touching glove's outer surface) to avoid skin contact with this product. Dispose of
contaminated gloves after use in accordance with applicable laws and good laboratory practices.
Wash and dry hands.
Body Protection
Choose body protection in relation to its type, to the concentration and amount of dangerous
substances, and to the specific work-place., The type of protective equipment must be selected
according to the concentration and amount of the dangerous substance at the specific workplace.
Respiratory protection
Respiratory protection is not required. Where protection from nuisance levels of dusts are desired,
use type N95 (US) or type P1 (EN 143) dust masks. Use respirators and components tested and
approved under appropriate government standards such as NIOSH (US) or CEN (EU).
Control of environmental exposure
No special environmental precautions required.
SECTION 9: Physical and chemical properties
Information on basic physical and chemical properties
a) Appearance Form: solid
b) Odour No data available
c) Odour Threshold No data available
d) pH No data available
e) Melting point/freezing No data available
point
f) Initial boiling point and No data available
boiling range
g) Flash point No data available
h) Evaporation rate No data available
i) Flammability (solid, gas) No data available
j) Upper/lower No data available
flammability or
explosive limits
k) Vapour pressure No data available
l) Vapour density No data available
m) Relative density No data available
n) Water solubility No data available
o) Partition coefficient: n- log Pow: 1,998
octanol/water
p) Auto-ignition No data available
temperature
q) Decomposition No data available
temperature
r) Viscosity No data available
s) Explosive properties No data available
t) Oxidizing properties No data available
Other safety information
No data available
SECTION 10: Stability and reactivity
Reactivity
No data available
Chemical stability
Stable under recommended storage conditions.
Possibility of hazardous reactions
No data available
Conditions to avoid
No data available
Incompatible materials
Strong oxidizing agents
Hazardous decomposition products
In the event of fire: see section 5
SECTION 11: Toxicological information
Information on toxicological effects
Acute toxicity
No data available
Skin corrosion/irritation
No data available
Serious eye damage/eye irritation
Respiratory or skin sensitisation
No data available
Germ cell mutagenicity
Carcinogenicity
IARC: No component of this product present at levels greater than or equal to 0.1% is identified as
probable, possible or confirmed human carcinogen by IARC.
Reproductive toxicity
Specific target organ toxicity - single exposure
Specific target organ toxicity - repeated exposure
Aspiration hazard
Additional Information
RTECS: Not available
SECTION 12: Ecological information
Toxicity
No data available
Persistence and degradability
No data available
Bioaccumulative potential
Mobility in soil
Results of PBT and vPvB assessment
This substance/mixture contains no components considered to be either persistent, bioaccumulative and
toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher.
Other adverse effects
No data available
SECTION 13: Disposal considerations
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company.
Contaminated packaging
Dispose of as unused product.
SECTION 14: Transport information
UN number
ADR/RID: - IMDG: - IATA: -
UN proper shipping name
ADR/RID: Not dangerous goods
IMDG: Not dangerous goods
IATA: Not dangerous goods
Transport hazard class(es)
ADR/RID: - IMDG: - IATA: -
Packaging group
ADR/RID: - IMDG: - IATA: -
Environmental hazards
ADR/RID: no IMDG Marine pollutant: no IATA: no
Special precautions for user
No data available
SECTION 15: Regulatory information
This safety datasheet complies with the requirements of Regulation (EC) No. 1907/2006.
Safety, health and environmental regulations/legislation specific for the substance or mixture
No data available
Chemical Safety Assessment
For this product a chemical safety assessment was not carried out
SECTION 16: Other information
Further information
Copyright 2014 Co. LLC. License granted to make unlimited paper copies for internal use
only.
The above information is believed to be correct but does not purport to be all inclusive and shall be
used only as a guide. The information in this document is based on the present state of our knowledge
and is applicable to the product with regard to appropriate safety precautions. It does not represent any
guarantee of the properties of the product. Corporation and its Affiliates shall not be held
liable for any damage resulting from handling or from contact with the above product. See
and/or the reverse side of invoice or packing slip for additional terms and conditions of sale.
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— trimethyl-pyridine-3,5-dicarboxylic acid monoethyl ester 14458-02-7 C12H15NO4 237.255 2,3,5-三甲基-4-吡咯羧酸乙酯 2,4,5-trimethyl-3-ethoxycarbonylpyrrole 2199-54-4 C10H15NO2 181.235 4-甲基吡啶-3,5-二甲醇 3,5-bis-hydroxymethyl-2,4,6-trimethyl-pyridine 633318-43-1 C10H15NO2 181.235
反应信息
-
作为反应物:描述:2,4,6-三甲基吡啶-3,5-二羧酸二乙基酯 在 三氟化硼乙醚 、 tetrabutylammonium tetrafluoroborate 、 水 作用下, 以 四氢呋喃 为溶剂, 反应 12.0h, 以79%的产率得到2,3,5-三甲基-4-吡咯羧酸乙酯参考文献:名称:通过将乙酸乙酯从芳香环中电化学挤出,将汉茨酯及其衍生物环缩合为吡咯摘要:开发了HEs及其吡啶衍生物的电化学环收缩法以获得多取代的吡咯。该方法提供了Hantzsch酯作为侧链或氢供体在正交文献中的正交应用。正式的转化显示了一步就将乙酸乙酯从吡啶环中挤出。除了新颖的转化,我们还发现了路易斯酸在分子内电化学过程中的区域选择性的分子间控制。该反应提供了许多从未得到的多取代的吡咯,包括药物中间体和光开关。不寻常的4电子连续还原反应驱动了前所未有的阴离子脱芳香化/环收缩/再芳香化途径。DOI:10.1039/d1gc00487e
-
作为产物:描述:参考文献:名称:Hantzsch 1,4-二氢吡啶在乙酸中在对甲苯磺酸存在下通过自氧化轻松芳构化摘要:使用乙酸中的对甲苯磺酸建立了以高产率实现 Hantzsch 二氢吡啶芳构化的简单方案,并且在室温下获得了 90% 的产率。对于衍生自带有一个或多个 α-氢的烷基醛的 Hantzsch 1,4-二氢吡啶,通过提出的自氧化机制获得了脱烷基产物。DOI:10.3184/174751918x15208600846791
文献信息
-
Synthesis of Spin-Labelled 1,4-Dihydropyridines and Pyridines作者:Olga H. Hankovszky、Cecilia P. Sár、Kálmán Hideg、Gyula JerkovichDOI:10.1055/s-1991-26389日期:——1,4-Dihydropyridines spin-labelled with 5- and 6-membered nitroxyl radicals in positions 1-4 of the pyridine ring were synthesized. The oxidation of these dihydropyridines to pyridines with active manganese dioxide was investigated.
-
Cobalt and Manganese Salts of p -Aminobenzoic Acid Supported on Silica Gel: A Versatile Catalyst for Oxidation by Molecular Oxygen作者:Mohammed M. Hashemi、Yusef AhmadibeniDOI:10.1007/s00706-002-0534-3日期:2003.2.1A 1:1 molar ratio of the cobalt and manganese salts of p -amino benzoic acid supported on silica gel is an effective catalyst for the oxidation of various organic compounds in reasonable yields using molecular oxygen. The catalyst can be reused several times.
-
Laccase-catalyzed oxidation of Hantzsch 1,4-dihydropyridines to pyridines and a new one pot synthesis of pyridines作者:Heba T. Abdel-Mohsen、Jürgen Conrad、Uwe BeifussDOI:10.1039/c2gc35950b日期:——laccase-catalyzed oxidation of 1,4-dihydropyridines to pyridines using aerial O2 as the oxidant exclusively delivers pyridines with yields up to 95% under mild reaction conditions. Combination of the Hantzsch 1,4-dihydropyridine synthesis with the newly developed laccase-catalyzed oxidation forms the basis of a facile and environmentally benign method for the synthesis of pyridines in one pot.
-
A new oxidation system for the oxidation of Hantzsch-1,4-dihydropyridines and polyhydroquinoline derivatives under mild conditions作者:Cui-Bing Bai、Nai-Xing Wang、Yan-Jing Wang、Xing-Wang Lan、Yalan Xing、Jia-Long WenDOI:10.1039/c5ra20977c日期:——
An oxidation system to synthesize pyridine derivatives has been developed by oxidation of 1,4-dihydropyridines and polyhydroquinoline derivatives in good yields.
-
A biomass-derived N-doped porous carbon catalyst for the aerobic dehydrogenation of nitrogen heterocycles作者:Jing-Jiang Liu、Fu-Hu Guo、Fu-Jun Cui、Ji-Hua Zhu、Xiao-Yu Liu、Arif Ullah、Xi-Cun Wang、Zheng-Jun QuanDOI:10.1039/d1nj05411b日期:——superior catalytic performance in the aerobic dehydrogenation of various heterocyclic nitrogen compounds (49 examples, up to 96% yield), similar to that of C3N4 and GO. Characterization by TEM, BET and XPS accompanied by the EPR analysis revealed that the enhanced catalytic properties of NC came from its high activation ability for both O2 and heterocyclic nitrogen, attributed to the porous structure and
表征谱图
-
氢谱1HNMR
-
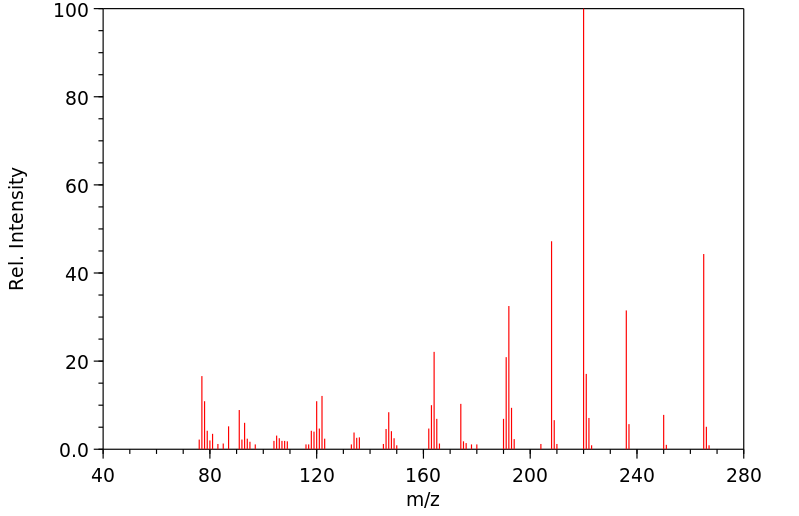
质谱MS
-
碳谱13CNMR
-
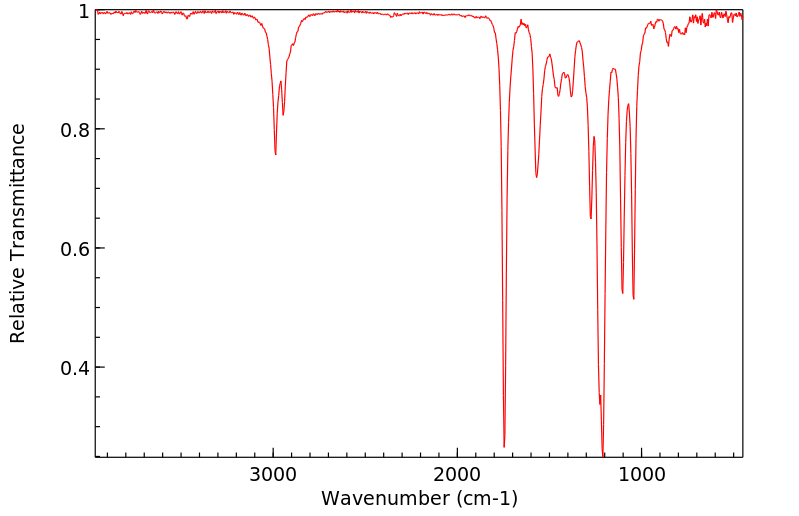
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-氨氯地平-d4
(R,S)-可替宁N-氧化物-甲基-d3
(R)-(+)-2,2'',6,6''-四甲氧基-4,4''-双(二苯基膦基)-3,3''-联吡啶(1,5-环辛二烯)铑(I)四氟硼酸盐
(R)-N'-亚硝基尼古丁
(R)-DRF053二盐酸盐
(5E)-5-[(2,5-二甲基-1-吡啶-3-基-吡咯-3-基)亚甲基]-2-亚磺酰基-1,3-噻唑烷-4-酮
(5-溴-3-吡啶基)[4-(1-吡咯烷基)-1-哌啶基]甲酮
(5-氨基-6-氰基-7-甲基[1,2]噻唑并[4,5-b]吡啶-3-甲酰胺)
(2S,2'S)-(-)-[N,N'-双(2-吡啶基甲基]-2,2'-联吡咯烷双(乙腈)铁(II)六氟锑酸盐
(2S)-2-[[[9-丙-2-基-6-[(4-吡啶-2-基苯基)甲基氨基]嘌呤-2-基]氨基]丁-1-醇
(2R,2''R)-(+)-[N,N''-双(2-吡啶基甲基)]-2,2''-联吡咯烷四盐酸盐
(1'R,2'S)-尼古丁1,1'-Di-N-氧化物
黄色素-37
麦斯明-D4
麦司明
麝香吡啶
鲁非罗尼
鲁卡他胺
高氯酸N-甲基甲基吡啶正离子
高氯酸,吡啶
高奎宁酸
马来酸溴苯那敏
马来酸氯苯那敏-D6
马来酸左氨氯地平
顺式-双(异硫氰基)(2,2'-联吡啶基-4,4'-二羧基)(4,4'-二-壬基-2'-联吡啶基)钌(II)
顺式-二氯二(4-氯吡啶)铂
顺式-二(2,2'-联吡啶)二氯铬氯化物
顺式-1-(4-甲氧基苄基)-3-羟基-5-(3-吡啶)-2-吡咯烷酮
顺-双(2,2-二吡啶)二氯化钌(II) 水合物
顺-双(2,2'-二吡啶基)二氯化钌(II)二水合物
顺-二氯二(吡啶)铂(II)
顺-二(2,2'-联吡啶)二氯化钌(II)二水合物
韦德伊斯试剂
非那吡啶
非洛地平杂质C
非洛地平
非戈替尼
非布索坦杂质66
非尼拉朵
非尼拉敏
雷索替丁
阿雷地平
阿瑞洛莫
阿扎那韦中间体
阿培利司N-6
阿伐曲波帕杂质40
间硝苯地平
间-硝苯地平
镉,二碘四(4-甲基吡啶)-
锌,二溴二[4-吡啶羧硫代酸(2-吡啶基亚甲基)酰肼]-