2,6-二氯甲基吡啶 | 3099-28-3
中文名称
2,6-二氯甲基吡啶
中文别名
2,6-双(氯甲基)吡啶;α,α'-二氯-2,6-二甲基吡啶;2,6-双(氯甲基)吡啶
英文名称
2,6-bis(chloromethyl)pyridine
英文别名
——
CAS
3099-28-3
化学式
C7H7Cl2N
mdl
MFCD00142844
分子量
176.045
InChiKey
IWQNFYRJSVJWQA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:73-78 °C (lit.)
-
沸点:110 °C / 3mmHg
-
密度:1.275±0.06 g/cm3(Predicted)
-
稳定性/保质期:
遵照规定使用和储存,则不会分解。
计算性质
-
辛醇/水分配系数(LogP):1.7
-
重原子数:10
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.285
-
拓扑面积:12.9
-
氢给体数:0
-
氢受体数:1
安全信息
-
安全说明:S26,S36/37/39
-
危险品运输编号:UN3335 9
-
WGK Germany:3
-
海关编码:2933399090
-
危险品标志:Xn
-
危险类别码:R22
-
危险标志:GHS05,GHS07
-
危险性描述:H302,H315,H318,H335
-
危险性防范说明:P261,P280,P305 + P351 + P338
-
包装等级:III
-
危险类别:8
-
储存条件:存于阴凉干燥处。
SDS
Section 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE
Product identifiers
Product name : 2,6-Bis(chloromethyl)pyridine
CAS-No. : 3099-28-3
Relevant identified uses of the substance or mixture and uses advised against
Identified uses : Laboratory chemicals, Manufacture of substances
Section 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
Classification according to Regulation (EC) No 1272/2008 [EU-GHS/CLP]
Acute toxicity, Oral (Category 4)
Skin irritation (Category 2)
Serious eye damage (Category 1)
Specific target organ toxicity - single exposure (Category 3)
Classification according to EU Directives 67/548/EEC or 1999/45/EC
Harmful if swallowed. Irritating to respiratory system and skin. Risk of serious damage to eyes.
Label elements
Labelling according Regulation (EC) No 1272/2008 [CLP]
Pictogram
Signal word Danger
Hazard statement(s)
H302 Harmful if swallowed.
H315 Causes skin irritation.
H318 Causes serious eye damage.
H335 May cause respiratory irritation.
Precautionary statement(s)
P261 Avoid breathing dust/ fume/ gas/ mist/ vapours/ spray.
P280 Wear protective gloves/ eye protection/ face protection.
P305 + P351 + P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove
contact lenses, if present and easy to do. Continue rinsing.
Supplemental Hazard none
Statements
According to European Directive 67/548/EEC as amended.
Hazard symbol(s)
R-phrase(s)
R22 Harmful if swallowed.
R37/38 Irritating to respiratory system and skin.
R41 Risk of serious damage to eyes.
S-phrase(s)
S26 In case of contact with eyes, rinse immediately with plenty of water and
seek medical advice.
S36/37/39 Wear suitable protective clothing, gloves and eye/face protection.
Other hazards
Lachrymator.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
: α,α′-Dichloro-2,6-lutidine
Synonyms
Formula : C7H7Cl2N
Molecular Weight : 176,04 g/mol
Component Concentration
2,6-Bis(chloromethyl)pyridine
CAS-No. 3099-28-3 -
Section 4. FIRST AID MEASURES
Description of first aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician.
In case of skin contact
Wash off with soap and plenty of water. Consult a physician.
In case of eye contact
Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician.
If swallowed
Never give anything by mouth to an unconscious person. Rinse mouth with water. Consult a physician.
Most important symptoms and effects, both acute and delayed
To the best of our knowledge, the chemical, physical, and toxicological properties have not been
thoroughly investigated.
Indication of any immediate medical attention and special treatment needed
no data available
Section 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
Special hazards arising from the substance or mixture
Carbon oxides, nitrogen oxides (NOx), Hydrogen chloride gas
Advice for firefighters
Wear self contained breathing apparatus for fire fighting if necessary.
Further information
no data available
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid dust formation. Avoid breathing vapors, mist or gas. Ensure
adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust.
Environmental precautions
Do not let product enter drains.
Methods and materials for containment and cleaning up
Pick up and arrange disposal without creating dust. Sweep up and shovel. Keep in suitable, closed
containers for disposal.
Reference to other sections
For disposal see section 13.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Avoid contact with skin and eyes. Avoid formation of dust and aerosols.
Provide appropriate exhaust ventilation at places where dust is formed.Normal measures for preventive fire
protection.
Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place.
Specific end uses
no data available
Section 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Control parameters
Components with workplace control parameters
Exposure controls
Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and
at the end of workday.
Personal protective equipment
Eye/face protection
Face shield and safety glasses Use equipment for eye protection tested and approved under
appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protection
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique
(without touching glove's outer surface) to avoid skin contact with this product. Dispose of
contaminated gloves after use in accordance with applicable laws and good laboratory practices.
Wash and dry hands.
The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and
the standard EN 374 derived from it.
Body Protection
Complete suit protecting against chemicals, The type of protective equipment must be selected
according to the concentration and amount of the dangerous substance at the specific workplace.
Respiratory protection
Where risk assessment shows air-purifying respirators are appropriate use a full-face particle
respirator type N100 (US) or type P3 (EN 143) respirator cartridges as a backup to engineering
controls. If the respirator is the sole means of protection, use a full-face supplied air respirator. Use
respirators and components tested and approved under appropriate government standards such
as NIOSH (US) or CEN (EU).
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
a) Appearance Form: crystalline
Colour: beige
b) Odour no data available
c) Odour Threshold no data available
d) pH no data available
e) Melting point/freezing Melting point/range: 73 - 78 °C - lit.
point
f) Initial boiling point and no data available
boiling range
g) Flash point no data available
h) Evaporation rate no data available
i) Flammability (solid, gas) no data available
j) Upper/lower no data available
flammability or
explosive limits
k) Vapour pressure no data available
l) Vapour density no data available
m) Relative density no data available
n) Water solubility no data available
o) Partition coefficient: n- log Pow: 1,797
octanol/water
p) Autoignition no data available
temperature
q) Decomposition no data available
temperature
r) Viscosity no data available
s) Explosive properties no data available
t) Oxidizing properties no data available
Other safety information
no data available
Section 10. STABILITY AND REACTIVITY
Reactivity
no data available
Chemical stability
no data available
Possibility of hazardous reactions
no data available
Conditions to avoid
no data available
Incompatible materials
Oxidizing agents
Hazardous decomposition products
Other decomposition products - no data available
Section 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity
no data available
Skin corrosion/irritation
no data available
Serious eye damage/eye irritation
no data available
Respiratory or skin sensitization
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
IARC: No component of this product present at levels greater than or equal to 0.1% is identified as
probable, possible or confirmed human carcinogen by IARC.
Reproductive toxicity
no data available
Specific target organ toxicity - single exposure
Inhalation - May cause respiratory irritation.
Specific target organ toxicity - repeated exposure
no data available
Aspiration hazard
no data available
Potential health effects
Inhalation May be harmful if inhaled. Causes respiratory tract irritation.
Ingestion Harmful if swallowed.
Skin May be harmful if absorbed through skin. Causes skin irritation.
Eyes Causes eye burns.
Signs and Symptoms of Exposure
To the best of our knowledge, the chemical, physical, and toxicological properties have not been
thoroughly investigated.
Additional Information
RTECS: Not available
Section 12. ECOLOGICAL INFORMATION
Toxicity
no data available
Persistence and degradability
no data available
Bioaccumulative potential
no data available
Mobility in soil
no data available
Results of PBT and vPvB assessment
no data available
Other adverse effects
no data available
Section 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company. Contact a licensed
professional waste disposal service to dispose of this material. Dissolve or mix the material with a
combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber.
Contaminated packaging
Dispose of as unused product.
Section 14. TRANSPORT INFORMATION
UN number
ADR/RID: - IMDG: - IATA: 3335
UN proper shipping name
ADR/RID: Not dangerous goods
IMDG: Not dangerous goods
IATA: Aviation regulated solid, n.o.s. (2,6-Bis(chloromethyl)pyridine)
Transport hazard class(es)
ADR/RID: - IMDG: - IATA: 9
Packaging group
ADR/RID: - IMDG: - IATA: -
Environmental hazards
ADR/RID: no IMDG Marine pollutant: no IATA: no
Special precautions for user
no data available
Section 15. REGULATORY INFORMATION
This safety datasheet complies with the requirements of Regulation (EC) No. 1907/2006.
Safety, health and environmental regulations/legislation specific for the substance or mixture
no data available
Chemical Safety Assessment
no data available
Section 16. OTHER INFORMATION
Further information
Copyright 2012 Co. LLC. License granted to make unlimited paper copies for internal use
only.
The above information is believed to be correct but does not purport to be all inclusive and shall be
used only as a guide. The information in this document is based on the present state of our knowledge
and is applicable to the product with regard to appropriate safety precautions. It does not represent any
guarantee of the properties of the product. Corporation and its Affiliates shall not be held
liable for any damage resulting from handling or from contact with the above product. See
and/or the reverse side of invoice or packing slip for additional terms and conditions of sale.
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 2-(Chloromethyl)-6-(dichloromethyl)pyridine 56533-58-5 C7H6Cl3N 210.49 2,6-二甲基吡啶 2,6-dimethylpyridine 108-48-5 C7H9N 107.155 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2,6-二(氯甲基)吡啶1-氧化物 2,6-bis(chloromethyl)pyridine N-oxide 201160-41-0 C7H7Cl2NO 192.045 —— 2-(azidomethyl)-6-(chloromethyl)pyridine 628308-62-3 C7H7ClN4 182.612
反应信息
-
作为反应物:描述:2,6-二氯甲基吡啶 在 sodium hydride 作用下, 以 四氢呋喃 为溶剂, 反应 16.0h, 生成 2,4,6,9,13,16-hexaoxa-11(2,6)-pyridina-1,5(2,3)-dinaphthalena-3(1,2)-benzenacyclohexadecaphane compound with tetrahydrofuran (1:1)参考文献:名称:Macroring-neutral molecule complexation. Synthesis of biconcave pyridino hosts, complex formation, and x-ray crystal structures of two inclusion compounds摘要:A series of pyridino macrocycles 1-3(a-e) incorporating rigid bi- and triaryl ether segments in different ring positions have been synthesized. The effect of incorporation of these building blocks into a given macroring framework on the host properties for uncharged-molecule inclusion has been studied. Symmetric 21-membered macrorings 1c, 1d, or 2c, 2d with tri-o-phenylene, tri-2,3-naphthylene, or mixed phenylene naphthylene ether units are efficient hosts in solid-state complexation of dipolar-aprotic and apolar guests such as nitro compounds and nitriles as well as DMF, DMSO, THF, dioxane, or benzene. X-ray analyses of the solid-state complexes of 1d with PhNO2 (1:1) and MeCN (1:1) have been studied. It is shown that the presence of the triaryl ether segment induces a biconcave nonequally sided host conformation suitable for guest inclusion. The packing characterizes the host-guest topologies of the two inclusion compounds to be of (H-bonded) cavitate-type for the MeCN case and of interstitial channel type for the PhNO2 case. A comparative discussion of the present and previous results support the interpretation of hemispherand preorganization for the new host structures.DOI:10.1021/jo00003a055
-
作为产物:描述:参考文献:名称:大环二氧戊胺:用于 1:1 镍 (II)-氧 (Ni(II)-O2) 加合物形成的新型配体摘要:La pentaaza-1,4,7,10,13 cyclohexadecanedione-14,16 forme des complexes stables 1:1 avec Ni(II) et Cu(II) possedant 2 donneurs deprotones en position equatoriale。Calcul des potentiels氧化还原。Cinetique de la fixation d'O 2 ensolutions aqueusesDOI:10.1021/ja00331a018
文献信息
-
腺苷受体拮抗剂
-
[EN] PROCESSES FOR MAKING TRIAZOLO[4,5D] PYRAMIDINE DERIVATIVES AND INTERMEDIATES THEREOF<br/>[FR] PROCÉDÉS DE PREPARATION DE DÉRIVÉS DE TRIAZOLO [4,5 D] PYRIMIDINE ET INTERMÉDIAIRES DE CEUX-CI
-
Design and Synthesis of Sn-Porphyrin Based Molecular Gates作者:Aurélie Guenet、Ernest Graf、Nathalie Kyritsakas、Mir Wais HosseiniDOI:10.1021/ic902265e日期:2010.2.15The design, synthesis, and structural characterization, both in solution by 1H NMR and in the solid state by X-ray diffraction on single crystals, of a series of molecular gates based on Sn-porphyrin derivatives are presented. The molecular system is based on a porphyrin core bearing at the meso positions either phenyl or pyridyl groups as a stator, octahedral Sn(IV) cation located at the center of
-
[EN] AMINOPYRAZINE COMPOUNDS WITH A2A ANTAGONIST PROPERTIES<br/>[FR] COMPOSÉS AMINOPYRAZINES AYANT DES PROPRIÉTÉS ANTAGONISTES DE L'A2A申请人:MERCK SHARP & DOHME公开号:WO2016081290A1公开(公告)日:2016-05-26Disclosed are compounds of Formula A and Formula A-1, or a salt thereof, and pharmaceutical formulations (pharmaceutical compositions) comprising those compounds, or a salt thereof; wherein "R1", "RA-1", "R2", "R3", and "Het" are defined herein above, which compounds are believed suitable for use in selectively antagonizing the A2a receptors, for example, those found in high density in the basal ganglia. Such compounds and pharmaceutical formulations are believed to be useful in treatment or management of neurodegenerative diseases, for example, Parkinson's disease, or movement disorders arising from use of certain medications used in the treatment or management of Parkinson's disease.
-
Pharmaceutically active compounds申请人:Astra Aktiebolag公开号:US06153641A1公开(公告)日:2000-11-28The present invention provides novel compounds of the formula (I): R.sub.20 --(CH.sub.2).sub.n --R (I) wherein: T.sub.20 is a bisindolylmaleimide moiety linked to the --(CH.sub.2).sub.n -group through an indolyl nitrogen, n is 0 or 1, R is a 5 or 6 membered aromatic carbocyclic or heterocyclic ring, the heterocyclic ring containing N or S, R is substituted by R.sub.1 and up to four of R.sub.2, R.sub.3, R.sub.4 and R.sub.5, wherein; R.sub.1 is aminomethyl, (N--(C.sub.1-4 -alkyl)amino)methyl, (N,N-di(C.sub.1-4 -alkyl)amino)methyl or pyridiniummethyl, and R.sub.2, R.sub.3, and R.sub.4 and R.sub.5 (if present), which may be the same or different, are each hydrogen, hydroxy, C.sub.1-4 -alkoxy, C.sub.1-4 -alkoxy) or halogen, or R.sub.2 when in a position contiguous to the bond connecting R to the --(CH.sub.2).sub.n -- group and n is 1 may, together with the 2-carbon atom on the indole to which the --(CH.sub.2) group is attached, form a ring, and pharmaceutically acceptable salts thereof; and the use of such compounds in medical therapy.本发明提供了一种新颖的公式(I)化合物:R.sub.20 --(CH.sub.2).sub.n --R (I) 其中:T.sub.20 是一个双吲哚基马来酰亚胺部分,通过吲哚基氮与--(CH.sub.2).sub.n -组连接,n为0或1,R是一个5或6成员的芳香碳环或杂环,杂环含有N或S,R被R.sub.1和最多四个R.sub.2,R.sub.3,R.sub.4和R.sub.5取代,其中;R.sub.1是氨基甲基,(N--(C.sub.1-4 -烷基)氨基)甲基,(N,N-二(C.sub.1-4 -烷基)氨基)甲基或吡啶甲基,R.sub.2,R.sub.3和R.sub.4和R.sub.5(如果存在),可能是相同的或不同的,每一个都是氢,羟基,C.sub.1-4 -烷氧基,C.sub.1-4 -烷氧基)或卤素,或者当R.sub.2处于连接R到--(CH.sub.2).sub.n --组的键的邻近位置且n为1时,R.sub.2可以与吲哚上连接--(CH.sub.2)组的2-碳原子一起形成一个环,以及它们的药用盐;以及将这些化合物用于医疗治疗的使用。
表征谱图
-
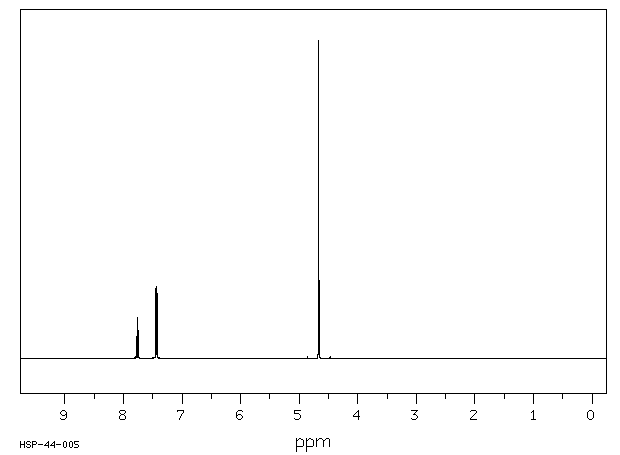
氢谱1HNMR
-
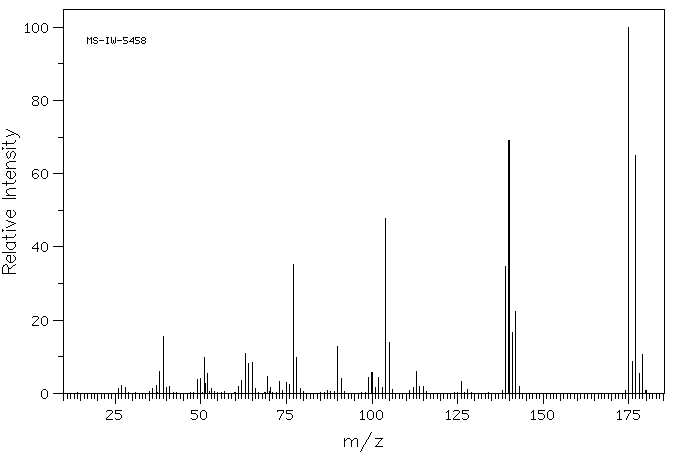
质谱MS
-
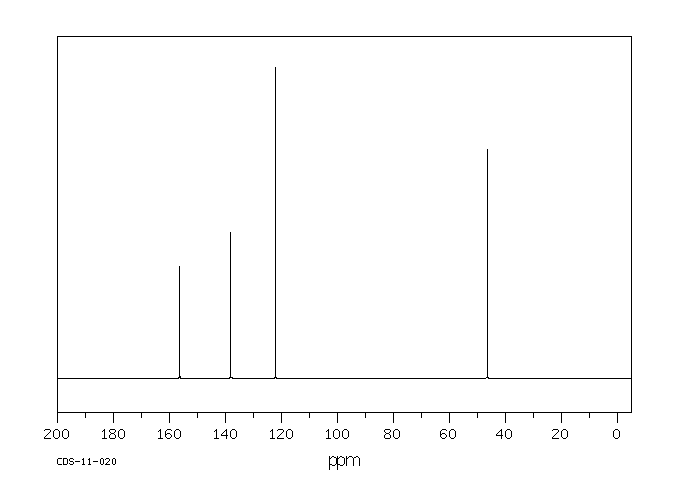
碳谱13CNMR
-
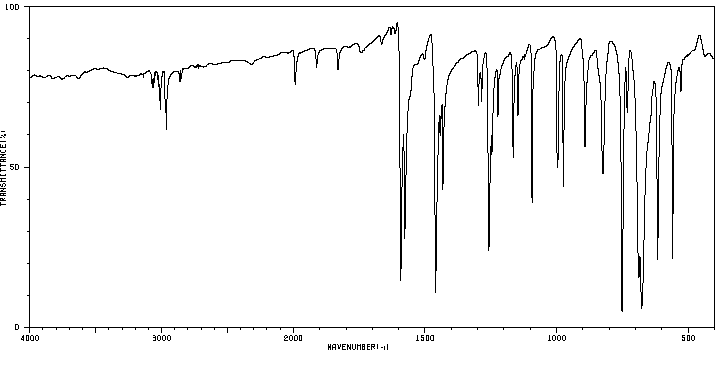
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-氨氯地平-d4
(R,S)-可替宁N-氧化物-甲基-d3
(R)-(+)-2,2'',6,6''-四甲氧基-4,4''-双(二苯基膦基)-3,3''-联吡啶(1,5-环辛二烯)铑(I)四氟硼酸盐
(R)-N'-亚硝基尼古丁
(R)-DRF053二盐酸盐
(5E)-5-[(2,5-二甲基-1-吡啶-3-基-吡咯-3-基)亚甲基]-2-亚磺酰基-1,3-噻唑烷-4-酮
(5-溴-3-吡啶基)[4-(1-吡咯烷基)-1-哌啶基]甲酮
(5-氨基-6-氰基-7-甲基[1,2]噻唑并[4,5-b]吡啶-3-甲酰胺)
(2S,2'S)-(-)-[N,N'-双(2-吡啶基甲基]-2,2'-联吡咯烷双(乙腈)铁(II)六氟锑酸盐
(2S)-2-[[[9-丙-2-基-6-[(4-吡啶-2-基苯基)甲基氨基]嘌呤-2-基]氨基]丁-1-醇
(2R,2''R)-(+)-[N,N''-双(2-吡啶基甲基)]-2,2''-联吡咯烷四盐酸盐
(1'R,2'S)-尼古丁1,1'-Di-N-氧化物
黄色素-37
麦斯明-D4
麦司明
麝香吡啶
鲁非罗尼
鲁卡他胺
高氯酸N-甲基甲基吡啶正离子
高氯酸,吡啶
高奎宁酸
马来酸溴苯那敏
马来酸氯苯那敏-D6
马来酸左氨氯地平
顺式-双(异硫氰基)(2,2'-联吡啶基-4,4'-二羧基)(4,4'-二-壬基-2'-联吡啶基)钌(II)
顺式-二氯二(4-氯吡啶)铂
顺式-二(2,2'-联吡啶)二氯铬氯化物
顺式-1-(4-甲氧基苄基)-3-羟基-5-(3-吡啶)-2-吡咯烷酮
顺-双(2,2-二吡啶)二氯化钌(II) 水合物
顺-双(2,2'-二吡啶基)二氯化钌(II)二水合物
顺-二氯二(吡啶)铂(II)
顺-二(2,2'-联吡啶)二氯化钌(II)二水合物
韦德伊斯试剂
非那吡啶
非洛地平杂质C
非洛地平
非戈替尼
非布索坦杂质66
非尼拉朵
非尼拉敏
雷索替丁
阿雷地平
阿瑞洛莫
阿扎那韦中间体
阿培利司N-6
阿伐曲波帕杂质40
间硝苯地平
间-硝苯地平
镉,二碘四(4-甲基吡啶)-
锌,二溴二[4-吡啶羧硫代酸(2-吡啶基亚甲基)酰肼]-