对异丁基甲苯 | 5161-04-6
中文名称
对异丁基甲苯
中文别名
1-甲基-4-异丁基苯
英文名称
1-isobutyl-4-methylbenzene
英文别名
1-methyl-4-(2-methylpropyl)benzene;4-Methyl-isobutylbenzol;Benzene, 1-methyl-4-(2-methylpropyl)-
CAS
5161-04-6
化学式
C11H16
mdl
MFCD00060990
分子量
148.248
InChiKey
VCGBZXLLPCGFQM-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:-44.72°C (estimate)
-
沸点:196°C
-
密度:0.8478
-
保留指数:1089.1;1105;1134;1149.1;1091.7;1133.6;1148
-
稳定性/保质期:
计算性质
-
辛醇/水分配系数(LogP):3.9
-
重原子数:11
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.454
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
海关编码:2902909090
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 alpha-异丙基-4-甲基苄醇 2-methyl-1-(p-tolyl)propan-1-ol 18228-44-9 C11H16O 164.247 2,4'-二甲基苯丙酮 4'-methylisobutyrophenone 50390-51-7 C11H14O 162.232 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 异丁芬酸 ibufenac 1553-60-2 C12H16O2 192.258
反应信息
-
作为反应物:描述:参考文献:名称:A one-pot synthesis of ibuprofene involving three consecutive steps of superbase metalation摘要:A one-pot reaction sequence consisting of three consecutive metalation and electrophilic substitution stages leads to 2-(4-isobutylphenyl)propanoic acid with 52% over-all yield. A crucial step is the alkylation of deprotonated p-ethyltoluene with isopropyl bromide. In general terms, sec-alkyl halides and benzyl or allyl type alkalimetal reagents undergo coupling reactions with surprising ease.DOI:10.1016/s0040-4039(00)92708-4
-
作为产物:参考文献:名称:Ruechardt,C., Chemische Berichte, 1961, vol. 94, p. 2609 - 2623摘要:DOI:
文献信息
-
一种以P(O)-OH化合物与含甲基取代芳香烃 制备有机磷酸酯类化合物的方法申请人:湖南理工学院公开号:CN107602609B公开(公告)日:2020-07-21
-
Coupling of Reformatsky Reagents with Aryl Chlorides Enabled by Ylide‐Functionalized Phosphine Ligands作者:Zhiyong Hu、Xiao‐Jing Wei、Jens Handelmann、Ann‐Katrin Seitz、Ilja Rodstein、Viktoria H. Gessner、Lukas J. GooßenDOI:10.1002/anie.202016048日期:2021.3.15organozinc reagents with aryl electrophiles using a cyclohexyl‐YPhos ligand bearing an ortho‐tolyl‐substituent in the backbone. This highly electron‐rich, bulky ligand enables the use of aryl chlorides in room temperature couplings of Reformatsky reagents. The reaction scope covers diversely functionalized arylacetic and arylpropionic acid derivatives. Aryl bromides and chlorides can be converted selectively
-
Role of Sterically Demanding Chiral Dirhodium Catalysts in Site-Selective C–H Functionalization of Activated Primary C–H Bonds作者:Changming Qin、Huw M. L. DaviesDOI:10.1021/ja504797x日期:2014.7.9sterically demanding dirhodium tetracarboxylate catalysts on the site selectivity of C-H functionalization by means of rhodium carbene-induced C-H insertion is described. The established dirhodium tetraprolinate-catalyzed reactions of aryldiazoacetates cause preferential C-H functionalization of secondary C-H bonds as a result of competing steric and electronic effects. The sterically more demanding dirhodium
-
An unprecedented iron-catalyzed cross-coupling of primary and secondary alkyl Grignard reagents with non-activated aryl chlorides作者:Marc C. Perry、Amber N. Gillett、Tyler C. LawDOI:10.1016/j.tetlet.2012.06.048日期:2012.8The use of N-heterocyclic carbene ligands in the iron-catalyzed cross-coupling of alkyl Grignards has allowed, for the first time, coupling of non-activated, electron rich aryl chlorides. Surprisingly, the tetrahydrate of FeCl2 was found to be a better pre-catalyst than anhydrous FeCl2. Primary Grignard reagents coupled in excellent yields while secondary Grignard reagents coupled in modest yields
-
Ligand-free nickel-catalyzed Kumada couplings of aryl bromides with tert-butyl Grignard reagents作者:Zhenghan Wu、Tengda Si、Guangqing Xu、Bin Xu、Wenjun TangDOI:10.1016/j.cclet.2018.12.027日期:2019.3Abstract A ligand-free nickel-catalyzed Kumada cross-coupling of aryl bromides and tert-butyl Grignard reagents led to the formation of a series of tert-butyl aryls in moderate to good yields, excellent tBu/iBu ratios, and good functional group compatibility. A radical coupling process is indicated and a mechanism with a Ni(I)-Ni(III) catalytic cycle is proposed.
表征谱图
-
氢谱1HNMR
-
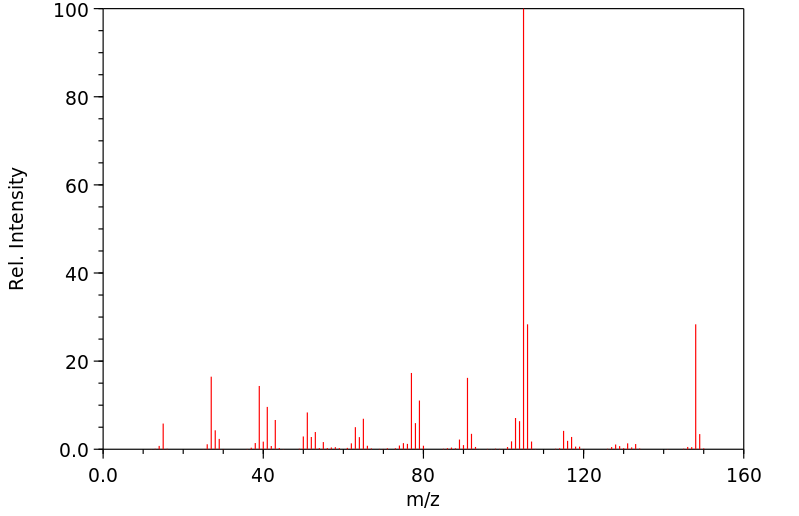
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫