2-(甲硫基)噻吩 | 5780-36-9
中文名称
2-(甲硫基)噻吩
中文别名
2-(甲基硫代)噻吩;2-甲硫基噻吩
英文名称
2-(methylsulfanyl)thiophene
英文别名
2-(methylthio)thiophene;2-methylmercaptothiophene;2-methylsulfanylthiophene
CAS
5780-36-9
化学式
C5H6S2
mdl
——
分子量
130.235
InChiKey
ZLSMPEVZXWDWEK-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:100 °C
-
密度:1.1752 g/cm3
-
闪点:82-84°C/15mm
-
保留指数:1035;1014;1014;1019;1036;1040;1045;1020
-
稳定性/保质期:
在常温常压下保持稳定
计算性质
-
辛醇/水分配系数(LogP):2.4
-
重原子数:7
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.2
-
拓扑面积:53.5
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:9
-
危险品标志:Xi
-
危险品运输编号:UN3334
-
海关编码:2934999090
-
危险类别:9
-
安全说明:S23,S24/25
-
危险性防范说明:P261,P280,P305+P351+P338,P304+P340,P405,P501
-
危险性描述:H319,H335
-
储存条件:常温下,应避免光照,存放在阴凉干燥处,并密封保存。
SDS
| Name: | 2-(Methylthio)thiophene 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 5780-36-9 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 5780-36-9 | 2-(Methylthio)thiophene | 97% | unlisted |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Not available.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
May cause respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 5780-36-9: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: yellow
Odor: stench - pungent odor
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 82 - 84 deg C @15mmHg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C5H6S2
Molecular Weight: 130
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, oxides of sulfur, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 5780-36-9 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-(Methylthio)thiophene - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: AVIATION REGULATED LIQUID, N.O.S.*
Hazard Class: 9
UN Number: 3334
Packing Group:
IMO
Shipping Name:
Hazard Class:
UN Number: 3334
Packing Group:
RID/ADR
Shipping Name:
Hazard Class:
UN Number: 3334
Packing group:
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 5780-36-9: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 5780-36-9 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 5780-36-9 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 2-ethylthiophene 6911-35-9 C6H8S2 144.262 2-噻吩硫醇 Thiophene-2-thiol 7774-74-5 C4H4S2 116.208 —— 2-(methylsulfinyl)thiophene 74166-42-0 C5H6OS2 146.234 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2,5-bis(methylsulfanyl)thiophene 50878-66-5 C6H8S3 176.328 —— 2-(methylsulfinyl)thiophene 74166-42-0 C5H6OS2 146.234 —— 5-Brom-2-methylmercapto-thiophen 86369-96-2 C5H5BrS2 209.131 —— 5-methyl-2-(methylthio)thiophene 40990-29-2 C6H8S2 144.262
反应信息
-
作为反应物:参考文献:名称:潜在的噻吩化学疗法。第三部分 一些5-取代的2-噻吩基硫醚和砜摘要:DOI:10.1039/jr9540000237
-
作为产物:参考文献:名称:Steinkopf; Leonhardt, Justus Liebigs Annalen der Chemie, 1932, vol. 495, p. 166,170摘要:DOI:
-
作为试剂:描述:参考文献:名称:Metal-free regioselective hydrochlorination of unactivated alkenesviaa combined acid catalytic system摘要:一个结合了盐酸/HCl、DMPU和乙酸的催化体系被用于对一系列未活化的烯烃进行氯化反应。DOI:10.1039/c7gc03665e
文献信息
-
Modulation of photochemical oxidation of thioethers to sulfoxides or sulfones using an aromatic ketone as the photocatalyst作者:Bin Zhao、Gerald B. Hammond、Bo XuDOI:10.1016/j.tetlet.2021.153376日期:2021.10We have developed an eco-friendly and chemo-selective photocatalytic synthesis of sulfoxides or sulfones via oxidation of sulfides (thioethers) at ambient temperature using air or O2 as the oxidant. An inexpensive thioxanthone was used as the photocatalyst. Our method offers excellent chemical yields and good functional group tolerance. The hydrogen bonding between hexafluoro-2-propanol (HFIP) and
-
Alloxan-Catalyzed Biomimetic Oxidations with Hydrogen Peroxide or Molecular Oxygen作者:Shiqi Zhang、Guangxun Li、Ling Li、Xiongfei Deng、Gang Zhao、Xin Cui、Zhuo TangDOI:10.1021/acscatal.9b04508日期:2020.1.3biomimetic catalysts for various oxidations, catalyzing oxidations of sulfides and amines with hydrogen peroxide or molecular oxygen under mild conditions with high yields in a short time. The whole catalytic cycle has been verified to be a biomimetic approach through the formation of the alloxan hydroperoxide reactive intermediate. Additionally, encouraging asymmetric catalytic results have been obtained
-
Synergistic cooperative effect of CF<sub>3</sub>SO<sub>2</sub>Na and bis(2-butoxyethyl)ether towards selective oxygenation of sulfides with molecular oxygen under visible-light irradiation作者:Kai-Jian Liu、Zheng Wang、Ling-Hui Lu、Jin-Yang Chen、Fei Zeng、Ying-Wu Lin、Zhong Cao、Xianyong Yu、Wei-Min HeDOI:10.1039/d0gc02663h日期:——visible-light-initiated oxygenation of sulfides at ambient temperature under transition-metal-, additives-free and minimal solvent conditions. The synergistic catalytic efforts between CF3SO2Na and 2-butoxyethyl ether represents the key promoting factor for the reaction.
-
Structural elaboration of dicyanopyrazine: towards push–pull molecules with tailored photoredox activity作者:Zuzana Hloušková、Milan Klikar、Oldřich Pytela、Numan Almonasy、Aleš Růžička、Veronika Jandová、Filip BurešDOI:10.1039/c9ra04731j日期:——provided a series of catalysts based on a pyrazine heterocyclic scaffold with easy synthesis and further modification, diverse photoredox characteristics and wide application potential across modern photoredox transformations. The photoredox catalytic activities of the target catalysts were examined in a benchmark cross-dehydrogenative coupling and novel and challenging annulation reactions.
-
[EN] PYRIMIDINE COMPOUNDS AS TUBERCULOSIS INHIBITORS<br/>[FR] COMPOSÉS PYRIMIDINE EN TANT QU'INHIBITEURS DE LA TUBERCULOSE申请人:VERTEX PHARMA公开号:WO2011019405A1公开(公告)日:2011-02-17The present invention relates to compounds II useful as inhibitors of treating tuberculosis. The invention also provides processes for preparing compounds of the invention.本发明涉及化合物II,用作治疗结核病的抑制剂。该发明还提供了制备本发明化合物的方法。
表征谱图
-
氢谱1HNMR
-
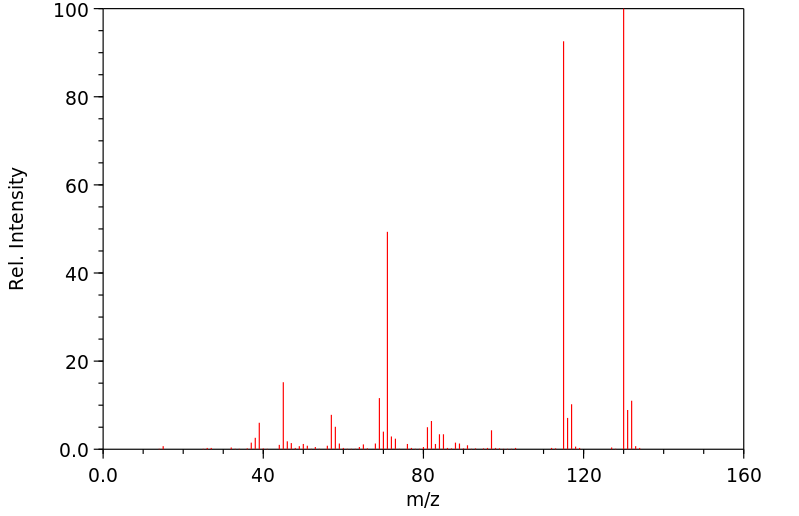
质谱MS
-
碳谱13CNMR
-
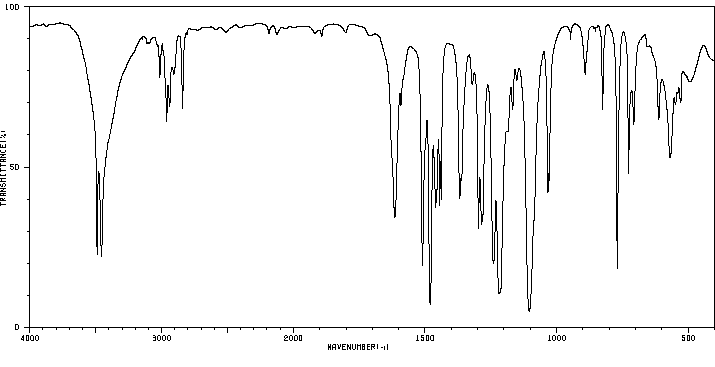
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(Rp)-2-(叔丁硫基)-1-(二苯基膦基)二茂铁
(1E)-1-{4-[(4-氨基苯基)硫烷基]苯基}乙酮肟
颜料红88
颜料紫36
顺式-1,2-二(乙硫基)-1-丙烯
非班太尔-D6
雷西那得中间体
阿西替尼杂质J
阿西替尼杂质C
阿西替尼杂质4
阿西替尼杂质
阿西替尼
阿拉氟韦
阿扎毒素
阿嗪米特
阔草特
银(I)(6-氨基-2-(甲硫基)-5-亚硝基嘧啶-4-基)酰胺水合物
钾三氟[3-(苯基硫基)丙基]硼酸酯(1-)
邻甲苯基(对甲苯基)硫化物
避虫醇
连翘脂苷B
还原红 41
还原紫3
还原桃红R
达索尼兴
辛硫醚
辛-1,7-二炔-1-基(苯基)硫烷
西嗪草酮
萘,2-[(2,3-二甲基苯基)硫代]-
莫他哌那非
茴香硫醚
苯醌B
苯酰胺,N-(氨基亚氨基甲基)-4-[(2-甲基苯基)硫代]-3-(甲磺酰)-,盐酸盐
苯酰胺,N-(氨基亚氨基甲基)-4-[(2-氯苯基)硫代]-3-(甲磺酰)-,盐酸盐
苯酰胺,N-(氨基亚氨基甲基)-4-[(2,6-二氯苯基)硫代]-3-(甲磺酰)-,盐酸盐
苯酰胺,2-[(2-硝基苯基)硫代]-
苯酚,3-氯-4-[(4-硝基苯基)硫代]-
苯酚,3-(乙硫基)-
苯酚,3,5-二[(苯基硫代)甲基]-
苯胺,4-[5-溴-3-[4-(甲硫基)苯基]-2-噻嗯基]-
苯胺,3-氯-4-[(1-甲基-1H-咪唑-2-基)硫代]-
苯胺,2-[(2-吡啶基甲基)硫代]-
苯硫醚-D10
苯硫胍
苯硫基乙酸
苯硫代磺酸S-(三氯乙烯基)酯
苯甲醇,2,3,4,5,6-五氟-a-[(苯基硫代)甲基]-,(R)-
苯甲酸,3-[[2-[(二甲氨基)甲基]苯基]硫代]-,盐酸
苯甲胺,5-氟-2-((3-甲氧苯基)硫代)-N,N-二甲基-,盐酸
苯甲二硫酸,4-溴苯基酯