N-甲基-3-硝基苯磺酰胺 | 58955-78-5
中文名称
N-甲基-3-硝基苯磺酰胺
中文别名
——
英文名称
N-methyl-3-nitrobenzenesulfonamide
英文别名
——
CAS
58955-78-5
化学式
C7H8N2O4S
mdl
MFCD00474673
分子量
216.218
InChiKey
FKHDGRMKIXBKNM-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:110-114
-
沸点:369.4±44.0 °C(Predicted)
-
密度:1.423±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):0.2
-
重原子数:14
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.142
-
拓扑面积:100
-
氢给体数:1
-
氢受体数:5
安全信息
-
危险品标志:Xi
-
海关编码:2935009090
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: N-Methyl 3-nitrobenzenesulfonamide
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: N-Methyl 3-nitrobenzenesulfonamide
CAS number: 58955-78-5
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
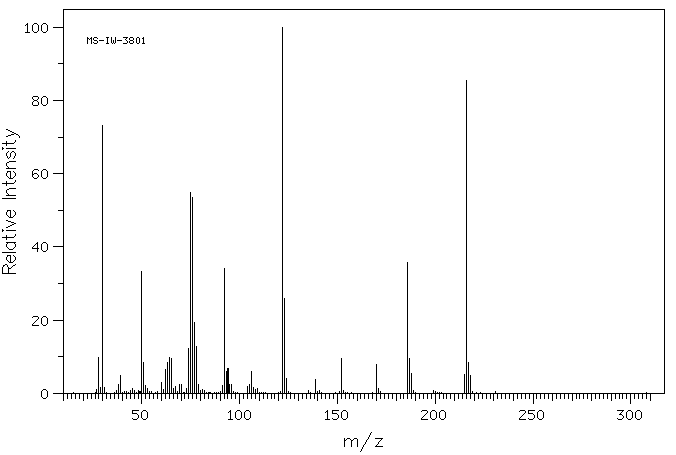
Molecular formula: C7H8N2O4S
Molecular weight: 216.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: N-Methyl 3-nitrobenzenesulfonamide
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: N-Methyl 3-nitrobenzenesulfonamide
CAS number: 58955-78-5
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C7H8N2O4S
Molecular weight: 216.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3-硝基苯磺酰氯 3-nitrobenzenesulphonyl chloride 121-51-7 C6H4ClNO4S 221.621 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 N-甲基-3-氨基苯磺酰胺 3-amino-N-methylbenzenesulfonamide 459434-40-3 C7H10N2O2S 186.235
反应信息
-
作为反应物:描述:参考文献:名称:新型3-(喹啉-4-基氨基)苯磺酰胺作为碳酸酐酶同工型I和II抑制剂的合成及生物学评价摘要:抽象的 碳酸酐酶(CAs,EC 4.2.1.1)是涉及多种生物过程的关键金属酶。我们报告了合成和生物学评估的新系列的苯磺酰胺结合未/取代的乙基喹啉-3-羧酸酯部分。新合成的化合物在体外评估为胞质人(h)亚型hCA I和II的抑制剂。此处报道的喹啉在不同程度上均抑制了hCA I和II亚型:h CA I在0.966–9.091μM范围内被K I s抑制,而hCA II在0.083–3.594μM范围内被抑制。主7-氯-6- flouro取代sulphfonamide衍生物6E(ķ我 = 0.083μM)被证明是抑制hCA II活性最强的喹啉,而其次磺酰胺类似物未能抑制hCA II直至10μM,从而证实了主要的磺酰胺类作为锌结合基团的关键作用。 CA抑制活性。DOI:10.1080/14756366.2019.1652282
-
作为产物:描述:参考文献:名称:[EN] METHODS FOR INHIBITING NECROPTOSIS
[FR] MÉTHODES POUR INHIBER LA NÉCROPTOSE摘要:本发明涉及抑制坏死程序的方法;用于识别抑制坏死程序的化合物的筛选方法;以及用于抑制坏死程序的化合物,可能在治疗与失调坏死程序相关的疾病中有用。公开号:WO2015172203A1
文献信息
-
Pyrazole compounds申请人:——公开号:US20030060453A1公开(公告)日:2003-03-27Pharmaceutical compositions and compounds are provided. The compounds of the invention demonstrate anti-proliferative activity, and may promote apoptosis in cells lacking normal regulation of cell cycle and death. In one embodiment of the invention, pharmaceutical compositions of the compounds in combination with a physiologically acceptable carrier are provided. The pharmaceutical compositions are useful in the treatment of hyperproliferative disorders, which disorders include tumor growth, lymphoproliferative diseases, angiogenesis. The compounds of the invention are substituted pyrazoles and pyrazolines.
-
Establishment of Heterolytic and Homolytic Y−NO<sub>2</sub> Bond Dissociation Energy Scales of Nitro-Containing Compounds in Acetonitrile: Chemical Origin of NO<sub>2</sub> Release and Capture作者:Xin Li、Xiao-Qing Zhu、Fan Zhang、Xiao-Xiao Wang、Jin-Pei ChengDOI:10.1021/jo702364a日期:2008.3.1The first heterolytic and homolytic N(O)−NO2 bond dissociation energy scales of three types Y−nitro (Y = N, O) compounds and corresponding radical anions in acetonitrile were established by using titration calorimetry combined with relevant electrochemical data through proper thermodynamic cycles.
-
[EN] COMPOUNDS AND METHODS<br/>[FR] COMPOSÉS ET PROCÉDÉS申请人:GLAXOSMITHKLINE LLC公开号:WO2011056739A1公开(公告)日:2011-05-12Disclosed are compounds having formula (I), wherein R1, R2, R3, R4, Z1, Z2, Z3 and Z4 are as defined herein, and methods of making and using the same.揭示了具有化学式(I)的化合物,其中R1、R2、R3、R4、Z1、Z2、Z3和Z4如本文所定义,并且揭示了制备和使用这些化合物的方法。
-
NOVEL COMPOSITIONS OF MATTER AND PHARMACEUTICAL COMPOSITIONS申请人:PSILERA INC.公开号:US20210346347A1公开(公告)日:2021-11-11Pharmaceutical formulations of novel indole compounds and psilocybin analogs are manufactured, provided in novel oral, transdermal, and nasal pharmaceutical compositions for use to treat neurological, mood or abuse diseases and disorders.
-
Sulfonyl amide inhibitors of calcium channel function申请人:Hangeland J. Jon公开号:US20050245535A1公开(公告)日:2005-11-03Compounds of formula I its stereoisomers, solvates, and salts, thereof, wherein: a, b, c, d, f, n, m and Ra are defined herein are are inhibitors of calcium channel function, and are useful in treating calcium channel-dependent disorders, including hypertension.
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
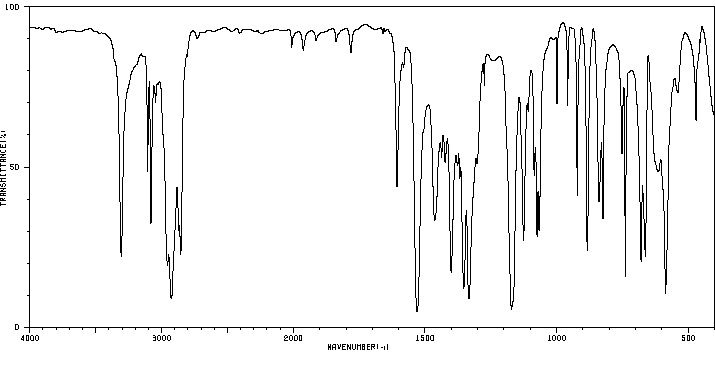
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫