1-BOC-哌啶-3-羧酸 | 163438-09-3
中文名称
1-BOC-哌啶-3-羧酸
中文别名
——
英文名称
BOC-nipecotic acid
英文别名
N-(tert-butyloxycarbonyl)nipecotic acid;1-(tert-butoxycarbonyl)piperidine-3-carboxylic acid;1-(tert-butoxycarbonyl)-3-piperidinecarboxylic acid;(±)-N-Boc-nipecotic acid;1-Boc-piperidine-3-carboxylic acid;1-[(2-methylpropan-2-yl)oxycarbonyl]piperidine-3-carboxylic acid
CAS
163438-09-3;71381-75-4;84358-12-3
化学式
C11H19NO4
mdl
MFCD00673775
分子量
229.276
InChiKey
NXILIHONWRXHFA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:159-162 °C(lit.)
-
沸点:353.2±35.0 °C(Predicted)
-
密度:1.164±0.06 g/cm3(Predicted)
-
稳定性/保质期:
常温常压下稳定,避免与强氧化剂接触。
计算性质
-
辛醇/水分配系数(LogP):1.1
-
重原子数:16
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.818
-
拓扑面积:66.8
-
氢给体数:1
-
氢受体数:4
安全信息
-
危险品标志:Xi
-
安全说明:S26,S36
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:2933399090
-
危险品运输编号:NONH for all modes of transport
-
危险标志:GHS07
-
危险性描述:H315,H319,H335
-
危险性防范说明:P261,P305 + P351 + P338
-
储存条件:常温下应存放在密封容器中,并避免光照。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
(R)-1-Boc-piperidine-3-carboxylic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
(R)-1-Boc-piperidine-3-carboxylic acid
Ingredient name:
CAS number: 163438-09-3
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C11H19NO4
Molecular weight: 229.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
(R)-1-Boc-piperidine-3-carboxylic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
(R)-1-Boc-piperidine-3-carboxylic acid
Ingredient name:
CAS number: 163438-09-3
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C11H19NO4
Molecular weight: 229.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 N-Boc-3-哌啶甲酸乙酯 ethyl N-(t-butyloxycarbonyl)nipecotate 130250-54-3 C13H23NO4 257.33 1-Boc-3-羟甲基哌啶 tert-butyl 3-(hydroxymethyl)piperidine-1-carboxylate 116574-71-1 C11H21NO3 215.293 1-Boc-3-羟甲基吡咯烷 tert-butyl 3-(hydroxymethyl)pyrrolidine-1-carboxylate 114214-69-6 C10H19NO3 201.266 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 N-Boc-3-哌啶甲酸甲酯 1-tert-butyl 3-methyl piperidine-1,3-dicarboxylate 148763-41-1 C12H21NO4 243.303 —— piperidine-1,3-dicarboxylic acid 3-allyl ester 1-tert-butyl ester 1263773-92-7 C14H23NO4 269.341 —— piperidine-1,3-dicarboxylic acid 3-((E)-but-2-enyl)ester 1-tert-butyl ester 1263773-82-5 C15H25NO4 283.368 —— piperidine-1,3-dicarboxylic acid 3-((E)-but-2-enyl) ester 1-tert-butyl ester 1263773-70-1 C15H25NO4 283.368 1-Boc-哌啶-3-甲醛 3-formylpiperidine-1-carboxylic acid tert-butyl ester 118156-93-7 C11H19NO3 213.277 N-Boc-3-甲基-3-哌啶甲酸甲酯 1-tert-butyl 3-methyl 3-methylpiperidine-1,3-dicarboxylate 888952-55-4 C13H23NO4 257.33 1-Boc-3-羟甲基哌啶 tert-butyl 3-(hydroxymethyl)piperidine-1-carboxylate 116574-71-1 C11H21NO3 215.293 —— piperidine-1,3-dicarboxylic acid 1-tert-butyl ester 3-(3,3-difluoro-allyl) ester 1263774-17-9 C14H21F2NO4 305.322 3-(2-丙基)-1,3-哌啶二羧酸-1-(1,1-二甲基乙基)酯 3-allylpiperidine-1,3-dicarboxylic acid 1-tert-butyl ester 959236-11-4 C14H23NO4 269.341 3-乙酰基哌啶-1-羧酸叔丁酯 tert-butyl 3-acetylpiperidine-1-carboxylate 858643-92-2 C12H21NO3 227.304 —— piperidine-1,3-dicarboxylic acid 3-benzyl ester 1-tert-butyl ester 139985-95-8 C18H25NO4 319.401 1-Boc-3-氨基甲酰基哌啶 tert-butyl 3-carbamoylpiperidine-1-carboxylate 91419-49-7 C11H20N2O3 228.291 —— 1-(tert-butyl) 3-methyl 3-allylpiperidine-1,3-dicarboxylate —— C15H25NO4 283.368 —— 3-methylcarbamoyl-piperidine-1-carboxylic acid tert-butyl ester 885698-91-9 C12H22N2O3 242.318 3-(3-甲氧基-3-氧代丙酰基)哌啶-1-羧酸叔丁酯 tert-butyl 3-(3-methoxy-3-oxopropanoyl)piperidine-1-carboxylate 891494-65-8 C14H23NO5 285.34 —— 3-(1-methyl-allyl)-piperidine-1,3-dicarboxylic acid 1-tert-butyl ester 1263773-71-2 C15H25NO4 283.368 —— 1-(t-butoxycarbonyl)nipecotoyl chloride 816455-27-3 C11H18ClNO3 247.722 —— tert-butyl 3-(dimethylcarbamoyl)piperidine-1-carboxylate 889942-54-5 C13H24N2O3 256.345 7-BOC-2-噁-7-氮杂螺[4,5]-1-癸酮 (RS)-1,1-dimethylethyl 2-oxa-1-oxo-7-azaspiro[4,5]decan-7-carboxylate 374795-33-2 C13H21NO4 255.314 —— tert-butyl-3-(ethylaminoformyl)piperidine-1-carboxylate 937724-98-6 C13H24N2O3 256.345 —— tert-butyl 3-(3-ethoxy-3-oxopropanoyl)piperidine-1-carboxylate —— C15H25NO5 299.367 —— tert-butyl 3-(isobutylcarbamoyl)piperidine-1-carboxylate 937725-01-4 C15H28N2O3 284.399 —— tert-butyl 3-(butylcarbamoyl)piperidine1-carboxylate 937725-02-5 C15H28N2O3 284.399 1-Boc-3-甲磺酰基氧甲基哌啶 tert-butyl 3-(((methylsulfonyl)oxy)methyl)piperidine-1-carboxylate 162166-99-6 C12H23NO5S 293.384 —— Tert-butyl 3-(diethylcarbamoyl)piperidine-1-carboxylate 937724-75-9 C15H28N2O3 284.399 —— tert-Butyl 3-(hydroxymethyl)-3-(prop-2-en-1-yl)piperidine-1-carboxylate 441773-93-9 C14H25NO3 255.357 —— Tert-butyl 3-[butyl(methyl)carbamoyl]piperidine-1-carboxylate 937724-95-3 C16H30N2O3 298.426 —— Tert-butyl 3-(propan-2-ylcarbamoyl)piperidine-1-carboxylate 937724-99-7 C14H26N2O3 270.372 —— Tert-butyl 3-(dipropylcarbamoyl)piperidine-1-carboxylate 937724-97-5 C17H32N2O3 312.453 1-Boc-3-(1-哌啶基羰基)哌啶 3-(piperidine-1-carbonyl)piperidine-1-carboxylic acid tert-butyl ester 937724-76-0 C16H28N2O3 296.41 —— succinimidyl 1-(tert-butoxycarbonyl)-3-piperidinecarboxylate 84358-14-5 C15H22N2O6 326.349 —— tert-butyl 3-[(cyclopropylamino)carbonyl]piperidine-1-carboxylate 1015446-12-4 C14H24N2O3 268.356 1-Boc-3-(1-吡咯烷羰基)哌啶 tert-Butyl 3-(pyrrolidine-1-carbonyl)piperidine-1-carboxylate 937724-78-2 C15H26N2O3 282.383 —— Tert-butyl 3-(tert-butylcarbamoyl)piperidine-1-carboxylate 937725-00-3 C15H28N2O3 284.399 3-((乙基氨基)甲基)哌啶-1-羧酸叔丁酯 tert-butyl 3-((ethylamino)methyl)piperidine-1-carboxylate 887587-98-6 C13H26N2O2 242.362 —— 1,1-Dimethylethyl 3-[3-(dimethylamino)-1-oxo-2-propen-1-yl]-1-piperidinecarboxylate 960201-84-7 C15H26N2O3 282.383 —— 1,1-Dimethylethyl 3-[(cyclopentylamino)carbonyl]-1-piperidinecarboxylate 937724-77-1 C16H28N2O3 296.41 3-(吗啉-4-甲酰基)哌啶-1-甲酸叔丁酯 3-(morpholine-4-carbonyl)-piperidine-1-carboxylic acid tert-butyl ester 889942-56-7 C15H26N2O4 298.382 —— tert-butyl 3-(cyclohexylcarbamoyl)piperidine-1-carboxylate 872508-06-0 C17H30N2O3 310.437 1-Boc-3-氰基哌啶 tert-butyl 3-cyanopiperidine-1-carboxylate 91419-53-3 C11H18N2O2 210.276 2-甲基-2-丙基3-(叠氮基甲基)-1-哌啶羧酸酯 tert-butyl 3-(azidomethyl)piperidine-1-carboxylate 155541-67-6 C11H20N4O2 240.305 —— tert-butyl 3-(difluoromethyl)piperidine-1-carboxylate —— C11H19F2NO2 235.274 —— tert-butyl 3-(phenethylcarbamoyl)piperidine-1-carboxylate —— C19H28N2O3 332.443 —— 1-N-Boc-3-carbamimidoyl-piperidine 1220039-60-0 C11H21N3O2 227.307 1-Boc-3-[甲氧基(甲基)氨基甲酰]哌嗪 tert-butyl 3-(methoxy(methyl)carbamoyl)piperidine-1-carboxylate 189442-78-2 C13H24N2O4 272.345 —— 1-Boc-piperidine-3-carboxylic acid phenyl amide 309747-36-2 C17H24N2O3 304.389 —— tert-butyl 3-((4-chlorophenyl)carbamoyl)piperidine-1-carboxylate —— C17H23ClN2O3 338.834 —— 1,1-dimethylethyl 3-benzoyl-1-piperidinecarboxylate 884510-91-2 C17H23NO3 289.375 3-(N-羟基甲脒基)哌啶-1-羧酸叔丁酯 tert-butyl 3-[(hydroxyamino)(imino)methyl]piperidine-1-carboxylate 479080-28-9 C11H21N3O3 243.306 —— 3-(1,1-difluoro-allyl)-piperidine-1,3-dicarboxylic acid 1-tert-butyl ester 1263774-18-0 C14H21F2NO4 305.322 —— 3-(2-acetylphenyl)-1-tert-butyl piperidine-1,3-dicarboxylate 868284-37-1 C19H25NO5 347.411 —— tert-Butyl 3-((pyridin-3-ylmethyl)carbamoyl)piperidine-1-carboxylate 204574-56-1 C17H25N3O3 319.404 - 1
- 2
- 3
- 4
- 5
- 6
反应信息
-
作为反应物:描述:1-BOC-哌啶-3-羧酸 在 palladium on activated charcoal lithium aluminium tetrahydride 、 ammonium acetate 、 benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate 、 氢气 、 三乙胺 作用下, 以 四氢呋喃 、 二氯甲烷 、 乙酸乙酯 、 甲苯 为溶剂, -15.0~150.0 ℃ 、303.98 kPa 条件下, 反应 38.5h, 生成 tert-butyl 3-[2-amino-3-cyano-6-(2-hydroxyphenyl)-4-pyridinyl]-1-piperidinecarboxylate参考文献:名称:新型IKK-β抑制剂的合成与构效关系。第2部分:提高体外活性。摘要:合成了一系列的2-氨基-3-氰基-4-烷基-6-(2-羟基苯基)吡啶衍生物,并将其评估为IkappaB激酶β(IKK-β)抑制剂。氨基吡啶基团取代核心吡啶环上4位的芳香族基团导致激酶酶和细胞效能的显着增加,并提供了具有低于100 nM的IC(50)值的强效IKK-β抑制剂。DOI:10.1016/j.bmcl.2004.05.040
-
作为产物:描述:参考文献:名称:新型杂环螺3-(N-甲基-N-苯基氨基)-2 H-叠氮基的合成及其作为氨基酸等价物在模型三肽制备中的应用摘要:描述了基于酰胺烯酸酯和二氯代氯代磷酸酯之间反应的新型杂环基3-氨基-2H-叠氮基的合成。已经表明该化合物是有用的氨基酸当量,并且以良好的总产率实现了模型三肽的合成。DOI:10.1016/s0040-4020(01)87199-4
文献信息
-
크로만 유도체 및 이를 유효성분으로 함유하는 신생혈관성 안질환 또는 암의 예방 또는 치료용 약학적 조성물申请人:Gachon University of Industry-Academic cooperation Foundation 가천대학교 산학협력단(220040376324) BRN ▼129-82-07687公开号:KR20200124630A公开(公告)日:2020-11-03본 발명은 크로만 유도체인 하기 화학식 1의 화합물, 이의 수화물, 이의 입체 이성질체, 또는 이의 약학적으로 허용 가능한 염을 포함하는 암 또는 신생혈관과 관련된 질환 치료용 약학적 조성물을 제공한다. [화학식 1] (상기 화학식 1에서 R, R, R, R, R, R, R은 발명의 설명에서 정의한 바와 같다.)
-
MACROCYCLIC COMPOUNDS AND THEIR USE AS KINASE INHIBITORS申请人:Combs Andrew Paul公开号:US20090286778A1公开(公告)日:2009-11-19The present invention relates to macrocyclic compounds of Formula I: or pharmaceutically acceptable salts thereof or quaternary ammonium salts thereof wherein constituent members are provided hereinwith, as well as their compositions and methods of use, which are JAK/ALK inhibitors useful in the treatment of JAK/ALK-associated diseases including, for example, inflammatory and autoimmune disorders, as well as cancer.
-
Carbonic anhydrase activators: amino acyl/dipeptidyl histamine derivatives bind with high affinity to isozymes I, II and IV and act as efficient activators作者:Claudiu T. Supuran、Andrea ScozzafavaDOI:10.1016/s0968-0896(99)00227-8日期:1999.12a series of compounds with the general formula AA-Hst (AA = amino acyl; dipeptidyl). The new derivatives were assayed as activators of three carbonic anhydrase (CA) isozymes, hCA I, hCA II (cytosolic forms) and bCA IV (membrane-bound form). Efficient activation was observed against all three isozymes, but especially against hCA I and bCA IV, with affinities in the nanomolar range for the best compounds组胺(Hst)与四溴邻苯二甲酸酐反应,并用三苯甲基磺酰氯保护其咪唑部分,然后进行肼解反应,得到N-1-三苯甲基磺酰-组胺,这是一种关键中间体,在其氨基乙基部分被进一步衍生。在咪唑类和氨基部分脱保护后,在碳二亚胺存在下,关键中间体与N-Boc-氨基酸/二肽(Boc-AA)反应,得到了一系列通式为AA-Hst的化合物(AA =氨基酰基;二肽基)。分析了新衍生物作为三种碳酸酐酶(CA)同工酶的激活剂,它们分别是hCA I,hCA II(胞质形式)和bCA IV(膜结合形式)。观察到针对所有三种同工酶的有效活化,尤其是针对hCA I和bCA IV的活化,其最佳化合物的亲和力在纳摩尔范围内。另一方面,hCA II可以被约10-20 nM的亲和力激活。这类新型的CA激活剂可能导致针对CA缺乏综合症(一种骨骼,脑和肾脏的遗传性疾病)的药物/诊断剂的开发。
-
Generation and Cross-Coupling of Organozinc Reagents in Flow作者:Ananda Herath、Valentina Molteni、Shifeng Pan、Jon LorenDOI:10.1021/acs.orglett.8b03156日期:2018.12.7formation of organozinc reagents and subsequent cross-coupling with aryl halides and activated carboxylic acids is reported. Formation of organozinc reagents is achieved by pumping organic halides, in the presence of ZnCl2 and LiCl, through an activated Mg-packed column under flow conditions. This method provides efficient in situ formation of aryl, primary, secondary, and tertiary alkyl organozinc reagents
-
Visible-Light-Mediated Alkylation of Thiophenols via Electron Donor–Acceptor Complexes Formed between Two Reactants作者:Yi-Ping Cai、Fang-Yuan Nie、Qin-Hua SongDOI:10.1021/acs.joc.1c01433日期:2021.9.3A metal-free, photocatalyst-free, photochemical system was developed for the direct alkylation of thiophenols via electron donor–acceptor (EDA) complexes (KEDA = 145 M–1) between two reactants, N-hydroxyphthalimide esters as acceptors and thiophenol anions as donors, in the presence of a tertiary amine. The EDA complexes in the reaction system have a broad range of visible-light absorption (400–650
表征谱图
-
氢谱1HNMR
-
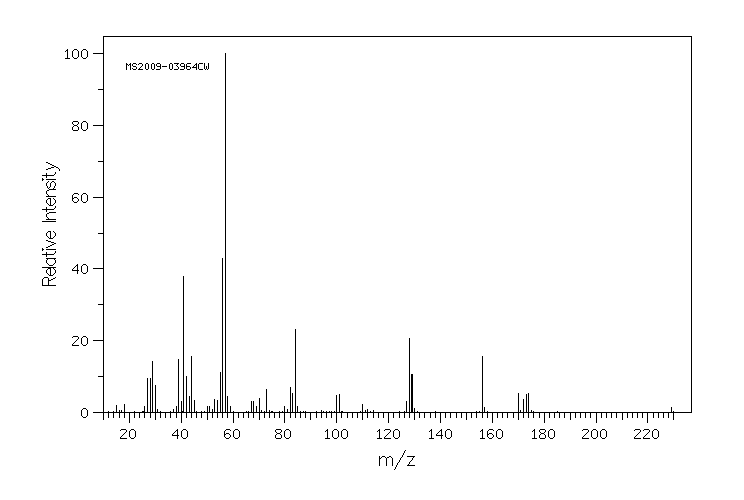
质谱MS
-
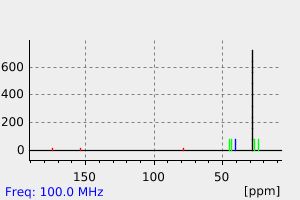
碳谱13CNMR
-
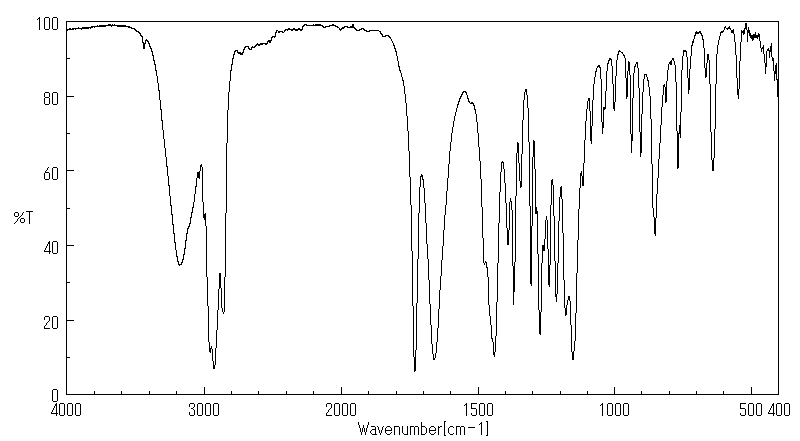
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(R)-3-甲基哌啶盐酸盐;
(R)-2-苄基哌啶-1-羧酸叔丁酯
((3S,4R)-3-氨基-4-羟基哌啶-1-基)(2-(1-(环丙基甲基)-1H-吲哚-2-基)-7-甲氧基-1-甲基-1H-苯并[d]咪唑-5-基)甲酮盐酸盐
高氯酸哌啶
高托品酮肟
马来酸帕罗西汀
颜料红48:4
顺式3-氟哌啶-4-醇盐酸盐
顺式2,6-二甲基哌啶-4-酮
顺式1-苄基-4-甲基-3-甲氨基-哌啶
顺式-叔丁基4-羟基-3-甲基哌啶-1-羧酸酯
顺式-6-甲基-哌啶-1,3-二甲酸1-叔丁酯
顺式-5-(三氟甲基)哌啶-3-羧酸甲酯盐酸盐
顺式-4-叔丁基-2-甲基哌啶
顺式-4-Boc-氨基哌啶-3-甲酸甲酯
顺式-4-(氮杂环丁烷-1-基)-3-氟哌
顺式-3-顺式-4-氨基哌啶
顺式-3-甲氧基-4-氨基哌啶
顺式-3-BOC-3,7-二氮杂双环[4.2.0]辛烷
顺式-3-(1-吡咯烷基)环丁腈
顺式-3,5-哌啶二羧酸
顺式-3,4-二溴-3-甲基吡咯烷盐酸盐
顺式-2,6-二甲基-4-氧代哌啶-1-羧酸叔丁基酯
顺式-1-叔丁氧羰基-4-甲基氨基-3-羟基哌啶
顺式-1-boc-3,4-二氨基哌啶
顺式-1-(4-叔丁基环己基)-4-苯基-4-哌啶腈
顺式-1,3-二甲基-4-乙炔基-6-苯基-3,4-哌啶二醇
顺-4-(4-氟苯基)-1-(4-异丙基环己基)-4-哌啶羧酸
顺-4-(2-氟苯基)-1-(4-异丙基环己基)-4-哌啶羧酸
顺-3-氨基-4-氟哌啶-1-羧酸叔丁酯
顺-1-苄基-4-甲基哌啶-3-氨基酸甲酯盐酸盐
非莫西汀
雷芬那辛
雷拉地尔
阿维巴坦中间体4
阿格列汀杂质
阿尼利定盐酸盐 CII
阿尼利定
阿塔匹酮
阿哌沙班杂质BMS-591455
阿哌沙班杂质87
阿哌沙班杂质52
阿哌沙班杂质51
阿哌沙班杂质5
阿哌沙班杂质
阿哌沙班杂质
阿哌沙班-d3
阿哌沙班
阻聚剂701
间氨基谷氨酰胺