2,4'-二氟二苯甲酮 | 342-25-6
中文名称
2,4'-二氟二苯甲酮
中文别名
2,4'-二氟苯甲酮;2,4-二氟二苯甲酮;2,4"-二氟二苯甲酮;粉唑醇二苯甲酮;2,4'-二氟二苯酮
英文名称
2,4'-difluorobenzophenone
英文别名
(2-fluorophenyl)-(4-fluorophenyl)methanone
CAS
342-25-6
化学式
C13H8F2O
mdl
——
分子量
218.203
InChiKey
LKFIWRPOVFNPKR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:22-24 °C(lit.)
-
沸点:176-178 °C16 mm Hg(lit.)
-
密度:1.244 g/mL at 25 °C(lit.)
-
闪点:>230 °F
-
溶解度:可溶于氯仿、甲醇
计算性质
-
辛醇/水分配系数(LogP):3.5
-
重原子数:16
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:3
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S24/25,S26,S37/39
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:2914700090
-
危险性防范说明:P305+P351+P338
-
危险性描述:H315,H319
-
储存条件:室温
SDS
| Name: | 2 4 -Difluorobenzophenone 98% Material Safety Data Sheet |
| Synonym: | None known |
| CAS: | 342-25-6 |
Synonym:None known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 342-25-6 | 2,4'-Difluorobenzophenone | 98 | 206-441-7 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. The toxicological properties of this substance have not been fully investigated.
Inhalation:
Causes respiratory tract irritation. The toxicological properties of this substance have not been fully investigated. Can produce delayed pulmonary edema.
Chronic:
Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid. Do NOT use mouth-to-mouth resuscitation.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Use water spray to keep fire-exposed containers cool. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Containers may explode when heated.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Cool containers with flooding quantities of water until well after fire is out. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Avoid runoff into storm sewers and ditches which lead to waterways.
Clean up spills immediately, observing precautions in the Protective Equipment section. Remove all sources of ignition. Absorb spill using an absorbent, non-combustible material such as earth, sand, or vermiculite. Do not use combustible materials such as sawdust.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Keep away from heat, sparks and flame. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 342-25-6: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear light yellow
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 176 - 178 deg C @ 16.00mm Hg
Freezing/Melting Point: 22.00 - 24.00 deg C
Autoignition Temperature: Not available.
Flash Point: > 112 deg C (> 233.60 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.2440g/cm3
Molecular Formula: C13H8F2O
Molecular Weight: 218.20
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen fluoride gas.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 342-25-6 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2,4'-Difluorobenzophenone - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 342-25-6: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 342-25-6 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 342-25-6 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— [4-(dimethylamino)phenyl](2-fluorophenyl)methanone 446018-27-5 C15H14FNO 243.281 —— 2,4'-difluorodiphenylmethane 132287-51-5 C13H10F2 204.219 —— (2-fluorophenyl)(4-fluorophenyl)methanol 153877-53-3 C13H10F2O 220.219 —— (R)-2-fluorophenyl-4'-fluorophenylmethanol —— C13H10F2O 220.219 —— (S)-(2-fluorophenyl)(4-fluorophenyl)methanol —— C13H10F2O 220.219 1-氟-2-[1-(4-氟苯基)乙烯基]苯 1-fluoro-2-(1-(4-fluorophenyl)vinyl)benzene 85000-56-2 C14H10F2 216.23
反应信息
-
作为反应物:描述:2,4'-二氟二苯甲酮 在 dirhodium tetraacetate 、 potassium carbonate 、 Selectfluor 、 三氟乙酸 、 三氟乙酸酐 作用下, 以 丙酮 为溶剂, 反应 12.0h, 生成 3-氟-占吨酮参考文献:名称:拓宽区域和化学选择性C–H氧合的催化剂和反应范围:通过吸引Rh(ii)和Ru(ii)催化,一种方便且可扩展的2-酰基苯酚方法†摘要:已开发出一种独特的Rh(II)和Ru(II)催化的芳基酮和其他芳烃的CH氧化反应,可以轻松合成各种功能化的酚。该反应显示出优异的反应性,区域和化学选择性,良好的官能团相容性和高产率。该方法的实用性已通过克级合成几种不同的2-酰基酚得到了证明。它的效用已在杂环合成和药物的直接修饰中的进一步应用中得到很好的例证非诺贝特。DOI:10.1039/c3ob27457h
-
作为产物:描述:2-氟甲苯 在 aluminum (III) chloride 、 iron(III) chloride 、 水 、 氯 、 三乙胺 、 lithium chloride 作用下, 反应 5.0h, 生成 2,4'-二氟二苯甲酮参考文献:名称:一种二氟二苯甲酮生产制造方法摘要:本发明属于化学合成技术领域,特别涉及一种二氟二苯甲酮生产制造方法。本发明以邻氟甲苯为原料,经光氯化、催化水解后与氟苯混合,在无水三氯化铝和氯化锂混合催化剂催化下得到高纯度2,4/‑二氟二苯甲酮,所用原料廉价易得,合成路线简单,易于操作,有利于工业化生产。运用氟苯做溶剂,氟苯可重复利用;且洗水循环用于淬灭反应,淬灭反应生成的三氯化铝溶液可用于净水剂生产,整个反应过程中不产生任何废水,环境友好,节约资源。公开号:CN113956141A
文献信息
-
Photoinduced Hydroarylation and Cyclization of Alkenes with Luminescent Platinum(II) Complexes作者:Hanchao Cheng、Tsz‐Lung Lam、Yungen Liu、Zhou Tang、Chi‐Ming CheDOI:10.1002/anie.202011841日期:2021.1.18Photoinduced hydroarylation of alkenes is an appealing synthetic strategy for arene functionalization. Herein, we demonstrated that aryl radicals generated from electron‐deficient aryl chlorides/bromides could be trapped by an array of terminal/internal aryl alkenes in the presence of [Pt(O^N^C^N)] under visible‐light (410 nm) irradiation, affording anti‐Markovnikov hydroarylated compounds in up to
-
Catalytic Enantioselective Borohydride Reduction of <i>Ortho</i>-Fluorinated Benzophenones作者:Ai Kokura、Saiko Tanaka、Taketo Ikeno、Tohru YamadaDOI:10.1021/ol060927p日期:2006.7.1[reaction: see text] In the presence of the optically active ketoiminatocobalt(II) complexes, the enantioselective borohydride reduction of benzophenones was successfully completed. The fluorine atom on the ortho position of the benzophenone and aryl ketones proved effective for obtaining high enantioselectivities. The combined use of modified lithium borohydride afforded the corresponding benzhydrols
-
Phthalide synthesis through dehydrogenated lactonization of the C(sp<sup>3</sup>)–H bond by photoredox catalysis作者:Hui Liu、Chao Liu、Shanyi Chen、Qihong Lai、Yulin Lin、Zhixiong Cai、Mingqiang Huang、Shunyou CaiDOI:10.1039/d1gc02297k日期:——A practical and efficient method is established for the direct oxidative lactonization of the C(sp3)–H bonds relying on visible-light-induced photoredox catalysis. This protocol expediently allows the delivery of diverse phthalides using oxygen as the sole terminal oxidant under metal-free conditions at room temperature. Notably, the choice of an appropriate hydrogen atom transfer (HAT) cocatalyst
-
Highly Efficient Hydrophosphonylation of Aldehydes and Unactivated Ketones Catalyzed by Methylene-Linked Pyrrolyl Rare Earth Metal Amido Complexes作者:Shuangliu Zhou、Zhangshuan Wu、Jiewei Rong、Shaowu Wang、Gaosheng Yang、Xiancui Zhu、Lijun ZhangDOI:10.1002/chem.201102207日期:2012.2.27characterized by 1H NMR spectroscopic analyses. The structures of complexes 2, 3, 4, and 6 were determined by single‐crystal X‐ray analyses. Study of the catalytic activities of the complexes showed that these rare earth metal amido complexes were excellent catalysts for hydrophosphonylations of aldehydes and unactivated ketones. The catalyzed reactions between diethyl phosphite and aldehydes in the presence通过甲硅烷基胺消除反应制备了一系列带有亚甲基连接的吡咯基-酰胺基配体的稀土金属酰胺配合物,在无溶剂条件下,液态底物在醛和未活化酮的氢膦酰化反应中显示出很高的催化活性。的治疗[(ME 3 Si)的2 N] 3 LN(μ-Cl)的锂(THF)3与2-(2,6--ME 2 ç 6 ħ 3 NHCH 2)C 4 H ^ 3 NH(1,1个当量)在甲苯,得到相应的式三价稀土金属酰胺(μ-η 5:η 1):η 1-2-[(2,6-Me 2 C 6 H 3)NCH 2 ](C 4 H 3 N)LnN(SiMe 3)2 } 2 [Ln = Y(2),Nd(3),Sm(4),Dy(5),Yb(6)],产量适中。所有化合物均通过光谱法和元素分析充分表征。钇配合物的特征还在于1 H NMR光谱分析。复合物的结构 2,3,4,和6由单晶X射线分析确定。配合物催化活性的研究表明,这些稀土金属酰胺配合物是醛和未活化
-
Pd-Catalyzed CH Oxygenation with TFA/TFAA: Expedient Access to Oxygen-Containing Heterocycles and Late-Stage Drug Modification作者:Gang Shan、Xinglin Yang、Linlin Ma、Yu RaoDOI:10.1002/anie.201207458日期:2012.12.21Acid brings the oxygen: A general method for palladium‐catalyzed CH oxygenation has been developed for the facile synthesis of a wide range of functionalized phenols with readily available aryl ketones, benzoates, benzamides, acetanilides, and sulfonamides. A trifluoroacetic acid/trifluoroacetic acid anhydride solvent system serves as the oxygen source and is the critical factor for CH activation
表征谱图
-
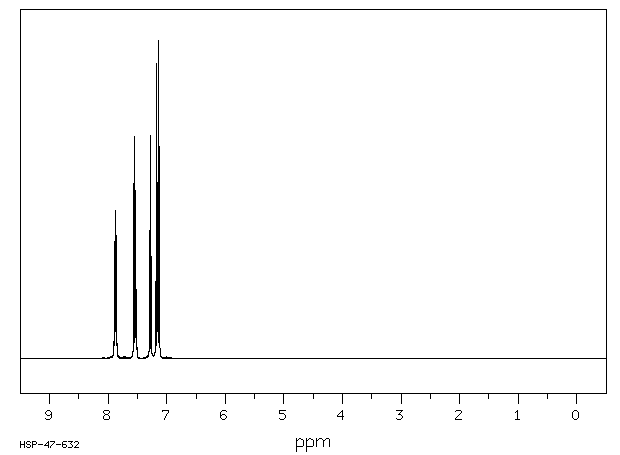
氢谱1HNMR
-
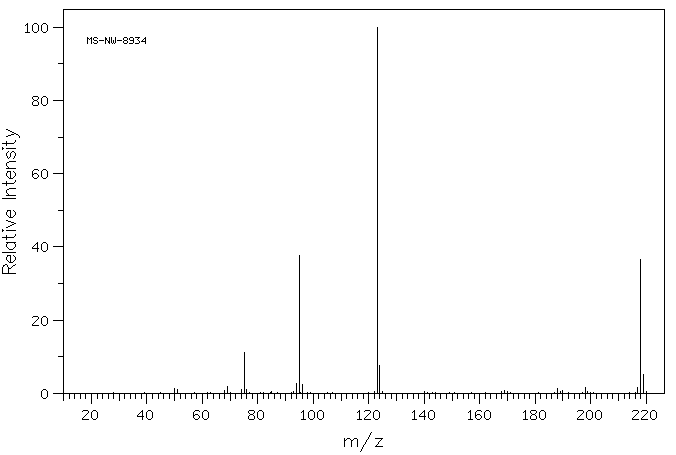
质谱MS
-
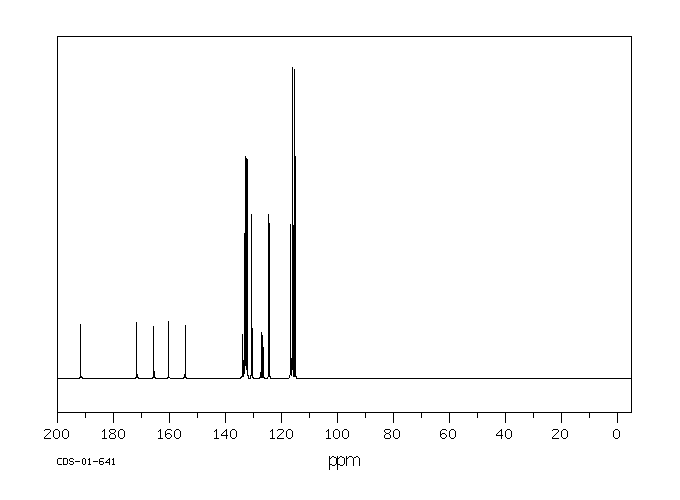
碳谱13CNMR
-
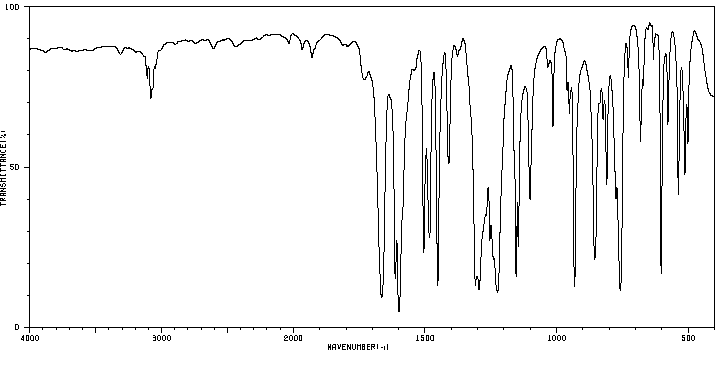
红外IR
-
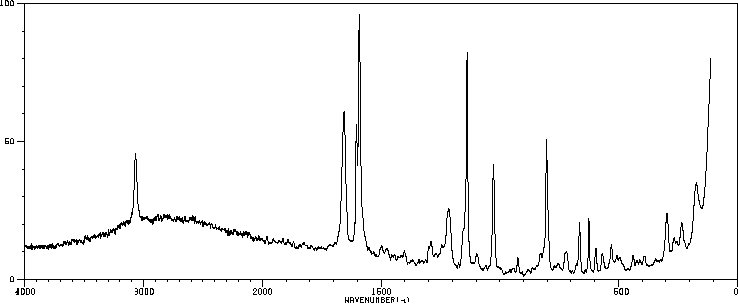
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫