(4E,6E)-1-(4-hydroxyphenyl)-7-phenylhepta-4,6-dien-3-one | 562810-28-0
中文名称
——
中文别名
——
英文名称
(4E,6E)-1-(4-hydroxyphenyl)-7-phenylhepta-4,6-dien-3-one
英文别名
1-(4-hydroxyphenyl)-7-phenyl-(4E,6E)-4,6-heptadien-3-one
CAS
562810-28-0
化学式
C19H18O2
mdl
——
分子量
278.351
InChiKey
GIXGHUAQPPUMRA-KBXRYBNXSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:129.3-130.0 °C(Solv: ethyl ether (60-29-7); hexane (110-54-3))
-
沸点:498.7±38.0 °C(Predicted)
-
密度:1.130±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):4.2
-
重原子数:21
-
可旋转键数:6
-
环数:2.0
-
sp3杂化的碳原子比例:0.11
-
拓扑面积:37.3
-
氢给体数:1
-
氢受体数:2
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— (4E,6E)-1-(4-benzoyloxyphenyl)-7-phenylhepta-4,6-dien-3-one 562810-31-5 C26H22O3 382.459 覆盆子酮 4-(4-hydroxyphenyl)-2-butanone 5471-51-2 C10H12O2 164.204 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— (4E,6E)-1-(4-methoxyphenyl)-7-phenylhepta-4,6-dien-3-one 1062082-92-1 C20H20O2 292.378 —— [4-[(4E,6E)-3-oxo-7-phenylhepta-4,6-dienyl]phenyl] acetate 1062083-04-8 C21H20O3 320.388
反应信息
-
作为反应物:描述:(4E,6E)-1-(4-hydroxyphenyl)-7-phenylhepta-4,6-dien-3-one 、 碘甲烷 在 吡啶 、 silver(l) oxide 作用下, 以 N,N-二甲基甲酰胺 为溶剂, 反应 2.0h, 以82%的产率得到(4E,6E)-1-(4-methoxyphenyl)-7-phenylhepta-4,6-dien-3-one参考文献:名称:Diarylheptanoids, new phytoestrogens from the rhizomes of Curcuma comosa: Isolation, chemical modification and estrogenic activity evaluation摘要:Three new diarylheptanoids, a 1: 2 mixture of (3S)- and (3R)-1-(4-methoxyphenyl)-7-phenyl-(6E)-6-hepten-3-ol (13a and 13b) and 1-(4-hydroxyphenyl)-7-phenyl-( 6E)-6-hepten-3- one ( 15), together with two synthetically known diarylheptanoids 1,7-diphenyl-(1E,3E,5E)-1,3,5-triene(9) and 1-(4-hydroxyphenyl)7-phenyl-(4E,6E)-4,6-heptadien-3- one ( 16), and nine known diarylheptanoids, 2, 8, 10 - 12, 14, a 3: 1 mixture of 17a and 17b, and 18, were isolated from the rhizomes of Curcuma comosa Roxb. The absolute stereochemistry of the isolated compounds has also been determined using the modified Mosher's method. The isolated compounds and the chemically modified analogues were evaluated for their estrogenic-like transcriptional activity using RT-PCR in HeLa cell line. Some of the isolated diarylheptanoids and their modified analogues exhibited estrogenic activity comparable to or higher than that of the phytoestrogen genistein. Based on the transcriptional activation of both estrogenic targets, Bcl-xL and ER beta gene expression, the structural features for a diarylheptanoid to exhibit high estrogenic activity are the presence of an phenolic function conjugated with the aromatic ring at the 7-position, a keto group at the 3-position, and a phenolic hydroxyl group at the p-position of the aromatic ring attached to the 1- position of the heptyl chain. (c) 2008 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmc.2008.05.051
-
作为产物:描述:4-(3-氧代丁基)苯基苯甲酸酯 在 4 A molecular sieve 、 potassium carbonate 、 对甲苯磺酸 、 lithium diisopropyl amide 作用下, 以 四氢呋喃 、 甲醇 、 苯 为溶剂, 反应 1.25h, 生成 (4E,6E)-1-(4-hydroxyphenyl)-7-phenylhepta-4,6-dien-3-one参考文献:名称:通过三种不同的策略合成新的生物合成重要的二芳基庚烷及其氧杂和氟类似物摘要:摘要 Wittig、aldol 和 Wittig-Horner 反应已被用于合成新的二芳基庚烷类化合物,它们是植物中苯菲酮生物合成的假定中间体。与其他方法相比,Wittig-Horner 方法是最合适的,并且产生了显着更高的产量。DOI:10.1081/scc-120016367
表征谱图
-
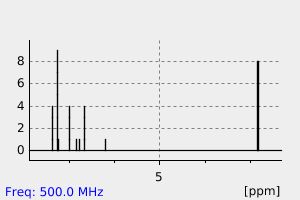
氢谱1HNMR
-
质谱MS
-
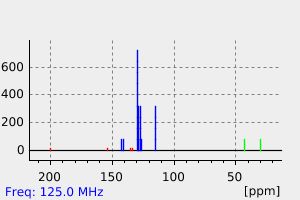
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(11aR)-3,7-双(3,5-二甲基苯基)-10,11,12,13-四氢-5-羟基-5-氧化物-二茚基[7,1-de:1'',7''-fg][1,3,2]二氧杂膦酸
龙血素C
顺-1,7-二苯基-1-庚烯基-5-醇
那洛西芬
赤杨酮
赤杨二醇
血竭素
蒙桑酮C
萘-2,7-二磺基酸,钠盐
苯酚,4-(1,3-二苯基丁基)-2-(1-苯基乙基)-
苯甲酸,2-[[2-[(2-羧基苯基)氨基]-5-(三氟甲基)苯基]氨基]-5-[[[(4-羟基-3-甲氧苯基)甲基]氨基]甲基]-
苯基-[4-(2-苯基乙炔基)苯基]甲酮
苯基-[2-[3-(三氟甲基)苯基]苯基]甲酮
苯基-[2-(2-苯基苯基)苯基]甲酮
苯基-(3-苯基萘-2-基)甲酮
苯基-(2-苯基环己基)甲酮
苯,[(二甲基苯基)甲基]甲基[(甲基苯基)甲基]-
苯,1,3-二[1-甲基-1-[4-(4-硝基苯氧基)苯基]乙基]-
脱甲氧姜黄
紫外吸收剂 234
粗糠柴苦素
硫酸姜黄素
矮紫玉盘素
益智醇
白桦林烯酮;1,7-双(4-羟基苯基)-4-庚烯-3-酮
甲酮,苯基(1,6,7,8-四氢-1-甲基-5-苯基环戊二烯并[g]吲哚-3-基)-
甲酮,[3-(4-甲氧苯基)-1-苯基-9H-芴-4-基]苯基-
甲酮,(4-氯苯基)[1-(4-氯苯基)-3-苯基-9H-芴-4-基]-
环香草酮
溴敌隆
波森
桤木酮
桑根酮D
杨梅醇
杨梅酮
杨梅联苯环庚醇-15-葡糖苷
替拉那韦
替吡法尼(S型对映体)
替吡法尼
曲沃昔芬
姜黄素葡糖苷酸
姜黄素beta-D-葡糖苷酸
姜黄素4,4'-二乙酸酯
姜黄素-d6
姜黄素
姜烯酮 A
奈帕芬胺杂质D
四甲基姜黄素
四氢脱甲氧基二阿魏酰甲烷
四氢姜黄素二乙酸酯