cis-1,3-cyclohexanediol | 823-18-7
中文名称
——
中文别名
——
英文名称
cis-1,3-cyclohexanediol
英文别名
(1S,3R)-cyclohexane-1,3-diol
CAS
823-18-7
化学式
C6H12O2
mdl
——
分子量
116.16
InChiKey
RLMGYIOTPQVQJR-OLQVQODUSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:81.0 to 85.0 °C
-
沸点:125°C/5mmHg(lit.)
-
密度:0.9958 (rough estimate)
计算性质
-
辛醇/水分配系数(LogP):0.2
-
重原子数:8
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:40.5
-
氢给体数:2
-
氢受体数:2
安全信息
-
储存条件:室温且干燥环境下使用。
SDS
Section I.Chemical Product and Company Identification
Chemical Name cis-1,3-Cyclohexanediol
Portland OR
Synonym 1,3-Cyclohexanediol, (1R,3S)-rel- (CA INDEX NAME);
cis-1,3-Dihydroxycyclohexane
Chemical Formula C6H12O2
CAS Number 823-18-7
Section II. Composition and Information on Ingredients
Toxicology Data
Chemical Name CAS Number Percent (%) TLV/PEL
cis-1,3-Cyclohexanediol 823-18-7 Min. 98.0 (GC) Not available. Not available.
Section III. Hazards Identification
Acute Health Effects No specific information is available in our data base regarding the toxic effects of this material for humans. However,
exposure to any chemical should be kept to a minimum. Skin and eye contact may result in irritation. May be harmful if
inhaled or ingested. Always follow safe industrial hygiene practices and wear proper protective equipment when handling
this compound.
Chronic Health Effects CARCINOGENIC EFFECTS : Not available.
MUTAGENIC EFFECTS : Not available.
TERATOGENIC EFFECTS : Not available.
DEVELOPMENTAL TOXICITY: Not available.
Repeated or prolonged exposure to this compound is not known to aggravate existing medical conditions.
Section IV. First Aid Measures
Eye Contact Check for and remove any contact lenses. In case of contact, immediately flush eyes with plenty of water for at least 15
minutes. Get medical attention.
Skin Contact In case of contact, immediately flush skin with plenty of water. Remove contaminated clothing and shoes. Wash clothing
before reuse. Thoroughly clean shoes before reuse. Get medical attention.
If the victim is not breathing, perform mouth-to-mouth resuscitation. Loosen tight clothing such as a collar, tie, belt or
Inhalation
waistband. If breathing is difficult, oxygen can be administered. Seek medical attention if respiration problems do not
improve.
INDUCE VOMITING by sticking finger in throat. Lower the head so that the vomit will not reenter the mouth and throat.
Ingestion
Loosen tight clothing such as a collar, tie, belt or waistband. If the victim is not breathing, perform mouth-to-mouth
resuscitation. Examine the lips and mouth to ascertain whether the tissues are damaged, a possible indication that the toxic
material was ingested; the absence of such signs, however, is not conclusive.
Section V. Fire and Explosion Data
Not available.
May be combustible at high temperature. Auto-Ignition
Flammability
Flash Points Flammable Limits Not available.
Not available.
These products are toxic carbon oxides (CO, CO2).
Combustion Products
Fire Hazards
Not available.
Risks of explosion of the product in presence of mechanical impact: Not available.
Explosion Hazards
Risks of explosion of the product in presence of static discharge: Not available.
Fire Fighting Media
SMALL FIRE: Use DRY chemical powder.
LARGE FIRE: Use water spray, fog or foam. DO NOT use water jet.
and Instructions
Consult with local fire authorities before attempting large scale fire-fighting operations.
Continued on Next Page
cis-1,3-Cyclohexanediol
Section VI. Accidental Release Measures
Spill Cleanup Use a shovel to put the material into a convenient waste disposal container. Finish cleaning the spill by rinsing any
contaminated surfaces with copious amounts of water. Consult federal, state, and/or local authorities for assistance on
Instructions
disposal.
Section VII. Handling and Storage
Handling and Storage Keep away from heat. Mechanical exhaust required. When not in use, tightly seal the container and store in a dry, cool
place. Avoid excessive heat and light. Do not breathe dust.
Information
Always store away from incompatible compounds such as oxidizing agents.
Section VIII. Exposure Controls/Personal Protection
Use process enclosures, local exhaust ventilation, or other engineering controls to keep airborne levels below recommended
Engineering Controls
exposure limits. If user operations generate dust, fume or mist, use ventilation to keep exposure to airborne contaminants
below the exposure limit.
Splash goggles. Lab coat. Dust respirator. Boots. Gloves. Suggested protective clothing might not be sufficient; consult a
Personal Protection
specialist BEFORE handling this product. Be sure to use a MSHA/NIOSH approved respirator or equivalent.
Exposure Limits Not available.
Section IX. Physical and Chemical Properties
Solid. Solubility
Physical state @ 20°C Not available.
Not available.
Specific Gravity
116.16
Molecular Weight Partition Coefficient Not available.
Boiling Point 125°C (257°F) @ 5 mmHg Vapor Pressure Not applicable.
83°C (181.4°F) Not available.
Melting Point Vapor Density
Not available. Volatility Not available.
Refractive Index
Not available.
Critical Temperature Not available. Odor
Viscosity Not available. Taste Not available.
Section X. Stability and Reactivity Data
Stability
This material is stable if stored under proper conditions. (See Section VII for instructions)
Conditions of Instability Avoid excessive heat and light.
Incompatibilities
Reactive with oxidizing agents.
Section XI. Toxicological Information
Not available.
RTECS Number
Routes of Exposure Eye Contact. Ingestion. Inhalation.
Not available.
Toxicity Data
Chronic Toxic Effects CARCINOGENIC EFFECTS : Not available.
MUTAGENIC EFFECTS : Not available.
TERATOGENIC EFFECTS : Not available.
DEVELOPMENTAL TOXICITY: Not available.
Repeated or prolonged exposure to this compound is not known to aggravate existing medical conditions.
Acute Toxic Effects No specific information is available in our data base regarding the toxic effects of this material for humans. However,
exposure to any chemical should be kept to a minimum. Skin and eye contact may result in irritation. May be harmful if
inhaled or ingested. Always follow safe industrial hygiene practices and wear proper protective equipment when handling this
compound.
Section XII. Ecological Information
Not available.
Ecotoxicity
Not available.
Environmental Fate
Continued on Next Page
cis-1,3-Cyclohexanediol
Section XIII. Disposal Considerations
Waste Disposal Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material with a
combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system. Observe all
federal, state and local regulations when disposing of the substance.
Section XIV. Transport Information
Not a DOT controlled material (United States).
DOT Classification
PIN Number Not applicable.
Proper Shipping Name Not applicable.
Packing Group (PG) Not applicable.
DOT Pictograms
Section XV. Other Regulatory Information and Pictograms
TSCA Chemical Inventory This product is NOT on the EPA Toxic Substances Control Act (TSCA) inventory. The following notices are required by 40
CFR 720.36 (C) for those products not on the inventory list:
(EPA)
(i) These products are supplied solely for use in research and development by or under the supervision of a technically
qualified individual as defined in 40 CFR 720.0 et sec.
(ii) The health risks of these products have not been fully determined. Any information that is or becomes available will be
supplied on an MSDS sheet.
WHMIS Classification Not controlled under WHMIS (Canada).
(Canada)
EINECS Number (EEC) Not available.
EEC Risk Statements Not available.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
反应信息
-
作为反应物:描述:cis-1,3-cyclohexanediol 在 sodium dichromate 、 乙醚 、 硫酸 作用下, 生成 2-cyclohexenone 2,4-dinitrophenylhydrazone参考文献:名称:339.间苯二酚。第一部分摘要:DOI:10.1039/jr9490001586
-
作为产物:描述:参考文献:名称:烯丙醇和高烯丙醇分子内氢化硅烷化的立体控制:1,3-二醇骨架立体选择性合成的新方法摘要:31.30; H,5.22;N,36.52。发现:C,31.34;H,5.25;N,36.70。低分辨率质谱分析显示预期的分子离子位于 m/e 345。红外光谱在 2180 cm-' 处包含一个氰基拉伸峰。31P NMR 光谱由-7.8 ppm 的单峰组成。通过缓慢蒸发 2 在己烷中的溶液来生长适用于 X 射线分析的晶体。2 的结构如图 2 所示,重要的结构参数在表 I.9 中给出。这两种化合物 1 和 2 与大气接触时稳定,加热至中等温度时不受影响。1和2的X射线结构信息与氰基而不是异氰基排列一致,1°,红外光谱证实了这一点。” 2中氰基的三键比1中的略长,DOI:10.1021/ja00279a097
文献信息
-
Acylative Desymmetrization of Cyclic <i>meso</i>-1,3-Diols by Chiral DMAP Derivatives作者:Hiroki Mandai、Tsubasa Hironaka、Koichi Mitsudo、Seiji SugaDOI:10.1246/cl.200809日期:2021.3.5An efficient enantioselective acylative desymmetrization of cyclic meso-1,3-diols was developed by using a chiral DMAP derivative 1e having a 1,1′-binaphthyl unit. The reactions required only 0.5 m...
-
Novel, Cyclically Substituted Furopyrimidine Derivatives and Use Thereof申请人:Lampe Thomas公开号:US20110124665A1公开(公告)日:2011-05-26The present application relates to novel, cyclically substituted furopyrimidine derivatives, methods for their production, their use for the treatment and/or prophylaxis of diseases and their use for the production of medicinal products for the treatment and/or prophylaxis of diseases, in particular for the treatment and/or prophylaxis of cardiovascular diseases.
-
PROCESS FOR PREPARING CHLOROACETALDEHYDE ACETALS申请人:Wacker Chemie AG公开号:US20160272610A1公开(公告)日:2016-09-22The invention relates to a process for preparing chloroacetaldehyde acetals of monohydric, dihydric or higher-functionality aliphatic alcohols, in which the chloroacetaldehyde acetal is obtained from an aqueous chloroacetaldehyde solution in the presence of the alcohol to be acetalized and an acid catalyst by azeotropic removal of water with the aid of a solvent, wherein the solvent is a halogenated solvent.
-
Efficient and Practical Arene Hydrogenation by Heterogeneous Catalysts under Mild Conditions作者:Tomohiro Maegawa、Akira Akashi、Kiichiro Yaguchi、Yohei Iwasaki、Masahiro Shigetsura、Yasunari Monguchi、Hironao SajikiDOI:10.1002/chem.200900361日期:2009.7.13An efficient and practical arene hydrogenation procedure based on the use of heterogeneous platinum group catalysts has been developed. Rh/C is the most effective catalyst for the hydrogenation of the aromatic ring, which can be conducted in iPrOH under neutral conditions and at ordinary to medium H2 pressures (<10 atm). A variety of arenes such as alkylbenzenes, benzoic acids, pyridines, furans, are
-
Chiral-Substituted Poly-<i>N</i>-vinylpyrrolidinones and Bimetallic Nanoclusters in Catalytic Asymmetric Oxidation Reactions作者:Bo Hao、Medha J. Gunaratna、Man Zhang、Sahani Weerasekara、Sarah N. Seiwald、Vu T. Nguyen、Alex Meier、Duy H. HuaDOI:10.1021/jacs.6b12113日期:2016.12.28synthesized from l-amino acids. The polymers, particularly 17, were used to stabilize nanoclusters such as Pd/Au for the catalytic asymmetric oxidations of 1,3- and 1,2-cycloalkanediols and alkenes, and Cu/Au was used for C-H oxidation of cycloalkanes. It was found that the bulkier the C5 substituent in the pyrrolidinone ring, the greater the optical yields produced. Both oxidative kinetic resolution of (±)-1由L-氨基酸合成了在吡咯烷酮环的C5处含有不对称中心的一类新的聚-N-乙烯基吡咯烷酮。这些聚合物,特别是17,用于稳定纳米团簇,例如Pd/Au,用于1,3-和1,2-环烷二醇和烯烃的催化不对称氧化,而Cu/Au用于环烷烃的CH氧化。研究发现,吡咯烷酮环中的C5取代基体积越大,产生的光学产率越大。 (±)-1,3- 和 1,2-反式环烷二醇的氧化动力学拆分以及内消旋顺式二醇的去对称化均在水中的氧气气氛下用 0.15 mol% Pd/Au (3:1)-17 进行,提供优异的 (S)-羟基酮的化学和光学收率。在 30 psi 氧气水中,用 0.5 mol% Pd/Au (3:1)-17 氧化各种烯烃,得到 >93% ee 的二羟基化产物。 (R)-柠檬烯在 25 °C 下在 C-1,2-环烯官能团处发生氧化,产生 (1S,2R,4R)-二羟基柠檬烯 49,产率 92%。重要的是,环烷烃用乙腈中的
表征谱图
-
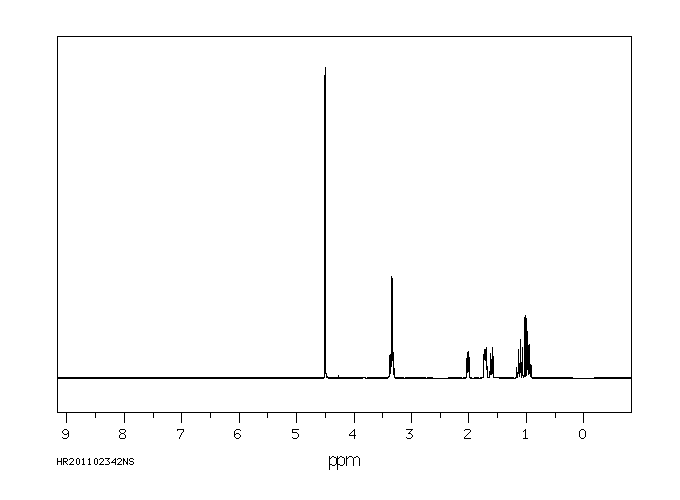
氢谱1HNMR
-
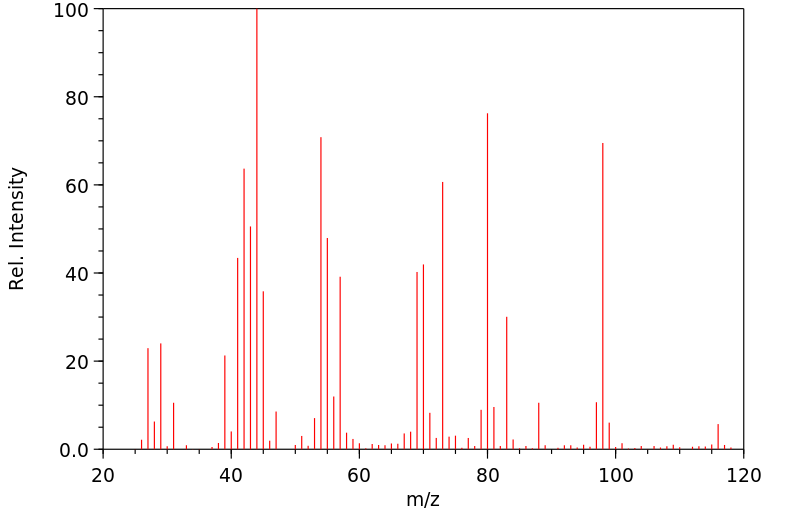
质谱MS
-
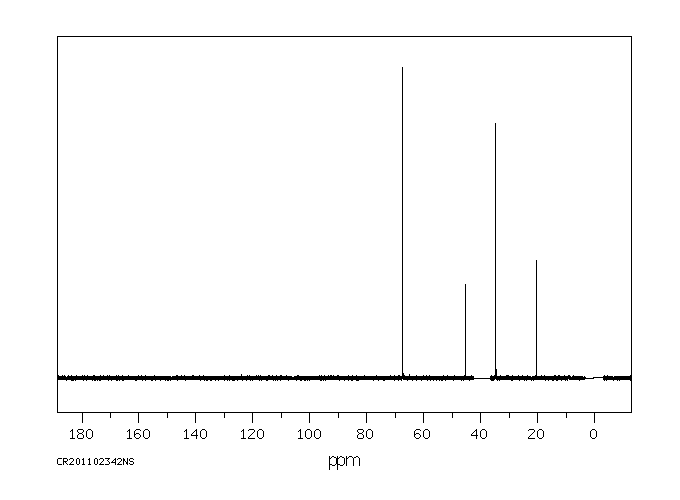
碳谱13CNMR
-
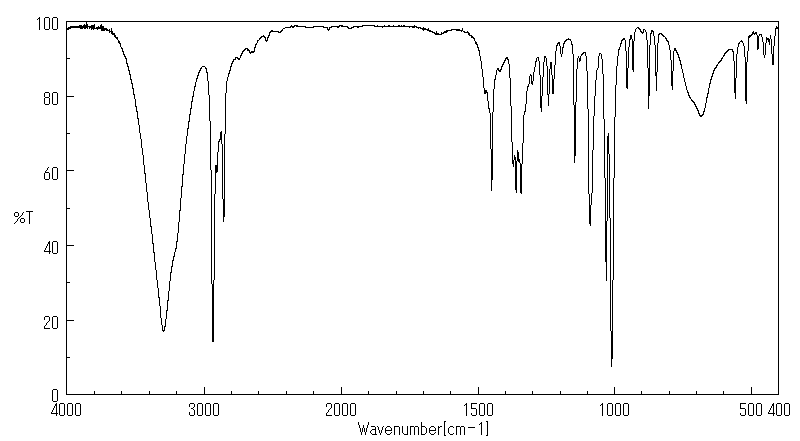
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(反式)-4-壬烯醛
(s)-2,3-二羟基丙酸甲酯
([1-(甲氧基甲基)-1H-1,2,4-三唑-5-基](苯基)甲酮)
(Z)-4-辛烯醛
(S)-氨基甲酸酯β-D-O-葡糖醛酸
(S)-3-(((2,2-二氟-1-羟基-7-(甲基磺酰基)-2,3-二氢-1H-茚满-4-基)氧基)-5-氟苄腈
(R)-氨基甲酸酯β-D-O-葡糖醛酸
(5,5-二甲基-2-(哌啶-2-基)环己烷-1,3-二酮)
(2,5-二氟苯基)-4-哌啶基-甲酮
龙胆苦苷
龙胆二糖甲乙酮氰醇(P)
龙胆二糖丙酮氰醇(P)
龙胆三糖
龙涎酮
齐罗硅酮
齐留通beta-D-葡糖苷酸
鼠李糖
黑芥子苷单钾盐
黑海棉酸钠盐
黑木金合欢素
黑曲霉三糖
黑介子苷
黄尿酸8-O-葡糖苷
麻西那霉素II
麦迪霉素
麦芽糖脎
麦芽糖基海藻糖
麦芽糖1-磷酸酯
麦芽糖
麦芽四糖醇
麦芽四糖
麦芽十糖
麦芽六糖
麦芽五糖水合物
麦芽五糖
麦芽五糖
麦芽五糖
麦芽三糖醇
麦芽三糖
麦芽三糖
麦芽三塘水合
麦芽七糖水合物
麦芽七糖
麦法朵
麦可酚酸-酰基-Β-D-葡糖苷酸
麦利查咪
麝香酮
鹤草酚
鸢尾酚酮 3-C-beta-D-吡喃葡萄糖苷
鸡矢藤苷