苦酮酸 | 550-74-3
中文名称
苦酮酸
中文别名
3-甲基-4-硝基-1-(对硝基苯)-2-吡唑啉酮-[5];3-甲-4-硝-1-對硝苯-5-吡唑啉酮;3-甲基-4-硝基-1-(对硝基苯基)-2-吡唑啉-5-酮;1-(对硝基苯)-3-甲基-4-硝基-5-吡唑啉酮;4-硝基-3-甲基对硝基苯基-5-吡唑啉酮;4-硝基-3-甲基-1-(对硝基苯)-5-吡唑啉酮;3-甲基-4-硝
英文名称
picrolonic acid
英文别名
Pikrolonsaeure;Picrolonsaeure;Pikrolonsaure;5-methyl-4-nitro-2-(4-nitrophenyl)-4H-pyrazol-3-one
CAS
550-74-3
化学式
C10H8N4O5
mdl
——
分子量
264.197
InChiKey
OVFUUSPKWADLNJ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:119 °C
-
沸点:548.8±50.0 °C(Predicted)
-
密度:1.65±0.1 g/cm3(Predicted)
-
溶解度:室温下在水中的浓度为0.27%,易溶于酒精。
-
稳定性/保质期:
计算性质
-
辛醇/水分配系数(LogP):1.2
-
重原子数:19
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.2
-
拓扑面积:124
-
氢给体数:0
-
氢受体数:6
安全信息
-
TSCA:Yes
-
危险品标志:Xn
-
安全说明:S26,S37/39
-
危险类别码:R68
-
海关编码:2933199090
-
RTECS号:UQ9510000
反应信息
-
作为反应物:描述:、 苦酮酸 以70%的产率得到参考文献:名称:Hein, F.; Schwartzkopff, O.; Hoyer, K., Berichte der Deutschen Chemischen Gesellschaft, 1928, vol. 61, p. 730 - 753摘要:DOI:
-
作为试剂:参考文献:名称:Aleksandrov, Yu. A.; Kolmakov, A. O.; Redoshkin, B. A., Journal of general chemistry of the USSR, 1988, vol. 58, # 10, p. 2071 - 2073摘要:DOI:
文献信息
-
Synthesis of substituted tetrahydropyridines and M-hydroxybenzoic acids作者:K. Clinch、C.J. Marquez、M.J. Parrott、R. RamageDOI:10.1016/0040-4020(89)80052-3日期:1989.14-dienylamines (10). The kinetics of these reactions have been investigated and an explanation of substituent effects is advanced. Also a new route to m-hydroxybenzoic acids has been established by cyclisation of substituted hexa-3,4:5,6-dienoic acids.一系列取代的1,2,3,6-(5)和1,2,5,6-四氢吡啶(6)的已合成经由分子内1,6-迈克尔加成甲氧基羰基-2,4- dienylamines的( 10)。已经研究了这些反应的动力学,并提出了取代基效应的解释。还到一个新的路由米5,6-二烯酸:对羟基酸已被取代的六- 3,4-环化建立。
-
N-substituted aporphines, a method of inducing emesis and a method of controlling psychosis utilising the same申请人:NORTHEASTERN UNIVERSITY公开号:EP0040074A1公开(公告)日:1981-11-18N-substituted aporphines of the formula wherein R, is lower alkyl, substituted lower alkyl, cycloalkyl, substituted cycloalkyl, lower alkenyl, substituted lower alkenyl, lower alkynyl, substituted lower alkynyl, phenyl lower alkyl, phenyl lower alkenyl and phenyl lower alkynyl, R2 is hydrogen or wherein R3 is lower alkyl and R4 is hydrogen or R20 and the acid addition salts thereof with the proviso that when R4 is hydrogen R1 is 2-chloroethyl. Representative compounds of this invention are potent emetics and dopamine antagonists.
-
1-((Benzoylphenyl) - lower-alkyl) piperidines and carbinol analogs and preparation thereof申请人:STERLING DRUG INC.公开号:EP0052311A1公开(公告)日:1982-05-261-[(3- or 4-Benzoylphenyl)-lower-alkyl]-[(CH2)n-N=B]-substituted-piperidines or α-hydroxy(3-or 4-benzyl), analogs thereof, useful as anti-asthmatic, anti-allergic, anti-cholinergic, bronchodilator and anti-inflammatory agents, are prepared by alkylation of an appropriate substituted piperidine with a (3- or 4-benzoyl-phenyl)-lower-alkyl halide or tosylate; by reaction of a 1-[2-(3- or 4-lithiophenyl)-lower-alkyl]-[(CH2)n-N=B]-substituted-piperidine with benzonitrile and hydrolysis of the resulting benzimidoyl compound; or by reduction of a 1-[a-(3- or 4-benzoylphenyl)-lower-alkanoyl]-[(CH2)n-N=B]-substituted-piperidine. The analogous carbinols are prepared by reduction, with an alkali metal borohydride, of the ketone.
-
Stable S-adenosylmethionine salts, the process for their preparation and therapeutic compositions which contain them as active principle申请人:BIORESEARCH S.p.A.公开号:EP0073376A1公开(公告)日:1983-03-09S-adenosylmethionine salts have been prepared wich are stable even at elevated temperatures for practically indefinite time periods, and which correspond to the formula in which X is the acid equivalent of a strong mineral acid of pK less than 2.5, and n is 4, 5 or 6. The new salts are practically free from toxicity, and find application in numerous fields of human therapy.
-
Coumarin derivatives, pharmaceutical compositions containing the same, and the use thereof in the treatment of cancer申请人:FIDIA S.p.A.公开号:EP0133766A2公开(公告)日:1985-03-06A compound having the formula: wherein R1 is naiogen or a substituted or unsubstituted hydrocarbon group; R2, R6 and R7, which may be the same or different, each represents hydrogen or a substituted or unsubstituted hydrocarbon group, or R6 and R7 take together may represent a hetero group having one or more hetero atoms; R3 and R4 which may be the same or different each represents a substituted or unsubstituted hydrocarbon group; or R3 and R4 taken together may represent an alkylene group or a hetero group having one or more hetero atoms and Rs is hydrogen, a substituted or unsubstituted hydrocarbon group, halogen or a free or protected hydroxy group, or a pharmaceutically acceptable salt thereof. The compounds have antiaggregating properties and some of them also have antitumor and antimetastatic activity.具有以下式子的化合物 其中 R1 为奈原或取代或未取代的烃基;R2、R6 和 R7 可以相同或不同,各自代表氢或取代或未取代的烃基,或 R6 和 R7 合在一起可代表具有一个或多个杂原子的杂基;R3 和 R4 可相同或不同,各自代表取代或未取代的烃基;或 R3 和 R4 合在一起可代表亚烷基或具有一个或多个杂原子的杂基,Rs 为氢、取代或未取代的烃基、卤素或游离或受保护的羟基,或其药学上可接受的盐。这些化合物具有抗聚集特性,其中一些还具有抗肿瘤和抗转移活性。
表征谱图
-
氢谱1HNMR
-
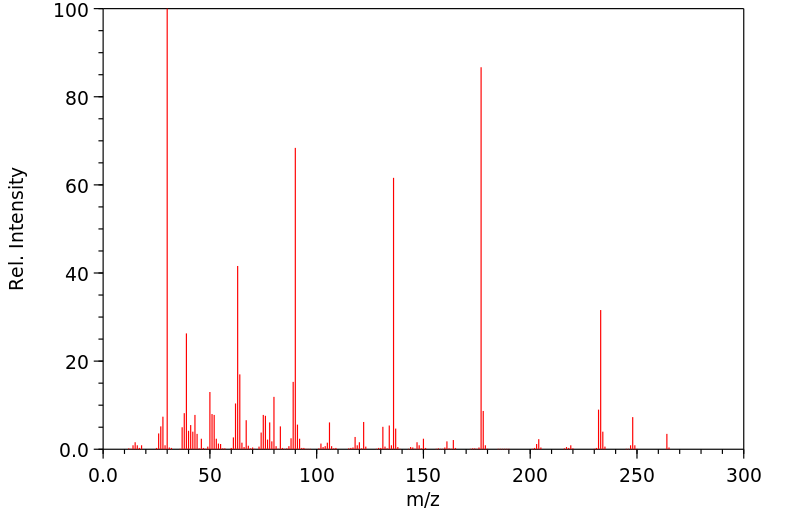
质谱MS
-
碳谱13CNMR
-
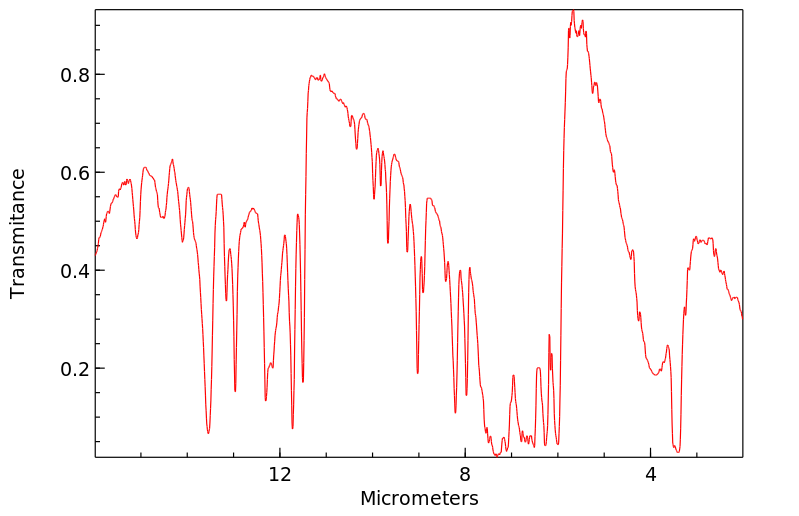
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(甲基3-(二甲基氨基)-2-苯基-2H-azirene-2-羧酸乙酯)
(±)-盐酸氯吡格雷
(±)-丙酰肉碱氯化物
(d(CH2)51,Tyr(Me)2,Arg8)-血管加压素
(S)-(+)-α-氨基-4-羧基-2-甲基苯乙酸
(S)-阿拉考特盐酸盐
(S)-赖诺普利-d5钠
(S)-2-氨基-5-氧代己酸,氢溴酸盐
(S)-2-[[[(1R,2R)-2-[[[3,5-双(叔丁基)-2-羟基苯基]亚甲基]氨基]环己基]硫脲基]-N-苄基-N,3,3-三甲基丁酰胺
(S)-2-[3-[(1R,2R)-2-(二丙基氨基)环己基]硫脲基]-N-异丙基-3,3-二甲基丁酰胺
(S)-1-(4-氨基氧基乙酰胺基苄基)乙二胺四乙酸
(S)-1-[N-[3-苯基-1-[(苯基甲氧基)羰基]丙基]-L-丙氨酰基]-L-脯氨酸
(R)-乙基N-甲酰基-N-(1-苯乙基)甘氨酸
(R)-丙酰肉碱-d3氯化物
(R)-4-N-Cbz-哌嗪-2-甲酸甲酯
(R)-3-氨基-2-苄基丙酸盐酸盐
(R)-1-(3-溴-2-甲基-1-氧丙基)-L-脯氨酸
(N-[(苄氧基)羰基]丙氨酰-N〜5〜-(diaminomethylidene)鸟氨酸)
(6-氯-2-吲哚基甲基)乙酰氨基丙二酸二乙酯
(4R)-N-亚硝基噻唑烷-4-羧酸
(3R)-1-噻-4-氮杂螺[4.4]壬烷-3-羧酸
(3-硝基-1H-1,2,4-三唑-1-基)乙酸乙酯
(2S,4R)-Boc-4-环己基-吡咯烷-2-羧酸
(2S,3S,5S)-2-氨基-3-羟基-1,6-二苯己烷-5-N-氨基甲酰基-L-缬氨酸
(2S,3S)-3-((S)-1-((1-(4-氟苯基)-1H-1,2,3-三唑-4-基)-甲基氨基)-1-氧-3-(噻唑-4-基)丙-2-基氨基甲酰基)-环氧乙烷-2-羧酸
(2S)-2,6-二氨基-N-[4-(5-氟-1,3-苯并噻唑-2-基)-2-甲基苯基]己酰胺二盐酸盐
(2S)-2-氨基-N,3,3-三甲基-N-(苯甲基)丁酰胺
(2S)-2-氨基-3-甲基-N-2-吡啶基丁酰胺
(2S)-2-氨基-3,3-二甲基-N-(苯基甲基)丁酰胺,
(2S)-2-氨基-3,3-二甲基-N-2-吡啶基丁酰胺
(2S,4R)-1-((S)-2-氨基-3,3-二甲基丁酰基)-4-羟基-N-(4-(4-甲基噻唑-5-基)苄基)吡咯烷-2-甲酰胺盐酸盐
(2R,3'S)苯那普利叔丁基酯d5
(2R)-2-氨基-3,3-二甲基-N-(苯甲基)丁酰胺
(2-氯丙烯基)草酰氯
(1S,3S,5S)-2-Boc-2-氮杂双环[3.1.0]己烷-3-羧酸
(1R,5R,6R)-5-(1-乙基丙氧基)-7-氧杂双环[4.1.0]庚-3-烯-3-羧酸乙基酯
(1R,4R,5S,6R)-4-氨基-2-氧杂双环[3.1.0]己烷-4,6-二羧酸
齐特巴坦
齐德巴坦钠盐
齐墩果-12-烯-28-酸,2,3-二羟基-,苯基甲基酯,(2a,3a)-
齐墩果-12-烯-28-酸,2,3-二羟基-,羧基甲基酯,(2a,3b)-(9CI)
黄酮-8-乙酸二甲氨基乙基酯
黄荧菌素
黄体生成激素释放激素(1-6)
黄体生成激素释放激素 (1-5) 酰肼
黄体瑞林
麦醇溶蛋白
麦角硫因
麦芽聚糖六乙酸酯
麦根酸